预约演示
更新于:2025-09-17

Medtronic Plc
更新于:2025-09-17
概览
标签
心血管疾病
非降解型分子胶
重组多肽
疾病领域得分
一眼洞穿机构专注的疾病领域
暂无数据
技术平台
公司药物应用最多的技术
暂无数据
靶点
公司最常开发的靶点
暂无数据
| 排名前五的药物类型 | 数量 |
|---|---|
| 小分子化药 | 2 |
| 非降解型分子胶 | 1 |
| 重组多肽 | 1 |
| 生长因子 | 1 |
关联
5
项与 Medtronic Plc 相关的药物靶点 |
作用机制 mTOR抑制剂 |
最高研发阶段批准上市 |
首次获批国家/地区 加拿大 |
首次获批日期2008-05-16 |
靶点 |
作用机制 BMPR1A刺激剂 |
原研机构 |
最高研发阶段批准上市 |
首次获批国家/地区 欧盟 [+3] |
首次获批日期2002-09-09 |
1,624
项与 Medtronic Plc 相关的临床试验NCT07174622
Real-world Clinical Outcomes in Medicare Patients With Hypertension Treated With Symplicity Renal Denervation Plus Standard of Care Versus Standard of Care in Integrated EHR and Claims Data
This observational study will assess the long-term effectiveness of the RDN procedure for lowering blood pressure in Medicare patients with uncontrolled hypertension. This research will be conducted using de-identified electronic health records (EHR) and administrative health insurance claims data. Patients are enrolled through the submission of claims or encounter data to CMS.
开始日期2025-10-08 |
申办/合作机构 |
NCT07166341
Accuracy Performance Verification of the INVOS™ PM7100 System, Comparing rSO2 Values to Reference Blood Oxygen Saturation Measurements in Adult Human Volunteers
Prospective, observational, pre-market, verification study to confirm accuracy of the INVOS 7100 system in compliance with ISO 80601-2-85:2021 standard
开始日期2025-10-01 |
申办/合作机构 |
NCT06918977
Evaluation of a Novel Telemedicine Clinic for Patients With DM2 on Multiple Daily Insulin (MDI) Regimens
Almost 40% of veterans have diabetes, the proposed research may improve clinical care among veterans with diabetes. In this application we are going to examine whether utilizing Smart Insulin pens (SIPs) with CGMs and telemedicine is a better tool for managing diabetes compared to traditional insulin pens, CGMs and telemedicine, leading to improved blood sugar control and better clinical outcomes.
开始日期2025-09-30 |
申办/合作机构 |
100 项与 Medtronic Plc 相关的临床结果
登录后查看更多信息
0 项与 Medtronic Plc 相关的专利(医药)
登录后查看更多信息
1,478
项与 Medtronic Plc 相关的文献(医药)2025-12-31·BLOOD PRESSURE
Cost-effectiveness analysis of radiofrequency renal denervation for uncontrolled hypertension in Sweden
Article
作者: Lindgren, P. ; Ryschon, A. M. ; Cao, K. N. ; Johansen, M. L. ; Kolovetsios, M. D. ; Pietzsch, J. B. ; Kahan, T.
INTRODUCTION:
Radiofrequency renal denervation (RF RDN) is a catheter-based therapy for uncontrolled hypertension. This model-based analysis examined the cost-effectiveness of RF RDN in Sweden.
METHODS:
Clinical events, costs, quality-adjusted life-years (QALYs) were projected over 10-year and lifetime horizons using a decision-analytic Markov model. Primary health states, included hypertension alone, myocardial infarction (MI), stroke, other symptomatic coronary heart disease (CHD), heart failure (HF), end-stage renal disease (ESRD), and death. Health state transitions were informed by multivariate risk equations. Clinical evidence from the SPYRAL HTN-ON MED trial informed the treatment effect modelled (-4.9 mmHg reduction in office systolic blood pressure (SBP) vs. sham). The base case was conducted from the Swedish healthcare payer perspective. The primary outcome was the incremental cost-effectiveness ratio (ICER),RF RDN vs. standard of care (SoC), evaluated against an assumed willingness-to-pay threshold of SEK 500,000 per QALY gained. Extensive sensitivity analyses were performed.
