预约演示
更新于:2025-07-15
Tenofovir Disoproxil Fumarate
富马酸替诺福韦二吡呋酯
更新于:2025-07-15
概要
基本信息
非在研机构 |
最高研发阶段批准上市 |
首次获批日期 美国 (2001-10-26), |
最高研发阶段(中国)批准上市 |
特殊审评加速批准 (美国)、孤儿药 (美国)、孤儿药 (日本)、优先审评 (中国) |
登录后查看时间轴
结构/序列
分子式C23H34N5O14P |
InChIKeyVCMJCVGFSROFHV-WZGZYPNHSA-N |
CAS号202138-50-9 |
关联
336
项与 富马酸替诺福韦二吡呋酯 相关的临床试验NCT06990438
Effect of Long-Term Use of Tenofovir (TDF) on Bone Density in Patients With Chronic Hepatitis B
This study aims to evaluate the effect of long-term use of Tenofovir Disoproxil Fumarate (TDF) on bone density in patients with chronic hepatitis B virus (HBV) infection. Tenofovir is a widely used antiviral medication for the treatment of HBV. While it is generally well tolerated, some studies have reported potential adverse effects on bone mineral density, particularly with long-term use.
The objective of this research is to assess whether extended TDF therapy is associated with reduced bone density or increased risk of osteopenia or osteoporosis in adult patients with chronic HBV infection. The study will involve clinical evaluation and radiological assessment of bone health using dual-energy X-ray absorptiometry (DEXA) scans, as well as relevant biochemical markers.
This investigation will provide important data on the long-term safety profile of Tenofovir in relation to bone health and help guide future clinical decisions for the management of chronic hepatitis B.
The objective of this research is to assess whether extended TDF therapy is associated with reduced bone density or increased risk of osteopenia or osteoporosis in adult patients with chronic HBV infection. The study will involve clinical evaluation and radiological assessment of bone health using dual-energy X-ray absorptiometry (DEXA) scans, as well as relevant biochemical markers.
This investigation will provide important data on the long-term safety profile of Tenofovir in relation to bone health and help guide future clinical decisions for the management of chronic hepatitis B.
开始日期2025-05-22 |
申办/合作机构 |
NCT06896630
Effect of Long-Term Use of Tenofovir (TDF) on Renal Function in Patients With Chronic Hepatitis B
Effect of Long-Term Use of Tenofovir (TDF) on Renal Function in Patients with Chronic Hepatitis B Over a Five-Year Period
开始日期2025-04-10 |
申办/合作机构 |
ChiCTR2500098499
Effects of TDF/3TC/ANV on cardiovascular disease risk score and intestinal microecology in HIV-1 patients with dyslipidemia
开始日期2025-03-10 |
申办/合作机构 |
100 项与 富马酸替诺福韦二吡呋酯 相关的临床结果
登录后查看更多信息
100 项与 富马酸替诺福韦二吡呋酯 相关的转化医学
登录后查看更多信息
100 项与 富马酸替诺福韦二吡呋酯 相关的专利(医药)
登录后查看更多信息
6,812
项与 富马酸替诺福韦二吡呋酯 相关的文献(医药)2025-12-31·HIV Research & Clinical Practice
Evaluating adherence in an active-controlled HIV pre-exposure prophylaxis trial (PrEPVacc) to inform the estimation of HIV incidence in a counterfactual placebo arm
Article
BACKGROUND:
Inferring the counterfactual placebo HIV incidence using the estimated effectiveness of Emtricitabine/Tenofovir (TDF/FTC) in active-controlled pre-exposure prophylaxis (PrEP) trials has been suggested. However, it has not yet been widely applied. In this article, we evaluate adherence to TDF/FTC in the PrEPVacc trial (NCT04066881) and consider how such adherence data could be used to estimate the effectiveness of TDF/FTC and subsequently, HIV incidence in a counterfactual placebo arm in a predominantly female population.
