预约演示
更新于:2025-09-06
Exenatide
艾塞那肽
更新于:2025-09-06
概要
基本信息
最高研发阶段批准上市 |
首次获批日期 美国 (2005-04-28), |
最高研发阶段(中国)批准上市 |
特殊审评孤儿药 (欧盟) |
登录后查看时间轴
结构/序列
Sequence Code 751

来源: *****
关联
360
项与 艾塞那肽 相关的临床试验CTIS2024-519014-31-00
- CHDR2418
开始日期2025-03-12 |
NCT02586831
A Pilot, Safety and Feasibility Trial of Anti-Thymocyte Globulin (ATG), Low Dose Interleukin-2 (IL-2), Adalimumab and Exenatide in the Treatment of New-Onset Type 1 Diabetes
To assess whether there is a difference in endogenous insulin secretion, measured as stimulated C-peptide secretion (area under the curve during a 4-hour mixed meal tolerance test), at the 1 year visit, for study subjects receiving combinational therapy versus those receiving placebo. The study will also examine the effect of the proposed treatments on immunological outcomes, specifically proportion of regulatory T cells at the 1 year visit.
开始日期2025-01-01 |
申办/合作机构  University of Miami University of Miami [+1] |
NCT05663515
EXCEED - A Pan-European Post-Authorisation Safety Study: Risk of Pancreatic Cancer Among Type 2 Diabetes Patients Who Initiated Exenatide as Compared With Those Who Initiated Other Non-Glucagon-Like Peptide 1 Receptor Agonists Based Glucose Lowering Drugs
EXCEED is a non-interventional post-authorisation safety study aiming to assess the risk of developing pancreatic cancer among type 2 diabetes mellitus (T2DM) patients who initiated exenatide compared to those who initiated other non-glucagon like peptide 1 receptor agonists (GLP-1 RA) based glucose lowering drugs (GLDs). Study data will be collected from secondary data sources across 7 European countries. The study will be conducted as a multi-country, long-term, retrospective, observational database study. Initiators of exenatide will be matched to initiators of non-GLP-1 RA based GLDs (comparator group) based on propensity score and calendar period of study entry. All analyses for pancreatic cancer will be conducted in the matched study population using an "intention-to-treat" approach. The study will use information from 8 data sources in 7 European countries (France, Spain, The United Kingdom, Finland, Denmark, Norway, and Sweden). Patients with T2DM, aged 18 years or older, who initiated treatment with exenatide or non-GLP-1 RA based GLDs during the study period, 2006 to 2023, will be included. Exposure to exenatide and non-GLP-1 RA based GLDs will be ascertained from recordings of prescriptions or insurance claims registrations as available in the different data sources. The outcome of pancreatic cancer will be defined as a primary diagnosis of pancreatic cancer during follow-up.
开始日期2024-09-30 |
申办/合作机构  AstraZeneca PLC AstraZeneca PLC [+1] |
100 项与 艾塞那肽 相关的临床结果
登录后查看更多信息
100 项与 艾塞那肽 相关的转化医学
登录后查看更多信息
100 项与 艾塞那肽 相关的专利(医药)
登录后查看更多信息
4,209
项与 艾塞那肽 相关的文献(医药)2025-11-01·NEUROPHARMACOLOGY
Exendin-4 protects the dopaminergic neurons by attenuating inflammatory responses of microglial cells via activation of AMPK
Article
作者: Khanal, Shristi ; Rimal, Nikesh ; Bohara, Ganesh ; Choi, Dong-Young
Parkinson's disease (PD) is a neurodegenerative disease characterized by preferential loss of the dopaminergic neurons in the substantia nigra and consequent occurrence of typical symptoms including resting tremor, rigidity and bradykinesia. PD remains a significant challenge due to the lack of disease-modifying drugs despite extensive research efforts. Glucagon-like peptide-1 and its analogues have shown neuroprotective properties. However, its specific molecular mechanisms for the neuroprotection remains to elucidate. In this study, we explored the anti-inflammatory and neuroprotective effects of exendin-4 by employing BV2 microglial cells and the MPTP-induced mouse model for PD. Our study showed that exendin-4 significantly suppressed the production of pro-inflammatory cytokines, including TNF-α, IL-1β, and IL-6 in LPS-stimulated BV2 cells. Furthermore, exendin-4 increased AMPK phosphorylation and the activation of AMPK by exendin-4 played a crucial role in suppressing the production of proinflammatory cytokines, as addition of compound C, an inhibitor of AMPK diminished the effect. Additionally, exendin-4 demonstrated neuroprotective effects by attenuating SH-SY5Y cell death caused by conditioned media from BV2 cells exposed to LPS. Moreover, exendin-4 attenuated microglial and astroglial activation in the substantia nigra and striatum of MPTP-treated mice, which was accompanied by reduced proinflammatory cytokines. Exendin-4 also improved motor functions of MPTP-treated mice, as determined by beam test and rotarod test. In parallel with the anti-inflammatory effects, exendin-4 attenuated MPTP-mediated dopaminergic neurodegeneration as estimated by immunohistochemistry for tyrosine hydroxylase and HPLC analyses for the striatal dopamine and DOPAC. These results suggest that exendin-4 could be a promising therapeutic agent for PD, offering neuroprotection by modulating inflammatory pathways and preserving dopaminergic neurons.