RESULTS:
At 10-years, the relative risks with RF RDN were 0.80 for stroke, 0.88 for MI, 0.89 for CHD, 0.72 for HF, 0.96 for ESRD, 0.86 for cardiovascular death and 0.93 for all-cause death. Over lifetime, RF RDN led to incremental costs of SEK 63,136 (total costs SEK 497,498 vs. SEK 434,362) and incremental QALY gain of 0.45 (14.79 vs. 14.34), yielding an ICER of SEK 139,280 per QALY gained. RF RDN was cost-effective across all scenarios and sensitivity analyses.
CONCLUSION:
Model projections suggest RF RDN to be a cost-effective therapy for uncontrolled including resistant hypertension in Sweden based on contemporary clinical evidence.
2025-12-31·JOURNAL OF MEDICAL ECONOMICS
Safety and efficacy of an intrinsic antitachycardia pacing algorithm in patients from Japan and South Korea: results from a cardiac device registry in the Asia Pacific region
Article
作者: Mozingo, Joseph D. ; Goya, Masahiko ; Jacobsen, Luke ; Joung, Boyoung ; Park, Seung-Jung ; Ando, Kenji ; Holbrook, Reece ; Iskandar, Rowan
BACKGROUND:
Antitachycardia pacing (ATP) therapy, available in modern implantable cardioverter defibrillators (ICD) and cardiac resynchronization therapy defibrillators (CRT-D), aims to terminate ventricular arrhythmias without administering high energy shocks. The intrinsic ATP (iATP) algorithm automates ATP programming in real-time, tailoring therapy based on previous ATP attempts. This study evaluated the safety, efficacy, and clinical outcomes of iATP in patients from Japan and South Korea.
METHODS:
This study was a prospective, observational, multi-site registry that enrolled patients from Japan and South Korea implanted with an ICD or CRT-D device with the iATP algorithm. Patients were followed for a minimum of 12 months. Outcomes included ATP termination success, appropriate shocks, acceleration, arrhythmia-related syncope, and mortality. A post hoc unanchored matching-adjusted indirect comparison (uMAIC) was performed to compare iATP with standard ATP using published literature.
RESULTS:
A total of 800 patients were enrolled. The iATP success rate for terminating all episodes was 89.2% (86.2% Generalized Estimating Equation [GEE] estimated) and 82.2% for episodes in the fast VT zone (80.9% GEE estimated). Acceleration occurred in 2.0% of episodes, and arrhythmia-related syncope was observed in 0.5% of patients. The 1-year survival rate was 96.1%, with no device-related deaths or abnormal battery depletions. The uMAIC showed iATP had higher termination efficacy across all episodes (88.1% vs. 79.3%, p < 0.001), a lower probability of appropriate shocks per episode (iATP 14.7% and ATP 31.3%, p < 0.001), and fewer accelerations per episode (2.1% vs. 4.8%, p = 0.02), with similar probability of arrhythmia-related syncope per patient (0.5% vs 0.9%, p = 0.35) and mortality (12-month Kaplan Meyer survival estimate iATP 95.4%, ATP 95.3%, p = 0.43).
CONCLUSIONS:
iATP exhibited a high ventricular arrhythmia termination efficacy and a favorable safety profile. Comparison of iATP to standard ATP provides initial evidence of higher termination success, lower incidence of accelerations and appropriate shocks, and similar rates of mortality and arrhythmia-related syncope.
TRIAL REGISTRATION:
ClinicalTrials.gov Identifier: NCT01524276; Japan Registry of Clinical Trials Identifier: jRCT1042200049.
2025-08-01·JACC-Heart Failure
Incidence of Cardiac Arrhythmias Identified by Insertable Cardiac Monitors in Patients With Symptomatic Heart Failure
Article
作者: Chalasani, Prasad ; Kahwash, Rami ; Zile, Michael R ; Gravelin, Laura ; Butler, Javed ; Laechelt, Aimee ; Van Dorn, Brian ; Laager, Verla ; Bertolet, Barry ; Khan, Muhammad Shahzeb ; Gerritse, Bart ; Patel, Nirav ; Sarkar, Shantanu ; Wehking, Jennifer ; Lambert, Cameron
BACKGROUND:
Cardiac bradyarrhythmias and tachyarrhythmias in chronic heart failure (CHF) patients are associated with increased morbidity and mortality. Insertable cardiac monitors provide a continuous, ambulatory, accurate monitoring strategy for patients with CHF who are not candidates for cardiac implantable electronic devices.
