更新于:2024-11-01

Chia Tai Tianqing Pharmaceutical Group Co., Ltd.
更新于:2024-11-01
概览
标签
肿瘤
其他疾病
皮肤和肌肉骨骼疾病
小分子化药
单克隆抗体
生物药
疾病领域得分
一眼洞穿机构专注的疾病领域
暂无数据
技术平台
公司药物应用最多的技术
暂无数据
靶点
公司最常开发的靶点
暂无数据
| 排名前五的药物类型 | 数量 |
|---|---|
| 小分子化药 | 72 |
| 单克隆抗体 | 22 |
| 生物药 | 18 |
| 生物类似药 | 12 |
| 化学药 | 10 |
关联
188
项与 正大天晴药业集团股份有限公司 相关的药物作用机制 ALK抑制剂 [+2] |
在研机构 |
原研机构 |
在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 中国 |
首次获批日期2024-06-11 |
靶点 |
作用机制 PDL1抑制剂 [+1] |
在研机构 |
原研机构 |
非在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 中国 |
首次获批日期2024-04-30 |
作用机制 ALK抑制剂 [+2] |
在研机构 |
原研机构 |
在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 中国 |
首次获批日期2024-04-24 |
970
项与 正大天晴药业集团股份有限公司 相关的临床试验A Multicenter, Open Label Phase I Clinical Trial Evaluating the Safety and Pharmacokinetics of TQB2252 Injection in Subjects With Advanced Malignant Tumors
This study is a multicenter, single arm, and open design Phase I clinical trial aimed at evaluating the safety, Pharmacokinetics (PK) characteristics, immunogenicity, and preliminary efficacy of TQB2252 injection in subjects with advanced malignant tumors.
开始日期2024-11-01 |
申办/合作机构 |
A Phase I Clinical Study of TQB3002 in Patients With Advanced Cancers
This is a Phase I study to evaluate the safety, tolerability, and efficacy of TQB3002 in subjects with advanced cancers
开始日期2024-11-01 |
申办/合作机构 |
Phase I Clinical Trial Evaluating Drug Drug Interactions of TQB3616 Capsules
To evaluate the effect of itraconazole/rifampicin on the pharmacokinetics of TQB3616 capsules.
To assess the safety of a single dose of oral TQB3616 capsules and in combination with itraconazole/rifampicin.
To assess the safety of a single dose of oral TQB3616 capsules and in combination with itraconazole/rifampicin.
开始日期2024-11-01 |
申办/合作机构 |
100 项与 正大天晴药业集团股份有限公司 相关的临床结果
登录后查看更多信息
0 项与 正大天晴药业集团股份有限公司 相关的专利(医药)
登录后查看更多信息
272
项与 正大天晴药业集团股份有限公司 相关的文献(医药)2024-11-01·Cancer Chemotherapy and Pharmacology
Pharmacokinetics, mass balance, and metabolism of [14C]envonalkib (TQ-B3139), a novel ALK tyrosine kinase inhibitor, in healthy Chinese subjects
Article
作者: Wang, Xin ; Yan, Shu ; Diao, Xingxing ; Ding, Dawei ; Miao, Liyan ; Zhang, Hua ; Yu, Ding ; Ma, Sheng ; Wan, Xiaojing ; Wang, Xunqiang
2024-11-01·Journal of Macromolecular Science, Part B