METHODS:
From December 2020 to March 2023 participants were recruited into the trial which included a comparison of Emtricitabine/Tenofovir Alafenamide (TAF/FTC) to TDF/FTC as PrEP over 26 weeks of follow-up, in Uganda, Tanzania, and South Africa. PrEP adherence was assessed in various ways.
RESULTS:
Of 697 participants dispensed TDF/FTC, 87% were female, 54% were ≥ 25 years, and 59% were sex workers. In a random sample (41%) assessed at visit 6 (week 8), 76% had detectable TFV-DP levels, with 22% reaching levels consistent with ≥2 pills/week. Males, Verulam and Mbeya participants, those ≥ 25 years, not single, subsistence fisheries workers, and those who had any STI at baseline were more likely to have higher adherence. Of those assessed at visit 6, 29% were identified as white coat dosing. Estimated (crude) HIV incidence risk reduction ranged from 10% to 65%.
CONCLUSIONS:
TDF/FTC adherence in the PrEPVacc trial was low, with considerable levels of white coat dosing. Inferring the counterfactual placebo HIV incidence using the estimated effectiveness of TDF/FTC is a promising approach, however, the approach requires further elaboration and evaluation.
2025-12-31·HIV Research & Clinical Practice
A combined prospective and retrospective comparative study evaluating renal outcomes after switching from TDF + FTC + EFV to TDF/3TC/DTG (TLD)
vs.
DTG + 3TC in virologically suppressed Thai people with HIV
Article
作者: Putcharoen, Opass ; Kanogtorn, Sathaporn ; Burimat, Phattarapon ; Patamatamkul, Samadhi
BACKGROUND:
TDF/3TC/DTG (TLD) has been adopted as the first-line therapy for all people with HIV according to the WHO 2019 and Thai HIV guidelines. As a result, people with HIV in Thailand on TDF/FTC/EFV are switched to TLD. However, increasing data demonstrate the efficacy and renal safety of the TDF-sparing dual therapy with DTG + 3TC compared with TDF-based combination ART (cART) as a switching therapy. A direct comparison of estimated glomerular filtration rates (eGFR) among people with HIV treated with TDF-based regimens who switch to TLD vs. DTG + 3TC is yet to be made.
METHODS:
We enrolled virologically suppressed people with HIV aged ≥18 years currently on TDF + FTC + EFV to either switch to TLD or DTG + 3TC at two tertiary care hospitals. The switching regimen was chosen at the physicians' discretion. The primary outcome was the change in eGFR, calculated by creatinine at 24 weeks. Secondary outcomes included changes in LDL, body weight, and BMI at 24 weeks.
RESULTS:
Among 53 recruited participants, 28 and 15 completed the second follow-up in the TLD and DTG + 3TC groups, respectively. The mean age was higher in the TLD group compared to the DTG + 3TC group (Table 1). The median time from HIV diagnosis to switching was 8.5 years. The eGFR reduction was significantly greater in the TLD group than in the DTG + 3TC group: -17.24 ± 9.24 vs. -8.4 ± 9.03 mL/min/1.73 m2 (p = 0.004). All participants in both groups achieved virological suppression. There was no significant change in CD4 counts between the two groups. Switching to DTG + 3TC was associated with a significantly smaller decline in eGFR post-switch (mean difference: 6.216 mL/min/1.73 m2; 95% CI 0.169-12.263; p = 0.044) compared to those who switched to TLD.
CONCLUSIONS:
There was a significant reduction in eGFR among people with HIV who switched to TLD compared to those switched to DTG + 3TC. Changes in LDL and BMI were comparable between groups. Dual therapy with DTG + 3TC may be a preferred switching option over TLD for individuals with renal safety concerns. Further randomized prospective trials with longer follow-up are warranted to confirm these findings.