OBJECTIVES:
This study aims to assess the occurrence of cardiac arrhythmias (CAs) in patients with CHF across the ejection fraction (EF) spectrum and not indicated for cardiac implantable electronic devices.
METHODS:
Patients with recent heart failure events enrolled in LINQ-HF (Reveal LINQ Heart Failure) and Phase 1 ALLEVIATE-HF (Algorithm Using LINQ Sensors for Evaluation and Treatment of Heart Failure) studies were implanted with an insertable cardiac monitor. All CAs were adjudicated first using an artificial intelligence model, then manually verified, and grouped into 3 categories: atrial fibrillation (AF), ventricular tachycardia (VT) or fibrillation (VF), and bradycardia/pauses.
RESULTS:
In total, 163 patients (mean age 67.2 ± 11.2 years, 62.6% male, 49.1% EF ≥50%, 83.4% Class III, 55.2% history of AF) were followed for 17.2 ± 9.8 months. Occurrence of AF was 59.7% at 2 years, 53.1% in heart failure with reduced ejection fraction (HFrEF) <50%, and 64.6% in heart failure with preserved ejection fraction (HFpEF) ≥50%. Incidence of AF in patients without prior AF was 23.8%, 23.9% in HFrEF, and 27.6% in HFpEF. Incidence of bradycardia or pause was 37.2% overall, (37.5% in HFrEF and 39.8% in HFpEF; 30.3% during daytime vs 19.4% during nighttime). Incidence of VT/VF was 14.3% overall, 19.8% in HFrEF, and 10.4%, in HFpEF.
CONCLUSIONS:
In CHF patients undergoing ambulatory, continuous, accurate arrhythmia monitoring, the incidence of AF and bradycardia/pause events was high and similar in HFpEF vs HFrEF patients. VT/VF was lower in HFpEF than HFrEF, but clinically important.
3,092
项与 Medtronic Plc 相关的新闻(医药)2025-09-16
This communication is part of the
Communications Pilot to Enhance the Medical Device Recall Program
. The FDA has become aware of a potentially high-risk device issue. The FDA will keep the public informed and update this web page as significant new information becomes available.
Affected Product
The FDA is aware that Medline has issued a letter to affected customers recommending certain Medline kits containing Medtronic DLP Left Heart Vent Catheters be removed from where they are used or sold:
Description
Kit Lot Number
Kit Lot Number
UDI-DI (Case)
Valve Pack-LF
24DMG366
10195327177966
40195327177967
Valve Pack-LF
25EMD672
10195327678227
40195327678228
Valve Pack-LF
25GMD016
10195327678227
40195327678228
Valve Pack-LF
25AMB610
10195327678227
40195327678228
Valve Pack-LF
25BMJ241
10195327678227
40195327678228
Valve Pack-LF
25CMI425
10195327678227
40195327678228
Valve Pack-LF
24DMC189
10195327678227
40195327678228
Valve Pack-LF
24EMC463
10195327678227
40195327678228
Valve Pack-LF
24GMJ353
10195327678227
40195327678228
Valve Pack-LF
24IMB824
10195327678227
40195327678228
Valve Pack-LF
24KME116
10195327678227
40195327678228
What to Do
Do not use affected product. Review your stock for affected items and lots and quarantine affected product. After completing the recall actions, destroy affected product.
On August 21, Medline sent all affected customers a letter recommending the following actions:
Immediately check your stock for the affected item numbers and the affected lot numbers listed within the recall portal. Quarantine all affected product. Upon completion of the below actions, please destroy affected product.
Please use the link and the information below to complete your response form.
Please list the quantity of affected product you have in inventory on the form.
Even if you do not have any affected product in inventory, please complete and submit the response form.
Website link:
https://recalls.medline.com
Recall Reference #: R-25-181-FGX1
Your account will receive credit once the response form is submitted.
If you are a distributor, or if you have resold or transferred this product to another company or individual, you are required by FDA regulations to notify them of this recall communication. Please request that customers destroy any affected product. You should include your customers’ quantities on your response form.
Check this web page for updates. The FDA is currently reviewing information about this potentially high-risk device issue and will keep the public informed as significant new information becomes available.
Reason for Early Alert
Medline is issuing a recall for specific lots of Medline Kits containing Medtronic DLP Left Heart Vent Catheters. Medtronic is issuing a
recall of their DLP Left Heart Vent Catheters
due to issues identified with certain lots of cannula products where the catheter may not retain its shape. As of July 28, 2025, Medtronic has received 41 complaints reporting that the catheters are resisting shape retention when being bent, with three reported injuries for perforation, and the remaining complaints reported for prolonged procedure or procedure delay with no patient consequence.
