恒瑞创新药达尔西利联合吡咯替尼晚期乳腺癌研究荣登《自然》子刊
2023-10-08
临床2期临床结果
近日,恒瑞医药创新药达尔西利联合吡咯替尼治疗HER2阳性晚期乳腺癌HER2阳性晚期乳腺癌的Ⅱ期探索性研究正式在线发表于Nature Communications(IF:16.6),该研究由河南省肿瘤医院闫敏教授发起。结果显示1:达尔西利联合吡咯替尼双口服方案治疗HER2阳性、任意HR状态的晚期一/二线乳腺癌HER2阳性、任意HR状态的晚期一/二线乳腺癌患者的客观缓解率(ORR)达70%,中位无进展生存期(mPFS)为11.0个月,安全性可控。该结果展示了达尔西利联合吡咯替尼在HER2阳性晚期乳腺癌HER2阳性晚期乳腺癌中的疗效,提示达尔西利在HER2阳性乳腺癌HER2阳性乳腺癌中的治疗潜力,为HER2阳性晚期乳腺癌HER2阳性晚期乳腺癌患者提供了新的治疗选择。

Preview
来源: 恒瑞医药
研究背景
Cyclin D-CDK4/6轴在细胞周期中发挥着关键作用,其调节异常是乳腺癌生物学的重要机制之一2。CDK4/6抑制剂通过抑制CDK/Cyclin复合物激酶的活性阻断细胞周期,已被证实能有效治疗激素受体(HR)阳性、HER2阴性乳腺癌HER2阴性乳腺癌3。而在HER2阳性乳腺癌HER2阳性乳腺癌中,CDK4/6同样是引人瞩目的靶点,它位于HER2信号通路的下游,促使肿瘤对抗HER2靶向治疗耐药4。已有研究发现,HER2阳性可致CDK4/6活性水平显著提高,这提示HER2阳性乳腺癌HER2阳性乳腺癌或能对CDK4/6抑制剂产生应答5。同时有临床前研究指出,CDK4/6抑制剂能抑制耐药及非耐药HER2阳性乳腺癌HER2阳性乳腺癌细胞系,并推迟HER2驱动的乳腺癌复发6。此外,动物实验发现,CDK4/6抑制剂能使肿瘤对抗HER2治疗增敏,并与抗HER2治疗产生协同作用4、6。临床试验已证实了CDK4/6抑制剂联合抗HER2治疗在HR阳性、HER2阳性晚期乳腺癌HER2阳性晚期乳腺癌中的疗效7、8,但该组合是否能使所有HER2阳性乳腺癌HER2阳性乳腺癌患者获益,这仍需进一步探索。
达尔西利是恒瑞医药自主研发的高度选择性小分子CDK4/6抑制剂,因其在III期随机试验(DAWNA-1)中表现出的出色疗效和可控安全性,在中国获批治疗HR阳性、HER2阴性晚期乳腺癌HER2阴性晚期乳腺癌9。吡咯替尼是恒瑞医药自主研发的靶向HER1、HER2、HER4的口服不可逆pan-ErbB受体酪氨酸激酶抑制剂(TKI),PHEOBE10和PHENIX11研究已证实了其疗效,该药已在中国获批治疗HER2阳性晚期乳腺癌HER2阳性晚期乳腺癌12。