恒瑞创新药氟唑帕利联合阿帕替尼治疗卵巢癌和乳腺癌研究发表于BMC Medicine
2023-10-11
临床1期临床结果上市批准临床2期
内容来源:爱医时空

Preview
来源: 恒瑞医药
研究背景
氟唑帕利是恒瑞医药研发的1类创新药,是我国首个拥有知识产权的PARP抑制剂。在中国获批用于胚系BRCA1/2(gBRCA1/2)突变的铂敏感复发性卵巢癌治疗,以及铂类敏感复发性卵巢癌维持治疗,无论胚系BRCA1/2突变状态如何[11-13]。阿帕替尼是恒瑞医药历时10年研发的具有自主知识产权的1类创新药,是一种高选择性的靶向血管内皮细胞生长因子受体2(VEGFR2)的抗血管生成药物。它在卵巢癌和乳腺癌等多种实体肿瘤中均显示良好的活性和安全性[14-17]。
研究设计
研究结果
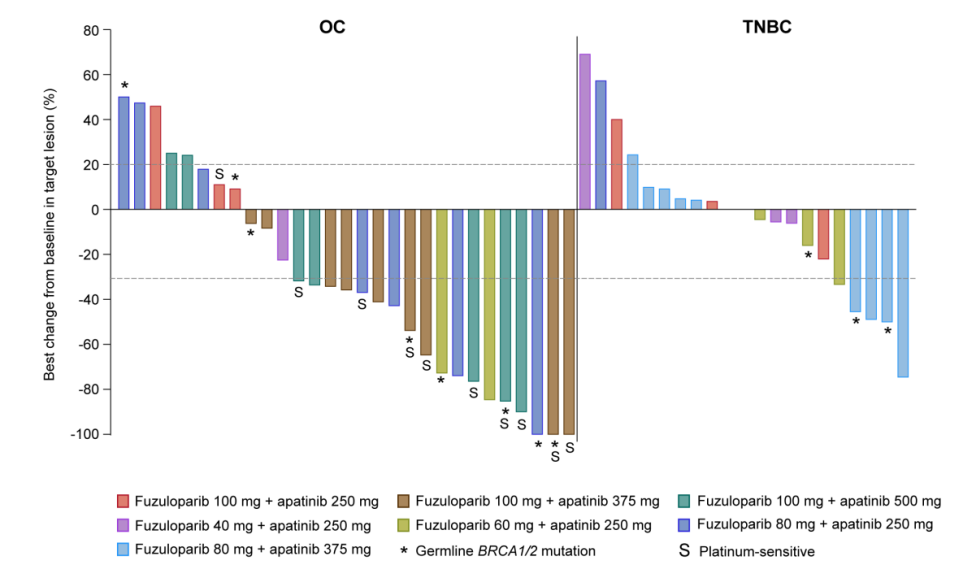
Preview
来源: 恒瑞医药
卵巢癌(OC)和三阴性乳腺癌(TNBC)靶病灶最佳反应
伴有gBRCA1/2突变的卵巢癌患者中位PFS为9.4个月,gBRCA野生型的卵巢癌患者中位PFS为6.7个月。伴有gBRCA1/2突变的三阴性乳腺癌患者的中位PFS为5.5个月,gBRCA野生型三阴性乳腺癌患者中位PFS为2.8个月。
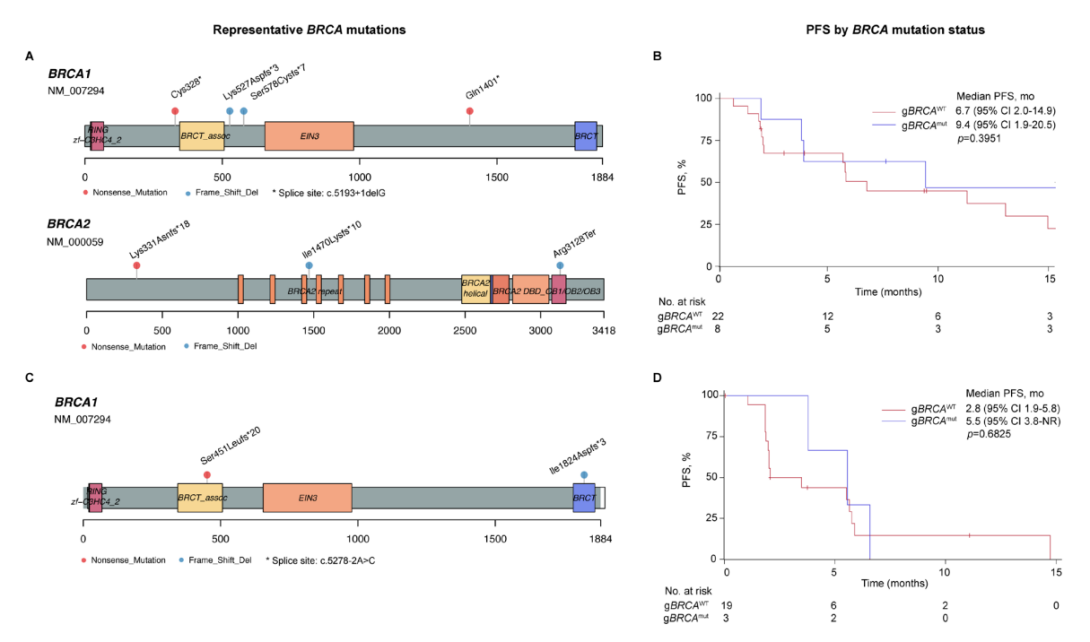
Preview
来源: 恒瑞医药
研究结论
氟唑帕利和阿帕替尼均是恒瑞医药自主研发的创新药,获批上市以来为广大肿瘤患者带去治疗新希望。作为中国首个自主研发的PARP抑制剂,氟唑帕利已获批上市两款适应症,今年8月,其新适应症上市申请获受理,用于晚期上皮性卵巢癌、输卵管癌或原发性腹膜癌一线维持治疗。除卵巢癌、乳腺癌外,氟唑帕利还在胰腺癌、前列腺癌等瘤种中开展相关研究,其单药或联合阿帕替尼的多个适应症开发已处在III期临床研究阶段,另有多种联合治疗方案,包括与阿比特龙、与SHR-A1811、与mFOLFIRINOX等联合治疗多种实体肿瘤已处于临床开发阶段。2014年10月获批上市的阿帕替尼,目前已有3个适应症获批,分别为单药用于晚期胃腺癌或胃-食管结合部腺癌、既往接受过至少一线系统性治疗后失败或不可耐受的晚期肝细胞癌、联合卡瑞利珠单抗用于不可切除或转移性肝细胞癌。前不久,卡瑞利珠单抗(艾瑞卡®)联合阿帕替尼(艾坦®)(“双艾”) 肝癌一线治疗适应症美国申报上市获FDA受理,该研究主论文全文在线发表于国际顶级医学期刊《柳叶刀》(The Lancet,IF:168.9)主刊上,这是肿瘤学领域中国学者主导的国际性III期临床研究首次问鼎《柳叶刀》主刊。
参考文献:
[1] Yaxin Liu, Wei Wang,et al. A phase 1 trial of fuzuloparib in combination with apatinib for advanced ovarian and triple negative breast cancer: efficacy, safety, pharmacokinetics and germline BRCA mutation analysis.
[2] Vanacker H, Harter P, Labidi-Galy SI, Banerjee S, Oaknin A, Lorusso D, et al. PARPinhibitors in epithelial ovarian cancer: Actual positioning and future expectations. Cancer Treat Rev. 2021; 99:102255.
[3] Mirza MR, Pignata S, Ledermann JA. Latest clinical evidence and further development of PARP inhibitors in ovarian cancer. Ann Oncol. 2018; 29:1366-76.
[4] Bianchini G, De Angelis C, Licata L, Gianni L. Treatment landscape of triple-negative breast cancer - expanded options, evolving needs. Nat Rev Clin Oncol. 2022; 19:91-113.
[5] Lim JJ, Yang K, Taylor-Harding B, Wiedemeyer WR, Buckanovich RJ. VEGFR3 inhibition chemosensitizes ovarian cancer stemlike cells through down-regulation of BRCA1 and BRCA2. Neoplasia. 2014; 16:343-353.e341-2.