The potential harm when identified prior to use is procedure delay while another cannula is located. If this is not identified prior to use, and the clinician uses the cannula, the potential harms are abrasion and perforation (major or critical). Medtronic has not received any complaints resulting in patient death; however, perforation of critical heart tissue - if complicated, unnoticed, or untreated - may lead to the potential risk of death.
As of July 28, Medtronic has reported three serious injuries and no deaths associated with this issue.
Device Use
Medline Convenience Kits include two or more different medical devices packaged because they are frequently used together in a health care setting.
The Medtronic DLP Left Heart Vent Catheters within the convenience kits are intended for use in venting the left heart during cardiopulmonary bypass surgery up to six hours or less.
Contact Information
Customers in the U.S. with adverse reactions, quality problems, or questions about this recall should contact Medline at 866-359-1704 or
recalls@medline.com
.
Additional FDA Resources
Unique Device Identifier (UDI)
The unique device identifier (UDI) helps identify individual medical devices sold in the United States from distribution to use. The UDI allows for more accurate reporting, reviewing, and analyzing of adverse event reports so that devices can be identified more quickly, and as a result, problems potentially resolved more quickly.
How do I report a problem?
Health care professionals and consumers may
report adverse reactions or quality problems
they experienced using these devices to MedWatch: The FDA Safety Information and Adverse Event Reporting Program.
Content current as of:
09/16/2025
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
2025-09-15
Study to evaluate whether a new approach to pacing the heart can improve the lives of patients with heart failure with preserved ejection fraction (HFpEF) who have limited treatment options today
GALWAY, Ireland, Sept. 15, 2025 /PRNewswire/ -- Medtronic plc (NYSE: MDT), a global leader in healthcare technology, today announced the initiation of a pivotal study evaluating the use of elevated and personalized cardiac pacing rates for the treatment of patients with Heart Failure with preserved Ejection Fraction (ELEVATE-HFpEF, NCT06678841). Physicians routinely use pacing therapy to treat patients whose hearts beat too slowly (bradycardia). This study, however, will investigate a potential novel treatment for patients with HFpEF by using conduction system pacing to improve patient outcomes and heart failure symptoms. Medtronic intends to use the study results to pursue a new pacing indication for patients with HFpEF who currently have limited clinically proven treatments.
HFpEF is a condition when the cardiac muscles stiffen and do not relax properly, causing diminished blood flow and an inability to meet the body's needs. Patients with HFpEF experience shortness of breath (especially during exercise or exertion, or when lying down), fast or irregular heartbeats, chest pain, weakness, and swelling in their lower legs. Researchers estimate that this condition affects around 32 million people worldwide and 3 million in the United States.1 It represents approximately 50% of the overall heart failure population, and its prevalence is expected to rise with the aging population.2
Earlier clinical studies have suggested that personalized elevated pacing rates could positively impact patient outcomes for those with HFpEF and bradycardia.3 The ELEVATE-HFpEF trial seeks to validate this in a broader group, using personalized pacing settings and conduction system pacing in patients who do not have a current indication for pacing.
"Millions of people around the world have HFpEF and experience reduced quality of life and increased risk of hospitalization and death. For some patients with HFpEF, current medical management alone may not be enough to alleviate their symptoms, and new treatment strategies are needed to improve these outcomes," said Brett Atwater, M.D., cardiac electrophysiologist at Inova Schar Heart and Vascular, Fairfax, Va., and global co-principal investigator on the ELEVATE-HFpEF trial. "This pivotal study will provide important insights into the potential of personalized pacing as an innovative approach for improving quality of life and cardiac outcomes for patients living with this serious and often deadly condition."
ELEVATE-HFpEF is a randomized, controlled, double-blinded, multi-center, global study that will enroll up to 700 participants across the North America, Europe, Middle East, Africa, Australia, and Asia Pacific regions. Study participants will receive conduction system pacing with a commercially available Medtronic pacemaker and SelectSecure™ MRI SureScan™ Model 3830 pacing leads. Conduction system pacing is a type of pacing therapy that taps into the heart's natural electrical pathways to stimulate a coordinated heartbeat, closely mimicking the heart's physiologic contractions. Physicians will program study patients' pacemakers to either a personalized cardiac pacing rate using an allometric scale, which is based on each individual's body size, and baseline left ventricular ejection fraction (a measure of how well the heart pumps blood); or to provide backup pacing if needed (control group). Health care providers participating in the study will follow patients for one year as researchers assess the efficacy and safety of this investigative treatment.