2025-12-01·SPECTROCHIMICA ACTA PART A-MOLECULAR AND BIOMOLECULAR SPECTROSCOPY
Development of colorimetric immunochromatographic assay based on different morphologies of gold nanoparticles for the detection of triadimefon
Article
作者: Luo, Ruifeng ; Zhu, Zixin ; Dong, Sa ; Feng, Jianguo ; Zhang, Yu ; Shi, Qiuyun ; Wang, Yulong
Triadimefon (TDF) is a highly effective, low toxicity, broad-spectrum triazole fungicide with strong systemic absorption. Excessive residues in tobacco and vegetables can affect human health. Therefore, it is urgent to establish a simple, rapid, and sensitive detection method for residual analysis of TDF in tobacco and vegetables. In this study, colloidal gold nanoparticles (Au NPs), gold nanoflowers (Au NFs), and gold nanorods (Au NRs) were synthesized and combined with anti-TDF monoclonal antibodies (mAb) to prepare probes for immunochromatographic strips (ICS). The lowest visual detection limits (vLODs) of the test strips using AuNPs-mAb, AuNFs-mAb, and AuNRs-mAb as probes were 7.8, 15.6, and 15.6 ng/mL, respectively. The three test strips showed no significant cross reactivity with the four structural analogues of tebuconazole, hexaconazole, myclobutanil, and difenoconazole, indicating that all three test strips have good specificity. To assess the presence of 0 and 62.5 ng/mL of TDF, ten test strips were used, and the results showed perfect accuracy with no false positives or false negatives. In addition, three test strips (same batch) were utilized to detect TDF (0 and 62.5 ng/mL) after storage for three months, and the results were still accurate, indicating that all three kinds of test strips have good repeatability and stability. The addition and recovery tests of TDF were conducted on cucumber and tobacco leaf samples (0, 2.5, 17.5, and 65 μg/kg), and the test results revealed that the strips performed with high accuracy. This study established three colorimetric test strips based on three different morphologies of gold nanoparticles, which possess the benefits of quick processing times, high cost-effectiveness, and minimal technical requirements, making them suitable for rapid on-site qualitative detection of TDF.
299
项与 富马酸替诺福韦二吡呋酯 相关的新闻(医药)2025-07-14
In collaboration with the Gates Foundation, Merck advances MK-8527 pre-exposure prophylaxis (PrEP) clinical trials globally
RAHWAY, NJ, USA I July 14, 2025 I
Merck (NYSE: MRK), known as MSD outside of the United States and Canada, today announced the initiation of the EXPrESSIVE Phase 3 clinical trials, evaluating the safety and efficacy of MK-8527, an investigational once-monthly, oral nucleoside reverse transcriptase translocation inhibitor (NRTTI) for HIV pre-exposure prophylaxis (PrEP). The EXPrESSIVE-11 (MK-8527-011,
NCT 07044297
) trial will evaluate the safety and efficacy of MK-8527 among people with greater likelihood of HIV-1 exposure in 16 countries and will begin enrolling in August 2025. In collaboration with the Gates Foundation, the EXPrESSIVE-10 (MK-8527-010) trial will evaluate the safety and efficacy of MK-8527 in women and adolescent girls in sub-Saharan Africa and will begin enrolling in the next few months.
“According to UNAIDS, 1.3 million people acquired HIV in 2023, highlighting the continued need for new PrEP options like our investigational once-monthly, oral PrEP candidate MK-8527, especially among women in sub-Saharan Africa and men who have sex with men, who experience disproportionately high rates of HIV,” said Dr. Eliav Barr, senior vice president, head of global clinical development and chief medical officer, Merck Research Laboratories. “Our collaboration with the Gates Foundation will help us explore the potential of MK-8527 to contribute to global efforts to reduce the number of HIV infections and help support opportunities to accelerate access around the world.”