Accelerated self-assembly of filipin proteins and formation of hydrogels
作者: Ma, Li ; Lu, Liyuan ; Wei, Yilong ; Liu, Yuan ; Ma, Doudou ; Li, Pingping ; Zhao, Xiaoliang ; Zhang, Chuanhui ; Zhang, Weijie
2024-09-01·Naunyn-Schmiedeberg's Archives of Pharmacology
Pharmacokinetics and safety of a new generic lurasidone: a phase I bioequivalence study in healthy Chinese subjects
Article
作者: Liu, Lang ; Xiao, Nan ; Chang, Tianying ; Cheng, Yang ; Liu, Guangwen ; Cui, Yingzi ; Wang, Yanli ; Wang, Wanhua ; Qu, Xinyao ; Chen, Jiahui ; Xue, Jinling ; Wang, Zeyu ; Deng, Qiaohuan ; Zhou, Yannan ; Zhang, Lixiu ; Zhao, Yicheng ; Liu, Zhengzhi ; Yang, Wei ; Yang, Haimiao
4,166
项与 正大天晴药业集团股份有限公司 相关的新闻(医药)2024-10-31
·米内网
精彩内容
日前,国家药监局官网显示,宜昌人福药业的复方醋酸钠林格注射液以仿制3类报产获批,视同过评。米内网数据显示,复方醋酸钠林格(钠钾镁钙葡萄糖)注射剂在近年中国公立医疗机构终端均超过10亿元销售规模,2024年上半年同比增长1.40%。今年以来,人福医药已有15款产品获批上市,19款产品报产在审。
来源:米内网一键检索
复方醋酸钠林格注射液主要用于循环血量及组织间液减少时细胞外液的补充,代谢性酸中毒的纠正。
近年中国公立医疗机构终端复方醋酸钠林格(钠钾镁钙葡萄糖)注射剂销售情况(单位:万元)来源:米内网中国公立医疗机构药品终端竞争格局
复方醋酸钠林格(钠钾镁钙葡萄糖)注射剂在近年中国城市公立医院、县级公立医院、城市社区中心以及乡镇卫生院(简称中国公立医疗机构)终端均超过10亿元销售规模,2024年上半年同比增长1.40%。米内网数据显示,复方醋酸钠林格注射液(钠钾镁钙葡萄糖注射液)有8家企业拥有生产批文,宜昌人福药业、江苏正大丰海制药、四川科伦药业等7家过评,石家庄四药、浙江医药新昌制药、辰欣药业等超10家企业以仿制3类报产在审,获批后视同过评。
10月26日,人福医药发布公告,其控股子公司宜昌人福药业的司美格鲁肽注射液以化学药品2.2类获批临床,同意本品开展2型糖尿病适应症的临床试验,目前全球尚无化学合成的司美格鲁肽产品上市。
今年以来,人福医药新品捷报频传,公司已有15款产品获批上市,19款产品报产在审,涵盖抗肿瘤和免疫调节剂、神经系统药物、消化系统及代谢药等多个治疗大类。资料来源:国家药监局官网、米内网数据库等注:米内网《中国公立医疗机构药品终端竞争格局》,统计范围是:中国城市公立医院、县级公立医院、城市社区中心以及乡镇卫生院,不含民营医院、私人诊所、村卫生室;上述销售额以产品在终端的平均零售价计算。如有疏漏,欢迎指正!
本文为原创稿件,转载请注明来源和作者,否则将追究侵权责任。投稿及报料请发邮件到872470254@qq.com稿件要求详询米内微信首页菜单栏商务及内容合作可联系QQ:412539092
●温馨提示●
因为微信公众号修改了推送规则,最近很多读者反映没有及时看到推文。
把米内网设为“星标”,就能每天与我们不见不散啦,具体操作如下:
【分享、点赞、在看】点一点不失联哦
上市批准一致性评价
2024-10-31
·米内网
精彩内容
10月31日,CDE官网显示,中玉制药(海口)的盐酸溴己新注射液以仿制3类报产在审。米内网数据显示,盐酸溴己新注射液在2023年中国公立医疗机构终端销售额突破10亿元大关,同比增长290.29%。
盐酸溴己新注射液用于在口服给药困难的情况下,慢性支气管炎及其他呼吸道疾病,如哮喘、支气管扩张、矽肺等有粘痰不易咳出的患者。
近年中国公立医疗机构终端盐酸溴己新注射液销售情况(单位:万元)
来源:米内网中国公立医疗机构药品终端竞争格局
盐酸溴己新注射液在近年中国城市公立医院、县级公立医院、城市社区中心以及乡镇卫生院(简称中国公立医疗机构)终端销售额快速增长,2023年突破10亿元大关,同比增长290.29%。
米内网数据显示,盐酸溴己新注射液有16家企业拥有生产批文,江苏正大清江制药、海南葫芦娃药业集团、珍宝岛药业等15家过评,中玉制药(海口)、辰欣药业、北京双鹭药业、以岭万洲国际制药等13家报产在审。
来源:米内网项目进度数据库
在此之前,中玉制药(海口)已有罗库溴铵注射液报产在审,在2023年中国公立医疗机构终端罗库溴铵注射液销售额超过5亿元,是肌肉松弛化药TOP3产品。
资料来源:CDE官网、米内网数据库
注:米内网《中国公立医疗机构药品终端竞争格局》,统计范围是:中国城市公立医院、县级公立医院、城市社区中心以及乡镇卫生院,不含民营医院、私人诊所、村卫生室;上述销售额以产品在终端的平均零售价计算。如有疏漏,欢迎指正!