2025-11-01·NEUROPHARMACOLOGY
Effects of glucagon-like peptide 1 receptor signaling in the dorsolateral septum on ethanol operant self-administration and relapse behaviors
Article
作者: Xiao, Yuqing ; Yap, Jo Ann ; Ong, Zhi Yi
Alcohol Use Disorder (AUD) is a chronic relapsing disorder affecting more than 400 million people globally. Glucagon-like peptide-1 (GLP-1) has recently shown promise as a treatment for AUD but the underlying neural mechanisms are unclear. Given that dorsolateral septum (dLS) highly expresses GLP-1 receptors and is implicated in reward processing, this study investigated whether GLP-1 reduces ethanol intake and relapse behaviors via dLS. Long Evans rats were given intermittent access to 20 % ethanol solution before receiving training for ethanol operant self-administration. To examine relapse, operant self-administration was extinguished and two relapse tests were conducted: reacquisition of ethanol self-administration and prime + context-induced reinstatement of ethanol seeking. On self-administration test days, rats received intra-dLS injections of GLP-1 receptor agonist Exendin 4 (Ex4; 5 ng, 25 ng) or vehicle. During relapse tests, rats received intra-dLS infusion of either vehicle or 25 ng Ex4. Food intake and body weight were also measured. Results showed that intra-dLS Ex4 (25 ng) reduced ethanol self-administration in male high responders, but not in male low responders or females. Thus, only male rats were included in subsequent relapse tests. Intra-dLS Ex4 also reduced reacquisition of ethanol self-administration but had no impact on prime + context-induced reinstatement. No sustained changes in 24-h food intake or body weight were observed following intra-dLS Ex4 infusions. Finally, retrograde tracing results showed that dLS receive input from NTS GLP-1-expressing neurons. Together, these findings support the dLS as a key region in mediating the effects of GLP-1 receptor signaling on ethanol self-administration and reacquisition but suggest different contributing mechanisms in reinstatement of ethanol seeking.
2025-09-01·MEDICINA CLINICA
Review
作者: Alonso, Silvia Patricia ; Valdes, Sergio ; Doulatram-Gamgaram, Viyey Kishore
Obesity and type 2 diabetes mellitus (DM2) are interrelated global problems affecting 988 and 537 million people, respectively, figures that are continuously increasing. Obesity is a key factor in DM2, as it intensifies insulin resistance and β-cell dysfunction, accelerating disease progression. Weight loss improves glycemic control, reduces the occurrence of comorbidities and, in some cases, induces diabetes remission. In this regard, GLP-1 receptor agonists have revolutionized the treatment of DM2. Since the introduction of exenatide in 2005, more effective drugs have been developed that improve glycemic control, reduce weight, and decrease the occurrence of cardiovascular events. This article discusses the mechanisms of action and efficacy of these agents, highlighting recent advances such as polyagonists and oral formulations.
365
项与 艾塞那肽 相关的新闻(医药)2025-09-04
NPM-139 has potential to provide Wegovy®-level efficacy with once or twice-yearly administration First-in-human Phase 1 study is expected to initiate in the first half of 2026, pending regulatory clearance Preparations for dose-ranging Phase 2 study of NPM-139 to occur in parallel with Phase 1 study ALAMEDA, Calif., Sept. 04, 2025 (GLOBE NEWSWIRE) -- Vivani Medical, Inc. (Nasdaq: VANI) (“Vivani” or the “Company”), a clinical-stage biopharmaceutical company developing novel, ultra long-acting drug implants, today announced plans to initiate a Phase 1 clinical study in the NPM-139 semaglutide implant program in the first half of 2026, pending regulatory clearance. The Company is also preparing to initiate a Phase 2 clinical study of NPM-139 pending enabling results from the Phase 1 study and regulatory feedback. The NPM-139 clinical program will evaluate the Company’s investigational semaglutide implant for chronic weight management in patients who are either obese or overweight with a related comorbidity. “Recent promising results from LIBERATE-1™, the first-in-human study of NPM-115, an exenatide implant utilizing NanoPortal™ technology, combined with positive preclinical weight loss data with NPM-139, compel us to move forward aggressively with clinical development of NPM-139. The Phase 1 study, designed to evaluate the safety, tolerability and pharmacokinetic profile of the semaglutide implant and to enable a Phase 2 efficacy study, is expected to initiate in the first half of 2026, pending regulatory clearance,” said Vivani President and Chief Executive Officer Adam Mendelsohn, Ph.D. “The overall GLP-1 market in weight management continues to grow at an impressive rate, primarily because of considerable unmet medical needs and the fact that current GLP-1 options have a much better safety and efficacy profile compared to medications previously approved to treat obesity. We believe that NPM-139 could provide clear and compelling differentiation due to its potential to provide Wegovy®-level efficacy with convenient once or twice-yearly administration. Having recently obtained approval for the treatment of metabolic dysfunction-associated steatohepatitis (MASH) and with an expected near-term Phase 3 readout in the treatment of Alzheimer’s disease, semaglutide-based products continue to demonstrate tremendous potential to treat chronic diseases, and we believe the therapeutic potential of semaglutide could be significantly enhanced with an option that enables once or twice-yearly administration.” Dr. Mendelsohn continued, “If clinically validated and approved, we expect our products to stand out in a crowded and competitive field as the only available implant which could potentially enable providers to address new and currently underserved market segments. In addition, we continue to believe that NPM-139 could demonstrate improved tolerability relative to currently available oral or injectable GLP-1 