The decision to initiate the Phase 3 clinical trial program was supported by the results of a double-blind, multicenter, Phase 2 trial (MK-8527-007,
NCT 06045507
) examining the safety and pharmacokinetics of MK-8527. The study enrolled 350 participants, 18–65 years of age, with low likelihood of HIV-1 exposure, who were randomized 2:2:2:1 to receive MK-8527 (3, 6, or 12 mg) or placebo once monthly for six months. In the trial, the rates of adverse events were similar among those in the MK-8527 arms and those in the placebo arm, and no clinically meaningful changes were seen in laboratory tests, including total lymphocyte and CD4 T-cell counts. The pharmacokinetics of MK-8527 and MK-8527-TP, the active form of MK-8527, support the continued development of MK-8527 as an oral, once-monthly option for PrEP. Results from the Phase 2 trial were highlighted in the opening press conference for IAS 2025, the 13th International AIDS Society Conference on HIV Science in Kigali, Rwanda, on Monday, July 14, at 15:30 – 16:30 CAT, titled “Breakthroughs Amid Crisis: The Future of HIV Innovation.” These research findings will be further detailed in a late-breaker oral session on Wednesday, July 16, at 12:15 – 13:15 CAT.
“Scientific advances against HIV have brought us further than ever imagined and are ushering in a new era in HIV prevention,” said Trevor Mundel, president of global health at the Gates Foundation. “With only 18% of global PrEP need currently met, there is a clear and urgent need for options like MK-8527 that may offer the ability to prevent infection. These Phase 3 trials are a key step toward translating progress into longer-acting options that could help turn the tide on HIV.”
In the EXPrESSIVE-10 trial, the International Clinical Research Center (ICRC) within the University of Washington Department of Global Health, in partnership with the University of Alabama at Birmingham, will receive grant funding from the Gates Foundation to support ICRC’s collaboration with 31 clinical research trial sites in Kenya, South Africa, and Uganda; sites will inform and engage communities and recruit, enroll, and follow women who participate in the EXPrESSIVE-10 trial. Merck will be the trial sponsor, gaining regulatory and customs approvals, and providing operational expertise and resources for management of the trials. The Gates Foundation will also provide support for global community advisory groups, which will offer insight into community perspectives on a monthly PrEP pill, participant recruitment materials and strategies, and cultural considerations for these trials. Separately, the Gates Foundation will provide grant funding for EXPrESSIVE-11 to support the establishment of a community advisory group and the development of recruiting and retention materials only.
About the EXPrESSIVE MK-8527 Phase 3 Clinical Trial Program
The EXPrESSIVE-11 trial (MK-8527-011,
NCT 07044297
) is a randomized, active-controlled study enrolling 4,390 sexually active people who could benefit from PrEP across trial sites in 16 countries. The primary objective of the study is to evaluate the efficacy, safety, and tolerability of once-monthly oral MK-8527 compared to daily emtricitabine/tenofovir disoproxil fumarate (FTC/TDF) as assessed by the incidence rate per year of adjudicated HIV-1 infections. The trial will begin enrolling in August 2025.
The EXPrESSIVE-10 trial (MK-8527-010) is a randomized, active-controlled study enrolling 4,580 sexually active women aged 16 to 30 in Kenya, South Africa, and Uganda. The primary objective of the study is to evaluate the efficacy, safety, and tolerability of once-monthly oral MK-8527 compared to daily emtricitabine/tenofovir disoproxil fumarate (FTC/TDF) as assessed by the incidence rate per year of adjudicated HIV-1 infections. The study will begin enrolling in the next few months.
About MK-8527
MK-8527 is being evaluated as a potential once-monthly oral prevention option for HIV-1. MK-8527 inhibits reverse transcriptase through multiple mechanisms of action, including inhibition of translocation and delayed chain termination. For an overview of Merck’s complete HIV treatment and prevention clinical development program, please click
here
.