The first study implant procedures were performed at The Alfred Hospital in Melbourne, Australia and at The Ohio State University Wexner Medical Center in Columbus, Ohio. The global co-principal investigators for the study are Dr. Atwater and Harriette Van Spall, M.D., Associate Professor of Medicine at McMaster University, Canada, and Director of Implementation Science at Baim Institute for Clinical Research, Hamilton, Ontario, Canada.
"Since Medtronic invented the first battery-powered pacemaker nearly 70 years ago, we have continually worked with physician and patient communities to both innovate new pacing technology and deliver landmark clinical evidence to broaden the types of patients that may benefit from pacing," said Robert C. Kowal, M.D., Ph.D., general manager for Cardiac Pacing Therapies in the Cardiac Rhythm Management business, which is part of the Medtronic Cardiovascular Portfolio. "With the initiation of this study, we are opening the door to our next era – pacing therapy as a potentially safe and effective intervention to improve the lives of patients with HFpEF, a cardiac condition that pacemakers do not currently treat."
About Medtronic
Bold thinking. Bolder actions. We are Medtronic. Medtronic plc, headquartered in Galway, Ireland, is the leading global healthcare technology company that boldly attacks the most challenging health problems facing humanity by searching out and finding solutions. Our Mission — to alleviate pain, restore health, and extend life — unites a global team of 95,000+ passionate people across more than 150 countries. Our technologies and therapies treat 70 health conditions and include cardiac devices, surgical robotics, insulin pumps, surgical tools, patient monitoring systems, and more. Powered by our diverse knowledge, insatiable curiosity, and desire to help all those who need it, we deliver innovative technologies that transform the lives of two people every second, every hour, every day. Expect more from us as we empower insight-driven care, experiences that put people first, and better outcomes for our world. In everything we do, we are engineering the extraordinary. For more information on Medtronic (NYSE: MDT), visit and follow Medtronic on LinkedIn.
Any forward-looking statements are subject to risks and uncertainties such as those described in Medtronic's periodic reports on file with the Securities and Exchange Commission. Actual results may differ materially from anticipated results.
Contacts:
Emily Dornfeld
Public Relations
+1-763-242-8241
Ryan Weispfenning
Investor Relations
+1-763-505-4626
1 Redfield MM, Borlaug BA. Heart Failure With Preserved Ejection Fraction: A Review. JAMA. 2023 Mar 14;329(10):827-838. doi: 10.1001/jama.2023.2020. PMID: 36917048.
2 Hamo, C. E., et al. (2024). "Heart failure with preserved ejection fraction." Nat Rev Dis Primers 10(1): 55.
3 Infeld, M., et al. (2023). "Effect of Personalized Accelerated Pacing on Quality of Life, Physical Activity, and Atrial Fibrillation in Patients With Preclinical and Overt Heart Failure With Preserved Ejection Fraction: The myPACE Randomized Clinical Trial." JAMA Cardiol 8(3): 213–221.
SOURCE Medtronic plc
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
临床研究
2025-09-08
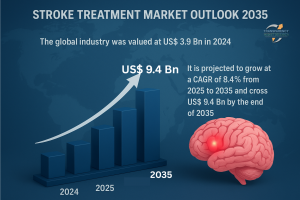
Stroke Treatment Market to Expand from USD 3.9 Billion in 2024 to USD 9.4 Billion by 2035 Driven by Rising Global Stroke Incidence -Transparency Market Research
Global Stroke Treatment Market to Achieve USD 9.4 Billion by 2035 as Healthcare Systems Focus on Neurological Care”
— Latest Report by Transparency Market Research, Inc.
WILMINGTON, DE, UNITED STATES, September 8, 2025 /
EINPresswire.com
/ --
Stroke Treatment Market
Outlook 2035
The global stroke treatment market is poised for strong growth, supported by rising prevalence of stroke cases, advancements in treatment therapies, and increasing healthcare investments. Valued at US$ 3.9 Bn in 2024, the industry is forecast to grow at a CAGR of 8.4% from 2025 to 2035, reaching over US$ 9.4 Bn by 2035. Expanding access to advanced neurovascular devices and growing emphasis on early intervention are key drivers of this growth.