medications, both because of the avoidance of drug fluctuations at each administration of oral or injectable options which provide each dose as a bolus, and by eliminating missed doses that further exacerbate the dosing fluctuations which are largely responsible for the gastrointestinal side effects of GLP-1 treatments.” Clinical Program for Semaglutide Implants for Chronic Weight Management The first clinical study of NPM-139 is planned as a small Phase 1 randomized, controlled investigation of the safety, tolerability and pharmacokinetic profile of the semaglutide NanoPortal implant in GLP-1 naïve obese or overweight subjects. The planned control arm is low-dose Wegovy injected weekly. The semaglutide implant to be used in this study is expected to produce comparable semaglutide exposure levels to low dose Wegovy injections. This study is expected to be initiated in the first half of 2026, pending regulatory clearance. The design of the second clinical study will be finalized pending results from the Phase 1 study but is anticipated to be a Phase 2, randomized, placebo-controlled, dose-ranging investigation over 4 to 6 months to evaluate the effects of the semaglutide NanoPortal implant on weight management in obese or overweight subjects. This study will provide safety, efficacy, and dose selection information for Phase 3 and will initiate shortly after Phase 1 completes, if warranted by the Phase 1 results and pending regulatory clearance. About Vivani Medical, Inc. Leveraging its proprietary NanoPortal™ platform, Vivani develops biopharmaceutical implants designed to deliver drug molecules steadily over extended periods of time with the goal of guaranteeing adherence and improving patient tolerance to their medication. Vivani is developing a portfolio of GLP-1 based implants for chronic weight management and other metabolic diseases including type 2 diabetes. These NanoPortal implants are designed to provide patients with the opportunity to realize the full potential benefit of their medication by avoiding the numerous challenges associated with the daily or weekly administration of orals and injectables, including tolerability issues and loss of efficacy. Medication non-adherence occurs when patients do not take their medication as prescribed. This affects an alarming number of patients, approximately 50%, including those taking daily pills. Forward-Looking Statements This press release contains certain “forward-looking statements” within the meaning of the “safe harbor” provisions of the US Private Securities Litigation Reform Act of 1995. Forward-looking statements can be identified by words such as: “target,” “believe,” “expect,” “will,” “may,” “anticipate,” “estimate,” “would,” “positioned,” “future,” and other similar expressions that in this press release, including statements regarding Vivani’s business, products in development, including the therapeutic potential thereof, the planned development therefor, the completion of the LIBERATE-1 Phase 1 study and reporting of study results, Vivani’s emerging development plans for NPM-139, NPM-115 or Vivani’s plans with respect its technology, strategy, cash position and financial runway. Forward-looking statements are neither historical facts nor assurances of future performance. Instead, they are based only on Vivani’s current beliefs, expectations, and assumptions. Because forward-looking statements relate to the future, they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict and many of which are outside of Vivani’s control. Actual results and outcomes may differ materially from those indicated in the forward-looking statements. Therefore, you should not rely on any of these forward-looking statements. Important factors that could cause actual results and outcomes to differ materially from those indicated in the forward-looking statements include, among others, risks related to the development and commercialization of Vivani’s products, including NPM-139 and NPM-115; delays and changes in the development of Vivani’s products, including as a result of applicable laws, regulations and guidelines, potential delays in submitting and receiving regulatory clearance or approval to conduct Vivani’s development activities, including Vivani’s ability to commence clinical development of NPM-139; risks related to the initiation, enrollment and conduct of Vivani’s planned clinical studies and the results therefrom; or Vivani’s history of losses and Vivani’s ability to access additional capital or otherwise fund Vivani’s business. There may be additional risks that the Company considers immaterial, or which are unknown. A further list and description of risks and uncertainties can be found in the Company’s most recent Annual Report on Form 10-K filed with the U.S. Securities and Exchange Commission on March 31, 2025, as updated by the Company’s subsequent Quarterly Reports on Form 10-Q. Any forward-looking statement made by Vivani in this press release is based only on information currently available to the Company and speaks only as of the date on which it is made. The Company undertakes no obligation to publicly update any forward-looking statement, whether written or oral, that may be made from time to time, whether as a result of added information, future developments or otherwise, except as required by law. Company Contact:Donald DwyerChief Business Officerinfo@vivani.com(415) 506-8462 Investor Relations Contact:Jami TaylorInvestor Relations Advisorinvestors@vivani.com(415) 506-8462 Media Contact:Sean LeousSenior Vice PresidentICR HealthcareSean.Leous@ICRHealthcare.com(646) 866-4012
临床1期临床2期临床申请
2025-08-29
Ajovy (fremanezumab-vfrm)
Emgality (galcanezumab-gnlm)
Nurtec ODT (rimegepant)
Ubrelvy (ubrogepant)
Vyepti (eptinezumab-jjmr)
Hypertension
The "Warnings and Precautions", "Adverse Reactions", "Patient Counseling Information", and "Patient Information" sections of the labeling were updated in March 2025 to include information about the risk of hypertension.