Merck’s Commitment to HIV
For more than 35 years, Merck has been committed to scientific research and discovery in HIV, leading to scientific breakthroughs that have helped change HIV treatment. Our work has helped pioneer the development of new options across multiple drug classes to help those impacted by HIV. Today, we are developing a series of antiviral options designed to help people manage their HIV and to help prevent HIV, with the goal of reducing the growing burden of HIV worldwide. We want to ensure people are not defined by HIV, and our work focuses on transformational innovations, collaborations with others in the global HIV community, and access initiatives aimed at helping to end the HIV epidemic for everyone.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable, and healthy future for all people and communities. For more information, visit
www.merck.com
and connect with us on
X (formerly Twitter)
,
Facebook
,
Instagram
,
YouTube
, and
LinkedIn
.
SOURCE:
Merck
临床2期临床结果临床3期
2025-07-10
Doravirine/islatravir would be the first FDA-approved two-drug regimen without an integrase inhibitor that demonstrated non-inferior efficacy and a generally comparable safety profile to the three-drug InSTI-based regimen, BIC/FTC/TAF, in a Phase 3 pivotal trial
RAHWAY, NJ, USA I July 10, 2025 I
Merck (NYSE: MRK), known as MSD outside of the United States and Canada, today announced that the U.S. Food and Drug Administration (FDA) has accepted for review the New Drug Application (NDA) for doravirine/islatravir (DOR/ISL), an investigational, once-daily, oral, two-drug regimen for adults with HIV-1 infection that is virologically suppressed on antiretroviral therapy. The FDA has set a target action date of April 28, 2026, for the application under the Prescription Drug User Fee Act (PDUFA).
“Merck has been at the forefront of HIV research for more than 35 years and we are pleased to continue our work to innovate and deliver new options that aim to meet the needs of the HIV community,” said Dr. Eliav Barr, senior vice president, head of global clinical development and chief medical officer, Merck Research Laboratories. “The health needs of people living with HIV often change over time – whether it’s managing comorbidities or navigating complex medication regimens. We believe DOR/ISL, if approved, will represent an important new complete regimen option designed to help meet their diverse needs.”
The NDA is based on findings at Week 48 of two pivotal Phase 3 clinical trials (
MK-8591A-051
and
MK-8591A-052
) where DOR/ISL was demonstrated to be non-inferior to baseline antiretroviral therapy (bART) in the open-label trial MK-8591A-051 and non-inferior to bictegravir/emtrictabine/tenofovir alafenamide
i
[BIC/FTC/TAF (50mg/200mg/25mg)] in the double-blind trial MK-8591A-052. Across both trials, the safety profile of DOR/ISL was generally comparable to comparator baseline antiretroviral regimens in trial MK-8591A-051 and BIC/FTC/TAF in trial MK-8591A-052. Data from these trials
were presented
during the 2025 Conference on Retroviruses and Opportunistic Infections (CROI) in San Francisco.
About the Phase 3 Trial MK-8591A-051
MK-8591A-051 is a Phase 3, open-label, randomized, active-controlled clinical trial evaluating the efficacy and safety of a switch to investigational, oral, once-daily DOR/ISL (100mg/0.25mg) in adults with HIV-1 infection that has been virologically suppressed using ART. The primary efficacy endpoint was the percentage of participants with HIV-1 RNA ≥50 copies/mL at Week 48 (non-inferiority margin 4%). In this trial, 551 adults with HIV-1 RNA <50 copies/mL for three months or more on oral 2- or 3-drug ART, with no history of treatment failure and no known virologic resistance to DOR, were randomized 2:1 and switched to DOR/ISL (n=366) or continued bART (n=185), stratified by bART regimen. The median age of participants was 51 years; 39.7% were assigned female sex at birth, 45.4% were Black or African American, and 14.5% were Hispanic or Latine. At baseline, 64.2% were treated with an InSTI-based regimen, 30.3% with an NNRTI-based regimen, and 5.4% with a protease inhibitor (PI)-based regimen, with median duration on current ART of 3.8 years (IQR 2.0-6.3).