本文为原创稿件,转载请注明来源和作者,否则将追究侵权责任。投稿及报料请发邮件到872470254@qq.com稿件要求详询米内微信首页菜单栏商务及内容合作可联系QQ:412539092
【分享、点赞、在看】点一点不失联哦
2024-10-31
·药闻康策
☝ 点击上方 一键预约 ☝
最新最热的医药健康新闻政策
前 言
十月中,随着山西省药械集中招标采购中心发布《关于报送第十批国家组织药品集中采购品种需求量的通知》,第十批国采的脚步愈加临近。根据法伯院内渠道数据显示,既往国家九批集采品种已覆盖各类治疗领域,对慢病领域影响较大,仿制药市场占比逐年被压缩。截止到2023年底已执行的前八批带量采购品种与2019年相比销售额下降45%,带动整体院内市场规模的降低。那么,国采药品的院内市场表现如何?受集采影响较大的企业有哪些?相关市场发生了怎样的变化?
01
历次国家集采药品整体分析
政策演变 | 院内市场表现
截至2024年上半年,国家层面共组织开展了九批药品集中带量采购,前八批在2023年底前已执行,第九批于2024年3月开始执行。几年来,国家集中带量采购执行政策不断优化,不断调整规则以缩小同品种不同企业中选价差(表1)。
表1-国家集采政策演变
数据来源:上海阳光采购网
在历次九批国家集中带量采购中,化学药和生物制剂类共覆盖品种数达374个,除生物制剂只涵盖胰岛素药物外,化学药品适应症涉及范围较广,覆盖从心血管疾病、糖尿病和抗感染药物等常用药物,到精神类疾病、抗肿瘤药物及呼吸系统药物等多种治疗领域。历次集采平均降价幅度从48%到58%不等(图1)。
图1-1-9批国家带量采购品种数量及治疗领域分布
数据来源:上海阳光采购网;法伯研究院统计
四大维度纵览带量采购药品院内市场表现
根据法伯院内渠道数据显示,从院内市场来看,在全国范围集采之前,1-9批集采品种占整体院内市场规模的36.9%。此外,与2019年相比,2023年已执行的1-8批国家集采品种销售额下降44.9%,2023年医疗机构节省医保基金超2,000亿元(图2)。
图2-国家集采药品整体市场变化
数据来源:法伯院内渠道数据
分渠道来看,带量采购的常态化执行对各个院内医疗机构渠道市场均产生较大影响。法伯数据显示,城市医院、县域医院和社区卫生中心渠道集采品种规模降幅均高于45%,而乡镇卫生院渠道的降幅相对较小,这与集采前后的渠道品种平均价格有关(图3)。
图3-1-8批集采品种院内渠道销售额变化
数据来源:法伯院内渠道数据
从治疗领域上看,国家集采品种主要集中在全身用抗感染、消化道和代谢、以及心血管系统用药等领域,根据法伯院内渠道数据显示,2019年这三个领域中VBP品种的销售额在本领域药品总规模中占比达到50%以上。此外,降幅较高的主要有消化代谢和抗肿瘤等领域(图4)。
图4-1-8批集采品种治疗领域分布及市场表现