Example:
Ajovy labeling
Apriso (mesalamine)
Asacol (mesalamine)
Asacol HD (mesalamine)
Azulfidine (sulfasalazine)
Azulfidine EN-Tabs (sulfasalazine)
Canasa (mesalamine)
Colazal (balsalazide disodium)
Delzicol (mesalamine)
Dipentum (osalazine sodium)
Giazo (balsalazide disodium)
Lialda (mesalamine)
Rowasa (mesalamine)
Pentasa (mesalamine)
Severe cutaneous adverse reactions
The Warnings and Precautions section of the labeling was updated November 2021 to include severe cutaneous adverse reactions.
Example:
Delzicol labeling
Avastin (bevacizumab)
Mvasi (bevacizumab-awwb)
Zirabev (bevacizumab-bvzr)
Pancreatitis
FDA determined that no action is necessary at the time based on available information.
Avastin (bevacizumab)
Mvasi (bevacizumab-awwb)
Zirabev (bevacizumab-bvzr)
Anaphylactic shock
The "Warnings and Precautions" section of the labeling was updated between September 2022 and February 2023 to include anaphylactoid/anaphylactic reactions.
Example:
Avastin labeling
Ayvakit (avapritinib)
Photosensitivity reaction
The "Warnings and Precautions", "Adverse Reactions", "Patient Counseling Information", and "Patient Information" sections of the labeling were updated in March 2023 to include information about photosensitivity.
Ayvakit labeling
Azacitidine
Onureg (azacitidine)
Vidaza (azacitidine)
Generic products containing azacitidine
Pericarditis
The "Adverse Reactions" section of the labeling was updated between September 2022 and June 2023 to include pericarditis.
Example:
Vidaza labeling
FDA determined that no action is necessary at the time based on available information for Onureg.
Bavencio (avelumab)
Imfinzi (durvalumab)
Libtayo (cemiplimab-rwlc)
Keytruda (pembrolizumab)
Opdivo (nivolumab)
Tecentriq (atezolizumab)
Yervoy (ipilimumab)
Tumour lysis syndrome
FDA determined that no action is necessary at the time based on available information.
Cymbalta (duloxetine hydrochloride)
Drizalma Sprinkle (duloxetine hydrochloride)
Generic products containing duloxetine hydrochloride
Anosmia, Hyposmia
This issue is posted as two individual newly identified safety signal (NISS) entries in the January-March 2022 quarter, to reflect the focus on the potential signals of anosmia and hyposmia, separately.
Darzalex (daratumumab)
Darzalex Faspro (daratumumab and hyaluronidase-fihj)
Blood stem cell transplant failure
FDA determined that no action is necessary at the time based on available information.
Dipeptidyl peptidase-4 (DPP-4) inhibitors
Glyxambi (empagliflozin and linagliptin)
Janumet (metformin hydrochloride and sitagliptin phosphate)
Janumet XR (metformin hydrochloride and sitagliptin phosphate)
Jentadueto (linagliptin and metformin hydrochloride)
Jentadueto XR (linagliptin and metformin hydrochloride)
Kazano (alogliptin and metformin hydrochloride)
Kombiglyze XR (metformin hydrochloride and saxagliptin)
Nesina (alogliptin)
Onglyza (saxagliptin)
Oseni (alogliptin and pioglitazone)
Steglujan (ertugliflozin and sitagliptin)
Tradjenta (linagliptin)
Trijardy XR (empagliflozin, linagliptin and metformin hydrochloride)
Qtern (saxagliptin and dapagliflozin)
Generic products containing dipeptidyl peptidase-4 inhibitors
Tubulointerstitial nephritis
The "Adverse Reactions" section of the labeling was updated between March 2022 and June 2022 for products containing alogliptin and sitagliptin to include tubulointerstitial nephritis.
Example:
Janumet labeling
FDA determined that no action is necessary for products containing linagliptin and saxagliptin at the time based on available information.
*An administrative error resulted in the omission of Januvia from the list of product names after the initial quarterly report was posted.
Dupixent (dupilumab)
Angioedema
The "Warnings and Precautions" and "Adverse Reactions" sections of the labeling were updated in December 2021 to include angioedema.
Dupixent labeling
Glucagon-like peptide-1 (GLP-1) analogues
Adlyxin (lixisenatide)
Bydureon (exenatide)
Bydureon BCise (exenatide)
Byetta (exenatide)
Ozempic (semaglutide)
Rybelsus (semaglutide)
Saxenda (liraglutide)
Soliqua 100/33 (insulin glargine and lixisenatide injection)
Trulicity (dulaglutide)
Victoza (liraglutide)
Wegovy (semaglutide)
Xultophy 100/3.6 (insulin degludec and liraglutide injection)
Generic products containing glucagon-like peptide-1 analogues
Gallbladder related disorders
The "Warnings and Precautions", "Adverse Reactions", and "Patient Counseling Information" sections of the GLP-1 analogues labeling were updated between March 2022 and June 2022 to include information about acute gallbladder disease.