About the Phase 3 Trial MK-8591A-052
MK-8591A-052 is a Phase 3, double-blind, randomized, active-controlled clinical trial to evaluate the efficacy and safety of a switch to investigational, oral, once-daily DOR/ISL (100mg/0.25mg) in adults with HIV-1 infection that has been virologically suppressed on BIC/FTC/TAF (50mg/200mg/25mg). The primary efficacy endpoint was the percentage of participants with HIV-1 RNA ≥50 copies/mL at Week 48 (non-inferiority margin 4%). In this trial, 513 adults with HIV-1 who had virologic suppression for three months or more on BIC/FTC/TAF, no history of treatment failure and no known resistance to DOR were randomized (2:1) and switched to DOR/ISL (n=342) or continued treatment with BIC/FTC/TAF (n=171). The median age of participants was 47 years; 21.4% were assigned female sex at birth, 30.8% were Black or African American, and 22.8% were Hispanic or Latine. The median duration of BIC/FTC/TAF treatment prior to trial enrollment was 3.4 years (IQR 2.0-5.0).
About Islatravir (MK-8591) and Merck’s HIV Research
Islatravir (MK-8591), Merck’s investigational nucleoside reverse transcriptase translocation inhibitor (NRTTI), blocks HIV-1 replication by multiple mechanisms including inhibition of reverse transcriptase translocation, resulting in immediate chain termination and induction of structural changes in the viral DNA, resulting in delayed chain termination. Islatravir is under evaluation in multiple ongoing early and late-stage clinical trials in combination with other antiretrovirals for potential daily and once-weekly treatments for HIV-1, with islatravir serving as the anchor medicine in the treatment regimens based on its potency and resistance profile. In addition to the MK-8591A-051 and MK-8591A-052 trials, ongoing Phase 3 trials of daily DOR/ISL (100mg /0.25mg) include
MK-8591A-053
in people with HIV who had not previously received treatment (treatment-naïve), and
MK-8591A-054
evaluating open-label DOR/ISL (100 mg/0.25 mg) in individuals who participated in earlier Phase 3 trials of DOR/ISL (100 mg/0.75 mg). Islatravir in combination with Gilead’s lenacapavir is in Phase 3 development as a novel oral once-weekly treatment for HIV-1, and islatravir in combination with our company’s investigational non-nucleoside reverse transcriptase inhibitor (NNRTI) ulonivirine (MK-8507) is in Phase 2 development as an oral once-weekly treatment.
Merck’s commitment to researching NRTTIs includes MK-8527, an investigational, novel oral, once-monthly NRTTI candidate that is in Phase 2 development for HIV-1 pre-exposure prophylaxis (PrEP). For an overview of Merck’s HIV treatment and prevention clinical development program, please click
here
.
Merck’s Commitment to HIV
For more than 35 years, Merck has been committed to scientific research and discovery in HIV leading to scientific breakthroughs that have helped change HIV treatment. Our work has helped pioneer the development of new options across multiple drug classes to help those impacted by HIV. Today, we are developing a series of antiviral options designed to help people manage their HIV and to help prevent HIV, with the goal of reducing the growing burden of infection worldwide. We want to ensure people are not defined by HIV, and our work focuses on transformational innovations, collaborations with others in the global HIV community, and access initiatives aimed at helping to end the HIV epidemic for everyone.
Indications and usage for PIFELTRO
®
(doravirine) and DELSTRIGO
®
(doravirine, lamivudine, and tenofovir disoproxil fumarate) in the U.S.
PIFELTRO is indicated in combination with other antiretroviral (ARV) agents for the treatment of HIV-1 infection in adult patients with no prior ARV treatment history or to replace the current ARV regimen in those who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) on a stable ARV regimen with no history of treatment failure and no known substitutions associated with resistance to doravirine.