HR阳性、HER2阳性乳腺癌HER2阳性乳腺癌细胞系及动物模型显示,达尔西利与吡咯替尼双药组合可增强抗肿瘤作用。本研究旨在探索该双口服方案在无化疗或内分泌治疗的情况下在HER2阳性、任意HR状态晚期乳腺癌HER2阳性、任意HR状态晚期乳腺癌患者中的应用。
研究设计
本研究是一项单臂、单中心II期临床试验。主要入排标准为:组织学确认的HER2阳性晚期乳腺癌HER2阳性晚期乳腺癌;既往晚期阶段接受过不超过1个系统治疗方案;至少有一个符合RECIST1.1标准的可测量病灶;患者既往可接受过曲妥珠单抗治疗;排除有临床症状的脑转移患者。入组受试者接受达尔西利(125mg/d口服21天、间隔7天)联合吡咯替尼(400mg/d连续口服)治疗,每28天为一个周期,治疗直至疾病进展、不可接受的毒性、死亡、撤回知情同意或研究判断需结束治疗的其他情况。研究主要终点为ORR,次要终点为PFS、总体生存(OS)和安全性。
研究结果
共计41例患者入组研究并接受了至少一次研究治疗,1例患者因不符合入
Preview
来源: 恒瑞医药
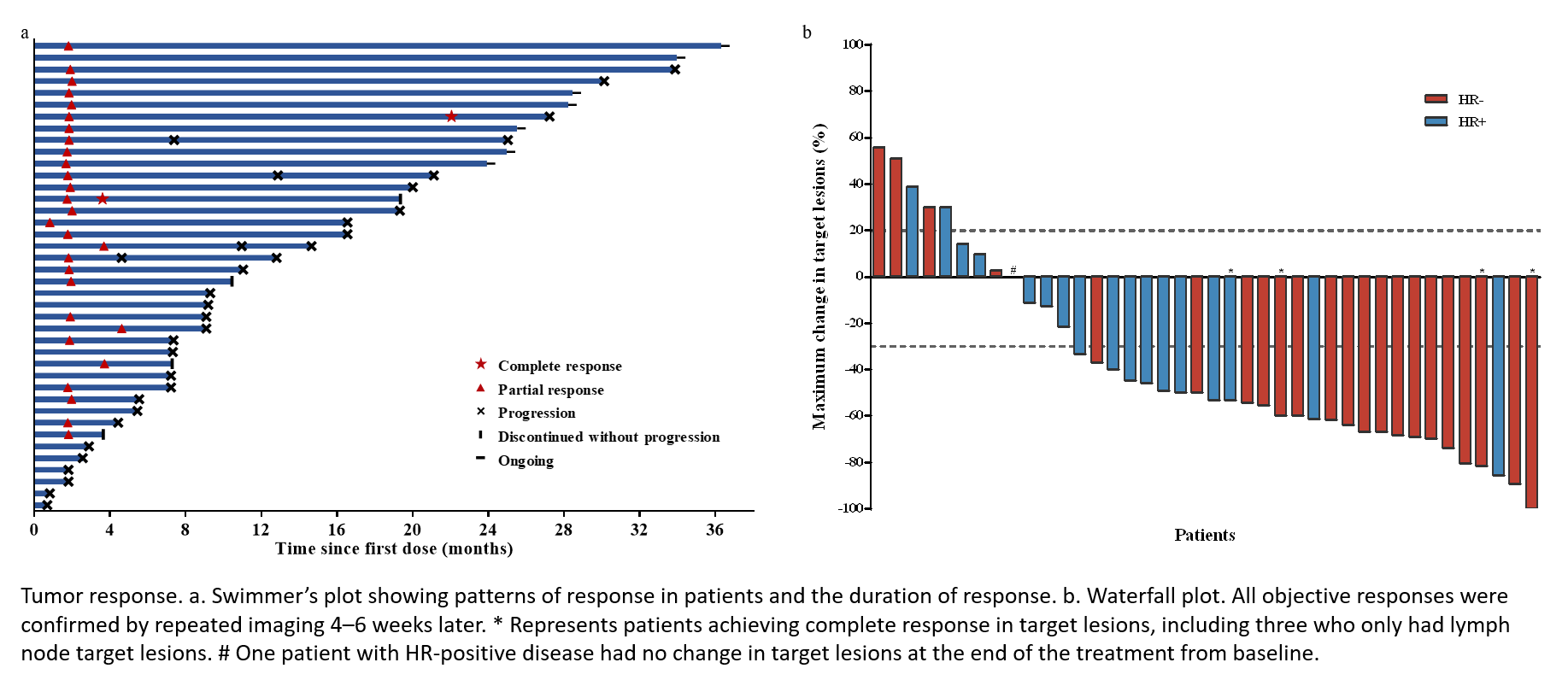
图1. 肿瘤缓解情况
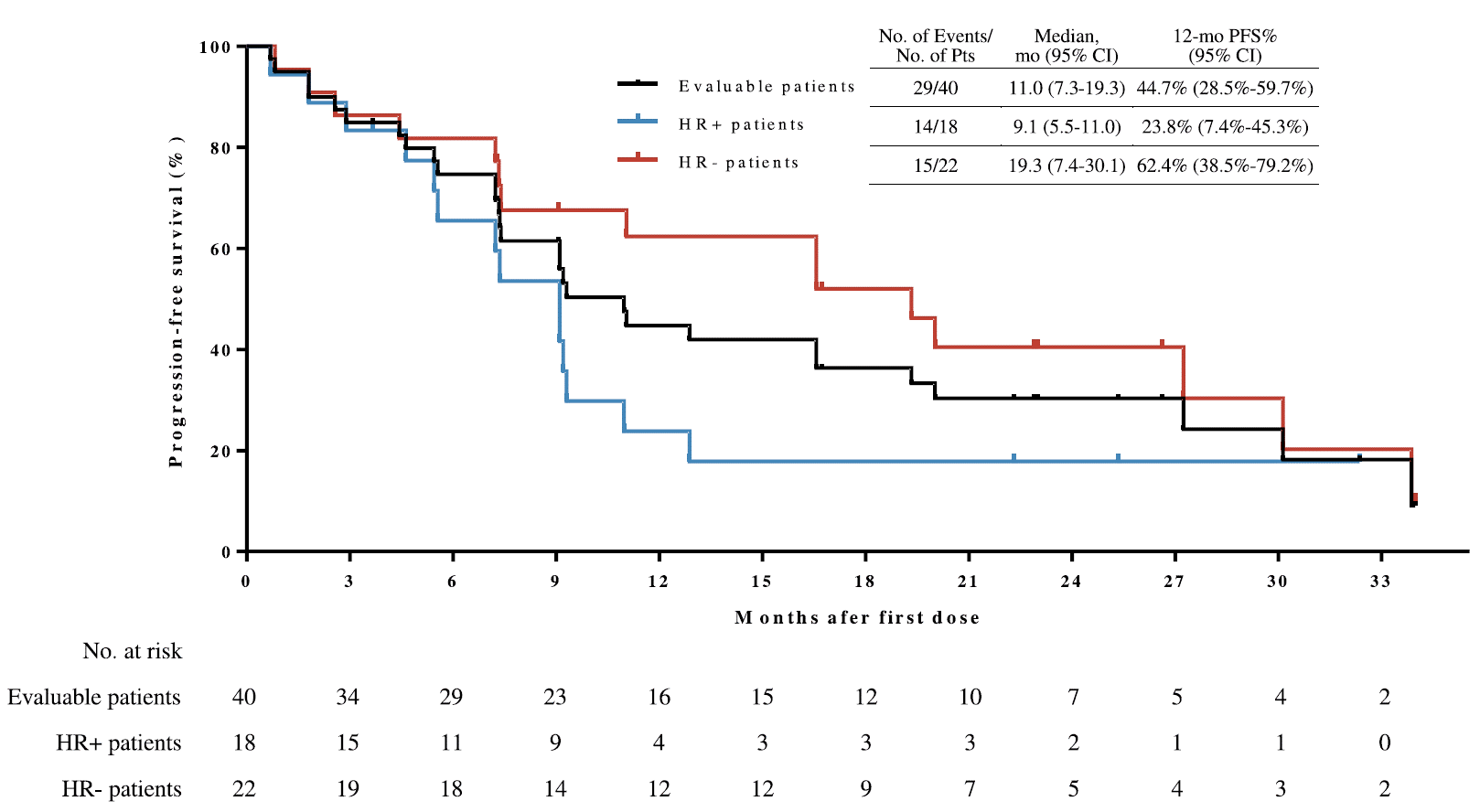
受试者的mPFS为11.0个月,12个月PFS率为44.7%。OS数据尚未成熟,预估的12个月和18个月OS率分别为90.0%和82.5%。研究整体安全性可控,无新的安全性信号。
Preview
来源: 恒瑞医药
图2. 无进展生存
非预设的事后亚组分析显示,HR阴性(81.8% vs 55.6%)和曲妥珠单抗敏感(80.0% vs 53.3%)的受试者ORR有更高的趋势,其PFS也趋于更长(分别为19.3 vs 9.1个月,和12.9 vs 9.1个月)。在13例基线具有无症状脑转移的患者中,整体ORR为84.6%,mPFS为11.0个月。
研究结论
在HER2阳性、任意HR状态的晚期乳腺癌HER2阳性、任意HR状态的晚期乳腺癌前线治疗中,达尔西利联合吡咯替尼显示了出色的疗效和可控的安全性,即使是具有无症状性活动性脑转移的患者也可获益。这一全口服方案为HER2阳性晚期乳腺癌HER2阳性晚期乳腺癌患者提供了另一种方便的用药选择。此外,该研究结果提示了CDK4/6抑制剂在(包括HR阴性、HER2阳性在内的)乳腺癌中的新的探索方向。