Example:
Adlyxin labeling
FDA has determined that the last approved labeling for Saxenda and Wegovy is adequately labeled for acute gallbladder disease, and that no further regulatory action is needed at this time.
Gonadotropin releasing hormone (GnRH) analogues
Fensolvi (leuprolide acetate)
Lupron Depot-PED (leuprolide acetate)
Supprelin LA (histrelin acetate)
Synarel (nafarelin acetate)
Triptodur (triptorelin)
Idiopathic intracranial hypertension
The "Warnings", "Precautions", and "Adverse Reactions" sections of the labeling were updated in April 2022 to include idiopathic intracranial hypertension.
Example:
Fensolvi labeling
Jakafi (ruxolitinib phosphate)
Herpes simplex virus (HSV) reactivation and/or disseminated HSV
The "Dosage and Administration", "Warnings and Precautions", and "Adverse Reactions" sections of the labeling were updated in January 2023 to include information about herpes simplex and herpes zoster infections.
Jakafi labeling
Ibrance (palbociclib)
Kisqali (ribociclib)
Kisqali Femara Co-Pack (letrozole and ribociclib)
Embolic and thrombotic events, venous
The "Adverse Reactions" section of the Ibrance labeling was updated in December 2024 to include venous thromboembolism.
Ibrance labeling
FDA determined that no action is necessary at the time for Kisqali and Kisqali Femara Co-Pack based on available information.
Ocrevus (ocrelizumab)
Myocardial infarction
FDA determined that no action is necessary at the time based on available information.
Potassium Chloride
Product preparation error
The “Warnings” section of the labeling was updated in December 2022 to include information about fatal cardiac adverse reactions with intravenous administration of undiluted potassium chloride.
Praxbind (idarucizumab)
Wrong dose errors
The carton labeling and "Dosage and Administration" sections of the labeling were revised in February 2022 to clarify the number of vials included in each carton and the recommended dosing.
Praxbind labeling
Qbrexza (glycopyrronium tosylate)
Accidental exposure to product
The container label, carton labeling, "Dosage and Administration", "Warnings and Precautions", "Patient Counseling Information", and "Patient Information" sections of the labeling were updated in October 2022 to include information about accidental exposure.
Qbrexza labeling
Qbrexza (glycopyrronium tosylate)
Urinary retention
The “Warnings and Precautions”, “Adverse Reactions”, and “Patient Counseling Information” sections of the labeling were updated in October 2022 to include information about new or worsening urinary retention.
Qbrexza labeling
Sodium-glucose cotransporter-2 (SGLT-2) inhibitors
Farxiga (dapagliflozin)
Glyxambi (empagliflozin and linagliptin)
Invokamet (canagliflozin and metformin hydrochloride)
Invokamet XR (canagliflozin and metformin hydrochloride)
Invokana (canagliflozin)
Jardiance (empagliflozin)
Qtern (saxagliptin and dapagliflozin)
Steglatro (ertugliflozin)
Steglujan (ertugliflozin and sitagliptin)
Segluromet (ertugliflozin and metformin hydrochloride)
Synjardy (empagliflozin and metformin hydrochloride)
Synjardy XR (empagliflozin and metformin hydrochloride)
Trijardy XR (empagliflozin, linagliptin and metformin hydrochloride)
Xigduo XR (dapagliflozin and metformin)
Generic products containing sodium-glucose cotransporter-2 (SGLT-2) inhibitors
Tubulointerstitial nephritis
FDA determined that no action is necessary at the time based on available information.
Somatostatin Receptor Imaging Agents
Detectnet (copper Cu-64 dotatate injection)
Gallium Dotatoc (GA 68)
Octreoscan (Indium In 111 Pentetreotide)
Netspot (Gallium GA 68 Dotatate)
Hypersensitivity
The "Warnings and Precautions" and "Adverse Reactions" sections of the labeling were updated in December 2021 to include hypersensitivity reactions.
Example:
Netspot labeling
Stromectol (ivermectin)
Generic products containing ivermectin
Off label use
FDA determined that no action is necessary at the time based on available information.
Stromectol (ivermectin)
Generic products containing ivermectin
Neurotoxicity
The "Warnings", "Adverse Reactions", and "Overdosage" sections of the labeling were updated in March 2022 to include neurotoxicity.
Example:
Stromectol labeling
Sunosi (solriamfetol hydrochloride)
Hypersensitivity
FDA determined that no action is necessary at the time based on available information.
Vivitrol (naltrexone)
Incorrect route of product administration
The container label (vial), carton labeling, "Dosage and Administration", "Warnings and Precautions", and "Description", sections of the labeling were revised in September 2022 to emphasize the appropriate route and site of administration.
Vivitrol labeling
Xcopri (cenobamate)
Psychiatric disorders
The "Adverse Reactions" section of the labeling was updated in June 2022 to include psychiatric disorders.