DELSTRIGO is indicated as a complete regimen for the treatment of HIV-1 infection in adult patients with no prior ARV treatment history or to replace the current ARV regimen in those who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) on a stable ARV regimen with no history of treatment failure and no known substitutions associated with resistance to the individual components of DELSTRIGO.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities. For more information, visit
www.merck.com
and connect with us on
X (formerly Twitter)
,
Facebook
,
Instagram
,
YouTube
and
LinkedIn
.
SOURCE:
Merck
临床3期临床2期临床结果申请上市
2025-07-08
July 8, 2025 6:45 am ET
RAHWAY, N.J.--(BUSINESS WIRE)-- Merck (NYSE: MRK), known as MSD outside of the United States and Canada, announced today that new data from its research pipeline for HIV prevention and treatment will be presented at the 13th International AIDS Society Conference on HIV Science (IAS 2025) taking place July 13-17, 2025, in Kigali, Rwanda.
Merck will share new scientific findings from its HIV clinical development programs, including Phase 2 data on the safety and pharmacokinetics of MK-8527, an investigational, novel nucleoside reverse transcriptase translocation inhibitor (NRTTI), dosed orally once monthly, in development for the prevention of HIV as pre-exposure prophylaxis (PrEP).
HIV clinical data presented at IAS 2025 from three Phase 3 trials (P051: NCT05631093; P052: NCT05630755; P054: NCT05766501) will examine the impact of preexisting resistance-associated mutations (RAMs) in proviral DNA on the virologic response to doravirine/islatravir (DOR/ISL), with a primary focus on M184I/V in proviral DNA.
Data will also be presented on the clinical development program for a once-weekly oral combination of islatravir and ulonivirine (ISL/ULO) for the treatment of adults with HIV-1 infection. Data from two Phase 1 trials evaluating the safety and tolerability of weekly ulonivirine and drug interactions between islatravir and ulonivirine dosed weekly in adults without HIV and a Phase 2b dose-ranging study utilizing a higher (20mg) dose of islatravir and 3 doses of ulonivirine in treatment naive adults living with HIV-1 will be presented. Based on results from these trials and pharmacokinetic modeling that will also be presented, a once-weekly oral combination of islatravir (2mg) and ulonivirine (200mg) (MK-8591B) is moving forward in clinical development.
“As we continue to advance our HIV research, we are excited to present data from across our HIV pipeline at IAS 2025, including new data from our nucleoside reverse transcriptase translocation inhibitors development programs,” said Dr. Elizabeth Rhee, vice president, global clinical development, Merck Research Laboratories. “With daily, weekly and monthly oral regimens in development, we aim to offer choices that can help address the evolving needs of individuals living with or impacted by HIV.”
Select abstracts in the IAS 2025 program include:
Abstract Title and Author
Date
HIV PREVENTION
Safety and Pharmacokinetics of Once-Monthly MK-8527: a Phase 2 Study in Adults at Low Risk of HIV-1 Exposure, Mayer, K, et al.
Late-Breaker
Oral Presentation
Wednesday, July 16, 2025
(15:00 – 16:00 CAT)
HIV TREATMENT
Switching to Doravirine/Islatravir (100mg/0.25mg) Once Daily Maintained Viral Suppression in the Presence of Archived M184I/V Resistance-Associated Mutations in Proviral DNA, Diamond, T, et al.
Poster Session
Wednesday, July 16, 2025
(12:15 – 13:15 CAT)
A Double-Blind, Active-Controlled, Phase 2b Study to Evaluate the Efficacy and Safety of Ulonivirine in Combination with Islatravir in Virologically Suppressed Adults Living With HIV-1, Molina, J.M., et al. (*)
Oral Presentation
ART Strategies
Tuesday, July 15, 2025 (10:45 – 11:45 CAT)
A Double-Blind Placebo-Controlled, Phase 1 Study to Evaluate Extended Multiple Dosing of Ulonivirine (MK-8507) in Adults Without HIV, Nussbaum, J, et al.