数据来源:法伯院内渠道数据
分内外资企业来看,聚焦前八批带量采购品种,相较于2019年,2023年外资企业总销售额降低了近60%,五年来贡献占比逐年降低,到2023年降至32.1%(图5)。
图5-1-8批集采品种内外资销售额贡献变化
数据来源:法伯院内渠道数据
02
受集采影响较大企业及市场表现
集采品种数 | 市场表现变化
可以分别从纳入集采的产品数量,以及集采产品市场销售表现两方面,来看国家集采为医药企业的带来的影响。
1-9批集采品种数量较多的企业
从纳入集采品种数量来看,内资企业上海医药集团、齐鲁制药、扬子江和科伦集团均有近100个带量采购品种,其中齐鲁制药中标产品数量最多,达59个。此外,正大天晴、扬子江药业、科伦集团中标产品数均超过40个。外资企业方面,集采产品数最多的是诺华(含山德士等仿制药子公司)、联邦集团和辉瑞集团,而之前辉瑞集团的立普妥(阿托伐他汀)、络活喜(氨氯地平)等多个大品种未中标原研产品已剥离至晖致医药。整体来看,外资企业的中标产品数普遍较少,中标产品数及其在集采产品中占比均较高的是赛诺菲,有6个产品中标,包括波立维(氢氯吡格雷)、安博诺(厄贝沙坦氢氯噻嗪)、乐沙定(奥沙利铂)和胰岛素等产品(图6)。
图6-1-9批集采品种(中标/未中标产品)数量较多的企业
数据来源:上海阳光采购网;法伯院内渠道数据
重点企业因国家集采导致市场表现变化
从集采对企业的市场表现影响看,根据法伯院内渠道数据显示,第1至9批集采产品在2019年份额较高的企业主要有外资企业阿斯利康、拜耳、赛诺菲、辉瑞、晖致、诺和诺德,以及内资企业恒瑞医药、鲁南制药等(表2)。各企业因应对带量采购的策略不同而显示出市场表现的差异,有的企业随着各批次陆续执行,因受影响较大导致销售额及排名发生较大变化;有些企业则市场应对能力较强,通过改变策略来减小带量采购的冲击;还有些企业能够从带量采购中获益。
表2-重点企业市场表现变化-23 vs. 19
(以2019年制药企业全部产品销售额排名)
数据来源:上海阳光采购网;法伯院内渠道数据
从2019到2023年企业销售表现变化来看,多数外资企业带量采购相关产品(原研产品为主)在企业全产品中占比较高,在集采的影响下,随着院内未中标原研产品销量和销售额下降,企业整体销售规模大幅下降,同时排名也相应下降。而有些企业通过创新药和药品升级上市发力,改变创新药和带量采购产品的营销策略,在市场中仍排名前列。以阿斯利康为例,虽然其集采相关产品份额从2019年的75.7%下降至2023年的29.3%,但是其他产品持续增长,如泰瑞沙(奥希替尼)对易瑞沙(吉非替尼,第1批)、信必可都保(布地奈德福莫特罗)对普米克令舒(布地奈德,第5批)的药品升级,抵消了部分带量采购的影响。再如辉瑞,剥离出带量采购大品种如立普妥、络活喜等到晖致经营,企业整体呈现正向增长(表2)。