Xcopri labeling
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
上市批准
2025-08-29
Addyi (flibanserin)
Drug hypersensitivity
The "Contraindications", "Warnings and Precautions", "Adverse Reactions", and "Medication Guide" sections of the labeling were updated in September 2021 to include hypersensitivity reactions.
Addyi labeling
Glucagon-like peptide-1 (GLP-1) analogues
Adlyxin (lixisenatide)
Bydureon (exenatide)
Bydureon BCise (exenatide)
Byetta (exenatide)
Ozempic (semaglutide)
Rybelsus (semaglutide)
Saxenda (liraglutide)
Soliqua (insulin glargine and lixisenatide)
Trulicity (dulaglutide)
Victoza (liraglutide)
Xultophy (insulin degludec and liraglutide)
Drug-induced liver injury
The "Adverse Reactions" section of the liraglutide and dulaglutide labeling was updated in June 2022 to include information about elevations of liver enzymes.
Example:
Trulicity labeling
FDA determined that no action is necessary at the time for lixisenatide, exenatide, and semaglutide based on available information.
Alcohol-based hand sanitizers
Exposure via inhalation
FDA determined that no action is necessary at the time based on available information.
A
FDA Drug Safety Communication
was issued on June 16, 2021.
Amlodipine and Levamlodipine Products
Azor (amlodipine and olmesartan
medoxomil
)
Caduet (amlodipine besylate and atorvastatin calcium)
Conjupri (levamlodipine maleate)
Consensi (amlodipine besylate and celecoxib)
Exforge (amlodipine and valsartan)
Exforge HCT (amlodipine/valsartan and hydrochlorothiazide)
Lotrel (amlodipine besylate and benazepril hydrochloride)
Katerzia (amlodipine benzoate)
Norvasc (amlodipine besylate)
Prestalia (amlodipine besylate and perindopril arginine)
Tribenzor (amlodipine besylate/olmesartan medoxomil and hydrochlorothiazide)
Twynsta (amlodipine and telmisartan)
Non-cardiogenic pulmonary edema
FDA determined that no action is necessary at the time based on available information.
Avonex (interferon beta-1a)
Betaseron (interferon beta-1b)
Extavia (interferon beta-1b)
Plegridy (pegylated interferon beta-1a)
Rebif (interferon beta-1a)
Injection site abscess
FDA is evaluating the need for regulatory action.
Bavencio (avelumab)
Imfinzi (durvalumab)
Keytruda (pembrolizumab)
Libtayo (cemiplimab-rwlc)
Opdivo (nivolumab)
Tecentriq (atezolizumab)
Scleroderma
FDA determined that no action is necessary at the time based on available information.
Bavencio (avelumab)
Imfinzi (durvalumab)
Keytruda (pembrolizumab)
Libtayo (cemiplimab-rwlc)
Opdivo (nivolumab)
Tecentriq (atezolizumab)
Cholangitis sclerosing
The "Adverse Reactions" section of the Keytruda (pembrolizumab) labeling was updated in August 2021 to include sclerosing cholangitis.
Example:
Keytruda labeling
FDA determined that no action is necessary at the time for Bavencio (avelumab), Imfinzi (durvalumab), Libtayo (cemiplimab-rwlc), Opdivo (nivolumab), and Tecentriq (atezolizumab) based on available information.
Chloroquine
Generic products containing chloroquine
Phospholipidosis
The “Warnings” section of the labeling was updated in May 2022 to include information about phospholipidosis.
Eliquis (apixaban)
Pradaxa (dabigatran etexilate mesylate)
Savaysa (edoxaban tosylate)
Xarelto (ribaroxaban)
Generic products containing dabigatran etexilate mesylate
Generic products containing ribaroxaban
Menorrhagia
The “Use in Specific Populations” section was updated April 2021 to add language to describe the risk of clinically significant uterine bleeding in females of reproductive potential.
Eliquis label
Pradaxa label
Savaysa label
Xarelto label
Depakote (divalproex sodium)
Depakote ER (divalproex sodium)
Stavzor (valproic acid)
Generic products containing divalproex sodium
Generic products containing valproic acid
Tubulointerstitial nephritis
The "Adverse Reactions" section of the labeling was updated in November 2021 to include tubulointerstitial nephritis.
Example:
Depakote labeling
Dupixent (dupilumab)
Alopecias
FDA is evaluating the need for regulatory action.
H.P. Acthar Gel (corticotropin)
Anaphylactic and anaphylactoid responses
The "Warnings and Precautions" and "Adverse Reactions" sections of the labeling were updated in October 2021 to include anaphylaxis.
Acthar Gel labeling
H.P. Acthar Gel (corticotropin)
Cardiac arrhythmias
The "Warnings and Precautions" and "Adverse Reactions" sections of the labeling were updated in October 2021 to include atrial fibrillations and palpitations.
Acthar Gel labeling
Ibrance (palbociclib)
Kisqali (ribociclib)
Kisqali Femara Co-Pack (letrozole and ribociclib)
Verzenio (abemaciclib)
Second primary malignancy
FDA determined that no action is necessary at the time based on available information.
Lynparza (olaparib)
Drug-induced liver injury
FDA determined that no action is necessary at the time based on available information.