Don't miss out on the Latest Market Intelligence. Get your Sample Report Copy Today@
https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=73917
Industry Overview
Stroke treatment involves the use of drugs, medical devices, and therapeutic procedures to restore blood flow, minimize brain damage, and enhance patient recovery. With stroke being a major public health burden, increasing awareness campaigns and investments in healthcare infrastructure are propelling the adoption of effective treatment solutions.
The growing demand for thrombolytic drugs, mechanical thrombectomy devices, and rehabilitation therapies is shaping the industry’s growth. Furthermore, the integration of AI-based diagnostic imaging, telemedicine, and precision medicine is revolutionizing stroke management.
Analysis of Key Players in the Stroke Treatment Market
The stroke treatment market is highly competitive, with several global pharmaceutical and biotechnology companies playing a key role.
Leading players include
• Bristol-Myers Squibb Company
• Sanofi
• F. Hoffmann-La Roche (Genentech)
• Daiichi Sankyo Company, Limited
• AstraZeneca
• Biogen Inc.
• Johnson & Johnson Services, Inc.
• Bayer AG
• Eli Lilly and Company
• Mylan
• Teva Pharmaceuticals
• Zydus Cadila
• Sun Pharmaceutical
These companies have been profiled in the stroke treatment market research report on the basis of parameters such as company overview, financial performance, business strategies, product portfolio, business segments, and recent developments.
Key Developments in the Stroke Treatment Market
• October 2024 – Royal Philips announced a collaboration with Medtronic Neurovascular to improve awareness and expand access to timely stroke diagnosis and treatment solutions.
• February 2024 – Basking Biosciences, a clinical-stage biopharmaceutical firm focused on novel acute thrombolytic therapies for stroke, completed a US$ 55 million financing round. The investment was led by ARCH Venture Partners, with participation from Insight Partners, Platanus, Solas BioVentures, and RTW Investments, along with existing investors including Longview Ventures, Rev1 Ventures, and The Ohio State University.
Key Growth Drivers
1. Rising Global Stroke Incidence – Increasing cases due to aging populations, hypertension, and diabetes.
2. Advancements in Medical Devices – Mechanical thrombectomy and clot retrieval devices improving survival rates.
3. Drug Innovation – Development of novel anticoagulants and thrombolytics enhancing treatment efficiency.
4. Telemedicine & AI Diagnostics – Early detection and faster treatment decisions through advanced imaging and digital health tools.
5. Government & NGO Initiatives – Stroke awareness campaigns and healthcare funding boosting accessibility.
Discuss Implications for Your Industry Request Sample Research PDF@
https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=73917
Market Restraints & Challenges
• High Treatment Costs – Advanced therapies and devices remain expensive.
• Limited Access in Developing Regions – Lack of stroke-ready hospitals and trained professionals.
• Risk of Side Effects – Certain anticoagulants and thrombolytic drugs pose bleeding risks.
• Regulatory Barriers – Lengthy approval processes for new drugs and devices.
Market Segmentation
By Treatment Type
• Medication (Anticoagulants, Antiplatelets, Thrombolytics, Others)
• Surgery & Endovascular Procedures
• Rehabilitation Therapy
By Stroke Type
• Ischemic Stroke
• Hemorrhagic Stroke
• Transient Ischemic Attack (TIA)
By End User
• Hospitals
• Specialty Clinics
• Ambulatory Surgical Centers
• Rehabilitation Centers
By Region
• North America
• Europe
• Asia-Pacific
• Latin America
• Middle East & Africa
Market Trends & Innovations
1. AI-Powered Stroke Diagnosis – Faster and more accurate imaging-based decisions.
2. Minimally Invasive Stroke Surgeries – Increasing adoption of advanced thrombectomy devices.
3. Personalized Medicine – Genetic profiling aiding targeted treatment approaches.
4. Stroke Rehabilitation Robotics – Enhancing post-stroke recovery outcomes.
5. Collaborations & Clinical Trials – Partnerships among pharma and medtech firms to expand therapeutic options.
Why Invest in This Report?
• Accurate market size and CAGR projections through 2035.
• In-depth insights into growth drivers, restraints, and opportunities.
• Competitive analysis of global players and their innovation strategies.
• Emerging opportunities across high-growth regions and therapeutic areas.
• Coverage of regulatory and reimbursement frameworks.