[6] Bindra RS, Crosby ME, Glazer PM. Regulation of DNA repair in hypoxic cancer cells. Cancer Metastasis Rev. 2007; 26:249-60.
[7] Bindra RS, Gibson SL, Meng A, Westermark U, Jasin M, Pierce AJ, et al. Hypoxiainduced down-regulation of BRCA1 expression by E2Fs. Cancer Res. 2005; 65:11597-604.
[9] Mirza MR, Åvall Lundqvist E, Birrer MJ, dePont Christensen R, Nyvang GB, Malander S, et al. Niraparib plus bevacizumab versus niraparib alone for platinum-sensitive recurrent ovarian cancer (NSGO-AVANOVA2/ENGOT-ov24): a randomised, phase 2, superiority trial. Lancet Oncol. 2019; 20:1409-19.
[11] Li N, Bu H, Liu J, Zhu J, Zhou Q, Wang L, et al. An open-label, multicenter, single-arm, phase II study of fluzoparib in patients with germline BRCA1/2 mutation and platinumsensitive recurrent ovarian cancer. Clin Cancer Res. 2021; 27:2452-8.
[12] Li N, Zhang Y, Wang J, Zhu J, Wang L, Wu X, et al. Fuzuloparib maintenance therapy in patients with platinum-sensitive, recurrent ovarian carcinoma (FZOCUS-2): a multicenter, randomized, double-blind, placebo-controlled, phase III trial. J Clin Oncol. 2022:Jco2101511.
[13] Li H, Liu R, Shao B, Ran R, Song G, Wang K, et al. Phase I dose-escalation and expansion study of PARP inhibitor, fluzoparib (SHR3162), in patients with advanced solid tumors. Chin J Cancer Res. 2020; 32:370-82.
[14] Li J, Qin S, Xu J, Xiong J, Wu C, Bai Y, et al. Randomized, double-blind, placebocontrolled phase III trial of apatinib in patients with chemotherapy-refractory advanced or metastatic adenocarcinoma of the stomach or gastroesophageal junction. J Clin Oncol. 2016; 34:1448-54.
[15] Qin S, Li Q, Gu S, Chen X, Lin L, Wang Z, et al. Apatinib as second-line or later therapy in patients with advanced hepatocellular carcinoma (AHELP): a multicentre, doubleblind,randomised, placebo-controlled, phase 3 trial. Lancet Gastroenterol Hepatol. 2021; 6:559-68.
更多内容,请访问原始网站
文中所述内容并不反映新药情报库及其所属公司任何意见及观点,如有版权侵扰或错误之处,请及时联系我们,我们会在24小时内配合处理。
热门报告
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。