内资企业方面,如扬子江、恒瑞医药和正大天晴等带量采购产品在企业全产品销售规模中占比较高,无论产品中标数量多少,都会受到带量采购的影响,销售规模和排名均下降,但是因为创新能力较强,在市场中仍保持较高排名。还有一类内资企业是在带量采购政策中获益的,如石药集团、科伦集团和齐鲁制药等,部分仿制产品新上市后集采中标,或中标仿制产品集采前份额相对较低,在集采执行后随着中标产品销量和销售额的大幅升高,带动整个企业的销售额升高。
(来源: 法伯科技Pharbers)
药闻康策
新媒体矩阵微信公众号
点击下方 一键关注
【免责声明】
1.“药闻康策”部分文章信息来源于网络转载是出于传递更多信息之目的,并不意味着赞同其观点或证实其内容的真实性。如对内容有疑议,请及时与我司联系。2.“药闻康策”致力于提供合理、准确、完整的资讯信息,但不保证信息的合理性、准确性和完整性,且不对因信息的不合理、不准确或遗漏导致的任何损失或损害承担责任。3.“药闻康策”所有信息仅供参考,不做任何商业交易或医疗服务的根据,如自行使用“药闻康策”内容发生偏差,我司不承担任何责任,包括但不限于法律责任,赔偿责任。
欢迎转发分享、点赞、点在看
带量采购
100 项与 正大天晴药业集团股份有限公司 相关的药物交易
登录后查看更多信息
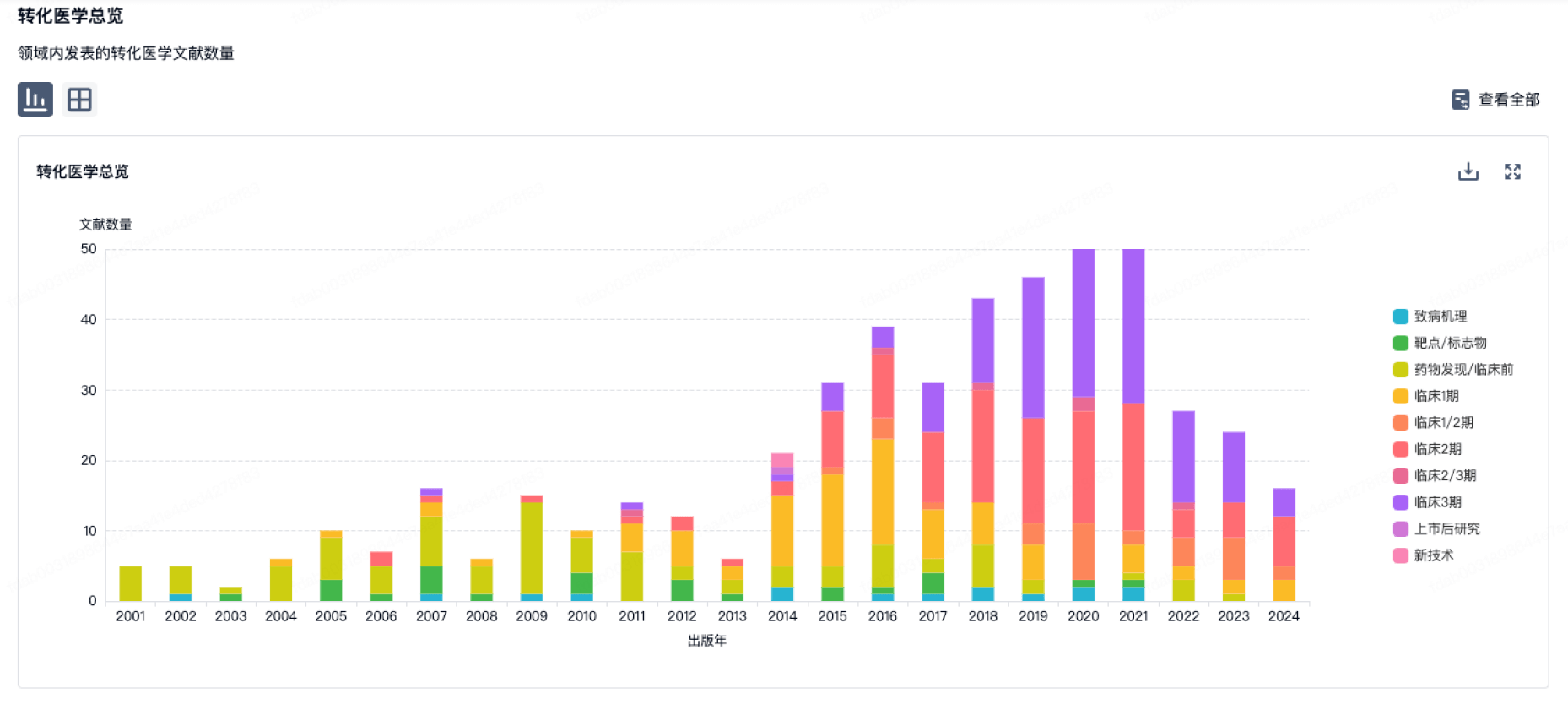
100 项与 正大天晴药业集团股份有限公司 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2024年11月17日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
药物发现
7
26
临床前
临床申请
2
12
临床申请批准
临床1期
40
20
临床2期
临床3期
18
6
申请上市
批准上市
26
31
其他
登录后查看更多信息
当前项目
| 药物(靶点) | 适应症 | 全球最高研发状态 |
|---|---|---|
贝伐珠单抗生物类似药 (正大天晴) ( VEGF-A ) | 复发性胶质母细胞瘤 更多 | 批准上市 |
曲妥珠单抗生物类似药 (正大天晴) ( HER2 ) | HER2阳性胃癌 更多 | 批准上市 |
甘草酸二铵 ( 11β-HSD1 ) | 慢性肝炎 更多 | 批准上市 |
盐酸安罗替尼 ( FGFRs x PDGFR x VEGFR1 x VEGFR2 x VEGFR3 x c-Kit ) | 分化型甲状腺癌 更多 | 批准上市 |
阿达木单抗生物类似药 (正大天晴) ( TNF-α ) | 类风湿关节炎 更多 | 批准上市 |
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