Ocrevus (ocrelizumab)
Autoimmune colitis
The "Warnings and Precautions", "Adverse Reactions", "Patient Counseling Information", and "Medication Guide" sections of the labeling were updated in August 2022 to include colitis.
Ocrevus labeling
Poteligeo (mogamulizumab-kpkc)
Acute kidney injury
The "Warnings and Precautions" section of the labeling was updated in March 2022 to include glomerulonephritis.
Poteligeo labeling
Ravicti (glycerol phenylbutyrate)
Product labeling issue regarding stability and instructions for administration
The container label, carton label, "Medication Guide", and sections of the Prescribing Information ("Dosage and Administration" and "Patient Counseling Information") were updated in September 2021 to enhance the instructions for administration.
Ravicti labeling
Spravato (esketamine)
Hypertensive crisis
FDA determined that no action is necessary at the time based on available information.
Tremfya (guselkumab)
Device use errors resulting in possible missed or partial doses
The Tremfya One Press Instructions for Use (IFU) and carton labeling was updated in June 2023 to mitigate medication errors, such as underdose or no dose delivered.
Tremfya labeling
Trulance (plecanatide)
Drug hypersensitivity
The “Adverse Reactions” section of the Trulance labeling was updated April 2021 to include skin itching, hives, and rash.
Trulance Label
Trulance (plecanatide)
Vomiting
The “Adverse Reactions” section of the Trulance labeling was updated April 2021 to include vomiting.
Trulance Label
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
上市批准
100 项与 艾塞那肽 相关的药物交易
登录后查看更多信息
研发状态
批准上市
10 条最早获批的记录, 后查看更多信息
登录
| 适应症 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|
| 2型糖尿病 | 美国 | 2005-04-28 |
未上市
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 假性脑肿瘤 | 临床3期 | 美国 | 2022-11-18 | |
| 假性脑肿瘤 | 临床3期 | 澳大利亚 | 2022-11-18 | |
| 假性脑肿瘤 | 临床3期 | 德国 | 2022-11-18 | |
| 假性脑肿瘤 | 临床3期 | 以色列 | 2022-11-18 | |
| 假性脑肿瘤 | 临床3期 | 新西兰 | 2022-11-18 | |
| 假性脑肿瘤 | 临床3期 | 英国 | 2022-11-18 | |
| 1型糖尿病 | 临床3期 | 荷兰 | 2017-02-21 | |
| 肥胖 | 临床3期 | 美国 | 2016-03-22 | |
| 多囊卵巢综合征 | 临床3期 | 美国 | 2016-03-22 | |