参考文献:
1.Min Y, et al. Dalpiciclib and pyrotinib in women with HER2-positive advanced breast cancer: a single-arm phase II trial. Nat Commun 14, 6272 (2023).
2.Spring LM, et al. Cyclin-dependent kinase 4 and 6 inhibitors for hormone receptor-positive breast cancer: past, present, and future. Lancet 395, 817-827 (2020).
3.George MA, et al. Clinical and Pharmacologic Differences of CDK4/6 Inhibitors in Breast Cancer. Front Oncol 11, 693104 (2021).
4.O'Sullivan CC, et al. The emerging role of CDK4/6i in HER2-positive breast cancer. Ther Adv Med Oncol 11, 1758835919887665 (2019).
5.Sinclair WD, et al. The Effects of HER2 on CDK4/6 Activity in Breast Cancer. Clin Breast Cancer 22, e278-e285 (2022).
6.Goel S, et al. Overcoming Therapeutic Resistance in HER2-Positive Breast Cancers with CDK4/6 Inhibitors. Cancer Cell 29, 255-269 (2016).
7.Tolaney SM, et al. Abemaciclib plus trastuzumab with or without fulvestrant versus trastuzumab plus standard-of-care chemotherapy in women with hormone receptor-positive, HER2-positive advanced breast cancer (monarcHER): a randomised, open-label, phase 2 trial. The Lancet Oncology 21, 763-775 (2020).
8.Spring LM, et al. Phase 1b clinical trial of ado-trastuzumab emtansine and ribociclib for HER2-positive metastatic breast cancer. NPJ Breast Cancer 7, 103 (2021).
9.Xu B, et al. Dalpiciclib or placebo plus fulvestrant in hormone receptor-positive and HER2-negative advanced breast cancer: a randomized, phase 3 trial. Nat Med 27, 1904-1909 (2021).
10.Xu B, et al. Pyrotinib plus capecitabine versus lapatinib plus capecitabine for the treatment of HER2-positive metastatic breast cancer (PHOEBE): a multicentre, open-label, randomised, controlled, phase 3 trial. Lancet Oncol 22, 351-360 (2021).
11.Yan M, et al. Pyrotinib plus capecitabine for human epidermal factor receptor 2-positive metastatic breast cancer after trastuzumab and taxanes (PHENIX): a randomized, double-blind, placebo-controlled phase 3 study. Transl Breast Cancer Res. 1, 13 (2020).
更多内容,请访问原始网站
文中所述内容并不反映新药情报库及其所属公司任何意见及观点,如有版权侵扰或错误之处,请及时联系我们,我们会在24小时内配合处理。
热门报告
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。