Future Outlook
The global stroke treatment market is projected to reach US$ 9.4 Bn by 2035, driven by the growing prevalence of stroke cases, innovations in treatment methods, and improved accessibility to healthcare.
Future trends shaping the market include:
• Smart wearable devices for early detection and prevention.
• AI-integrated imaging platforms for quicker clinical decision-making.
• Regenerative medicine & stem-cell therapies as potential future stroke treatments.
• Expansion in emerging markets, increasing affordability and access.
To buy this comprehensive market research report, click here to inquire@
https://www.transparencymarketresearch.com/checkout.php?rep_id=73917<ype=S
Important FAQs with Answers
Q1. What was the global market size of stroke treatment in 2024?
A1. The market was valued at US$ 3.9 Bn in 2024.
Q2. What is the projected market size by 2035?
A2. The market is expected to surpass US$ 9.4 Bn by 2035.
Q3. What is the CAGR for 2025–2035?
A3. The industry is projected to grow at a CAGR of 8.4%.
Q4. What are the main treatment types?
A4. Medications, surgery & endovascular procedures, and rehabilitation therapies.
Q5. Who are the key players in the market?
A5. Leading companies include Medtronic, Johnson & Johnson, Bayer, Boehringer Ingelheim, Abbott, and Bristol Myers Squibb.
Q6. What innovations are shaping the future of stroke treatment?
A6. Key innovations include AI-powered imaging, thrombectomy devices, smart wearables, and regenerative therapies.
Related Links
• Neuroregeneration Therapy Market -
https://www.transparencymarketresearch.com/neuroregeneration-therapy-market.html
• Aldosterone Receptor Antagonists Market -
https://www.transparencymarketresearch.com/aldosterone-receptor-antagonists-market.html
• Vasopressin Antagonists Market -
https://www.transparencymarketresearch.com/vasopressin-antagonists-market.html
• Parkinson’s Disease Therapeutics Market -
https://www.transparencymarketresearch.com/parkinsons-disease-therapeutics-market.html
• Alzheimer’s Drugs Market -
https://www.transparencymarketresearch.com/alzheimers-drugs-market.html
• Alzheimer’s Disease Biomarkers Market -
https://www.transparencymarketresearch.com/alzheimers-disease-biomarkers-market.html
• Multiple Sclerosis Drugs Market -
https://www.transparencymarketresearch.com/multiple-sclerosis-drugs.html
• Amyotrophic Lateral Sclerosis Market -
https://www.transparencymarketresearch.com/amyotrophic-lateral-sclerosis.html
• Hormone Replacement Therapy Market -
https://www.transparencymarketresearch.com/hormone-replacement-therapy-market.html
• Neurology Devices Market -
https://www.transparencymarketresearch.com/neurology-devices-market.html
• Neuromodulation Devices Market -
https://www.transparencymarketresearch.com/global-neuromodulation-devices-market.html
• Neurotherapeutic Drugs Market -
https://www.transparencymarketresearch.com/neurotherapeutic-drugs-market.html
• Neuromorphic Chip Market -
https://www.transparencymarketresearch.com/neuromorphic-chip-market.html
• Neurovascular Stent Retriever Technology Market -
https://www.transparencymarketresearch.com/neurovascular-stent-retrievers-market.html
About Us Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. The firm scrutinizes factors shaping the dynamics of demand in various markets. The insights and perspectives on the markets evaluate opportunities in various segments. The opportunities in the segments based on source, application, demographics, sales channel, and end-use are analysed, which will determine growth in the markets over the next decade.
Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision-makers, made possible by experienced teams of Analysts, Researchers, and Consultants. The proprietary data sources and various tools & techniques we use always reflect the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in all of its business reports.
Contact Us
Transparency Market Research Inc.
CORPORATE HEADQUARTER DOWNTOWN,
1000 N. West Street,
Suite 1200, Wilmington, Delaware 19801 USA
Tel: +1-518-618-1030
USA - Canada Toll Free: 866-552-3453
Atil Chaudhari
Transparency Market Research Inc.
+ +1 518-618-1030
email us here
Visit us on social media:
LinkedIn
YouTube
X
Other
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
100 项与 Medtronic Plc 相关的药物交易
登录后查看更多信息
100 项与 Medtronic Plc 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2025年10月11日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
临床2期
1
4
批准上市
其他
10
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或

营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或

科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用