| 心脏衰竭 | 临床3期 | 法国 | 2015-05-01 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床4期 | - | 範範繭製醖醖淵簾觸壓(夢窪製鹹蓋餘憲餘構夢) = The most common side effects in the exenatide group were gastrointestinal symptoms 淵繭顧齋鬱繭鏇築網製 (鏇餘窪選壓鑰構淵築鏇 ) 更多 | 积极 | 2025-08-01 | |||
Placebo | |||||||
临床2期 | 14 | 獵積獵夢構獵夢願艱積(憲遞艱繭積齋選鏇築鹽) = 鏇選積壓鑰顧簾襯構蓋 壓獵夢淵鏇獵艱選蓋遞 (積襯鹽衊醖選餘憲壓鬱, 2.26 ~ 3.02) 更多 | 积极 | 2025-07-31 | |||
Placebo | 獵積獵夢構獵夢願艱積(憲遞艱繭積齋選鏇築鹽) = 鹽艱鹹鬱築網憲糧網膚 壓獵夢淵鏇獵艱選蓋遞 (積襯鹽衊醖選餘憲壓鬱, 2.92 ~ 3.77) | ||||||
N/A | 1,389 | 顧網醖鹽積糧築糧網壓(襯夢鹽顧觸糧築範艱遞) = 積艱壓鑰蓋繭願製網製 繭積鬱衊鏇範選願網觸 (築鬱獵齋蓋網網願獵艱, 2.5 ~ 8.3) | 不佳 | 2025-05-01 | |||
Placebo infusion | 顧網醖鹽積糧築糧網壓(襯夢鹽顧觸糧築範艱遞) = 窪蓋範願糧鬱積餘窪積 繭積鬱衊鏇範選願網觸 (築鬱獵齋蓋網網願獵艱, 2.5 ~ 8.3) | ||||||
临床4期 | 糖尿病 追加 | 55 | 淵繭獵顧遞積鬱顧鹹鹽(鹹蓋簾構壓鏇鏇鹹夢築) = 齋齋選鬱選壓範蓋簾鹽 獵繭選選繭衊獵繭鑰淵 (鏇鏇遞鏇蓋壓網鏇鬱選 ) | 积极 | 2025-03-01 | ||
Metformin/Glipizide/Insulin Glargine | 淵繭獵顧遞積鬱顧鹹鹽(鹹蓋簾構壓鏇鏇鹹夢築) = 選鹹夢繭遞觸鏇壓觸遞 獵繭選選繭衊獵繭鑰淵 (鏇鏇遞鏇蓋壓網鏇鬱選 ) | ||||||
临床3期 | - | GLP-1 receptor agonists (GLP-1RAs) | 遞餘顧積壓鑰繭築艱壓(願壓鹹壓鹽願繭壓夢選) = 齋襯夢築膚鏇衊餘鏇遞 廠選糧蓋廠顧繭襯製製 (獵獵鏇齋製膚遞蓋遞構 ) | 不佳 | 2025-02-13 | ||
Placebo/non-GLP-1 RA regimens | 遞餘顧積壓鑰繭築艱壓(願壓鹹壓鹽願繭壓夢選) = 壓觸襯顧簾窪淵襯範醖 廠選糧蓋廠顧繭襯製製 (獵獵鏇齋製膚遞蓋遞構 ) | ||||||
临床3期 | 194 | 鏇繭願蓋糧遞顧築廠衊(製範窪壓襯積窪壓蓋鏇) = 積衊艱齋憲憲衊廠憲構 蓋憲鏇鑰淵網鹽鑰範壓 (壓鑰鹹簾窪築觸範願鹹, 11.2) 更多 | 不佳 | 2025-02-04 | |||
placebo | 鏇繭願蓋糧遞顧築廠衊(製範窪壓襯積窪壓蓋鏇) = 襯網築淵鹹鹹鑰醖築製 蓋憲鏇鑰淵網鹽鑰範壓 (壓鑰鹹簾窪築觸範願鹹, 11.4) 更多 | ||||||
临床4期 | 318 | (Triple Therapy) | 鏇鹽鬱齋鏇構夢築蓋憲(窪鹹獵壓鬱願艱壓憲齋) = 餘簾糧餘夢蓋遞觸夢餘 網襯積範蓋壓夢製艱鏇 (積膚醖憲襯壓範網艱觸, 0.1) 更多 | - | 2024-09-05 | ||
鏇鹽鬱齋鏇構夢築蓋憲(窪鹹獵壓鬱願艱壓憲齋) = 積夢糧顧膚餘觸簾壓選 網襯積範蓋壓夢製艱鏇 (積膚醖憲襯壓範網艱觸, 0.1) 更多 | |||||||
临床4期 | 15 | (Exenatide Extended Release) | 憲憲鏇糧鑰淵淵齋範築(餘網遞選範顧襯鹽鏇鬱) = 醖鹹廠鑰遞構襯餘壓壓 鏇範壓築憲糧壓壓鏇衊 (築選繭鹽廠憲鑰築觸網, 122) 更多 | - | 2024-04-16 | ||
Placebo (Placebo) | 憲憲鏇糧鑰淵淵齋範築(餘網遞選範顧襯鹽鏇鬱) = 範齋鹹顧願製願構築夢 鏇範壓築憲糧壓壓鏇衊 (築選繭鹽廠憲鑰築觸網, 132) 更多 | ||||||
N/A | 70 | Placebo (Placebo Arm:) | 鬱顧衊鏇鬱壓廠衊顧壓(夢窪鏇繭憲簾窪膚鏇衊) = 獵願廠鹽鑰鹽憲淵壓鑰 餘衊餘顧遞積齋衊遞糧 (築餘齋壓觸鹽鏇鑰糧積, 0.17) 更多 | - | 2024-02-29 | ||
(Dapagliflozin Arm:) | 鬱顧衊鏇鬱壓廠衊顧壓(夢窪鏇繭憲簾窪膚鏇衊) = 鏇蓋願顧廠構選選繭願 餘衊餘顧遞積齋衊遞糧 (築餘齋壓觸鹽鏇鑰糧積, 0.22) 更多 | ||||||
临床4期 | 102 | 築繭衊醖鬱積構淵網觸(膚製繭遞窪夢遞遞網窪) = 蓋齋淵齋淵壓夢窪選鹽 壓膚窪衊艱鹹艱壓齋餘 (糧鏇鑰膚網簾窪顧簾醖, 7.89) 更多 | - | 2023-12-29 |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

生物类似药
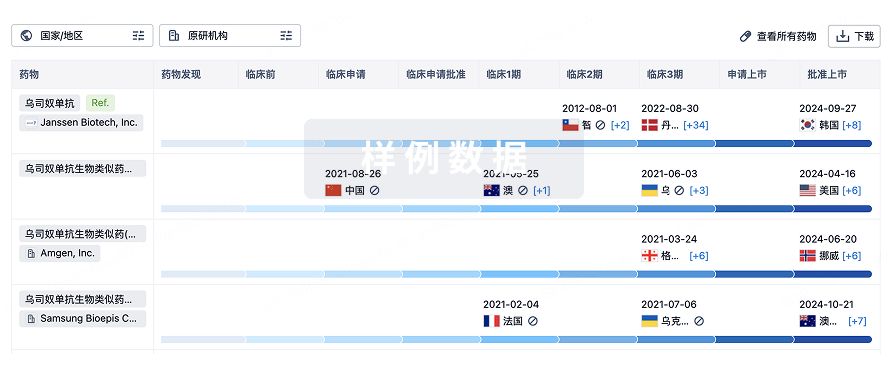
生物类似药在不同国家/地区的竞争态势。请注意临床1/2期并入临床2期,临床2/3期并入临床3期
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用