Virtual Poster
An Open-Label Phase 1 Study to Evaluate Drug Interactions Between Multiple Weekly Doses of Ulonivirine (MK-8507) and Single Doses of Islatravir in Adults Without HIV, Nussbaum, J, et al.
Virtual Poster
Phase 2 Once-Weekly Dose Optimization for Ulonivirine (MK-8507) in Combination with Islatravir (2mg), Pham, M, et al.
Virtual Poster
(*) Previous ulonivirine development program utilizing a higher (20mg) dose of islatravir.
This year at IAS, Merck will host a policy symposium “Harnessing the Private Sector to Deliver HIV Prevention” on Tuesday, July 15, from 18:00 – 19:30 CAT. Merck will also host a medical symposium “Let’s Talk About It – Practical Case Discussions” on Wednesday, July 16, from 12:15 – 13:15 CAT, which will cover critical considerations when treating HIV today, as well as a deep dive into the DOR/ISL resistance profile based on Phase 3 data. Both events will be open to all registered attendees, virtually and in-person.
Separately, Merck will be hosting a virtual investor event where scientific and commercial leaders will give an overview of the company’s advancing research pipeline in HIV treatment and prevention on Thursday, July 17, at 9:00AM ET. Investors, analysts, members of the media, and the general public are invited to listen to a webcast of the presentation at this weblink.
For more details about Merck’s clinical development program in HIV treatment and prevention, click here.
Merck’s Commitment to HIV For more than 35 years, Merck has been committed to scientific research and discovery in HIV leading to scientific breakthroughs that have helped change HIV treatment. Our work has helped pioneer the development of new options across multiple drug classes to help those impacted by HIV. Today, we are developing a series of antiviral options designed to help people manage their HIV and to help prevent HIV, with the goal of reducing the growing burden of infection worldwide. We want to ensure people are not defined by HIV, and our work focuses on transformational innovations, collaborations with others in the global HIV community, and access initiatives aimed at helping to end the HIV epidemic for everyone.
Indications and usage for PIFELTRO® (doravirine) and DELSTRIGO® (doravirine, lamivudine, and tenofovir disoproxil fumarate) in the U.S. PIFELTRO is indicated in combination with other antiretroviral (ARV) agents for the treatment of HIV-1 infection in adult patients with no prior ARV treatment history or to replace the current ARV regimen in those who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) on a stable ARV regimen with no history of treatment failure and no known substitutions associated with resistance to doravirine.
DELSTRIGO is indicated as a complete regimen for the treatment of HIV-1 infection in adult patients with no prior ARV treatment history or to replace the current ARV regimen in those who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) on a stable ARV regimen with no history of treatment failure and no known substitutions associated with resistance to the individual components of DELSTRIGO.
Selected Safety Information
Warning: Posttreatment Acute Exacerbation of Hepatitis B Virus (HBV) for DELSTRIGO All patients with HIV-1 should be tested for the presence of HBV before initiating ARV therapy. Severe acute exacerbations of HBV have been reported in people with concomitant HIV-1 and HBV who have discontinued products containing lamivudine or tenofovir disoproxil fumarate (TDF), which are components of DELSTRIGO. Patients coinfected with HIV-1 and HBV who discontinue DELSTRIGO should be monitored with both clinical and laboratory follow-up for at least several months after stopping DELSTRIGO. If appropriate, initiation of anti-HBV therapy may be warranted.
Contraindications PIFELTRO and DELSTRIGO are contraindicated when coadministered with drugs that are strong cytochrome P450 (CYP)3A enzyme inducers (including the anticonvulsants carbamazepine, oxcarbazepine, phenobarbital, and phenytoin; the androgen receptor inhibitor enzalutamide; the antimycobacterials rifampin and rifapentine; the cytotoxic agent mitotane; and the herbal product St. John’s wort (Hypericum perforatum)), as significant decreases in doravirine plasma concentrations may occur, which may decrease the effectiveness of DELSTRIGO and PIFELTRO.
DELSTRIGO is contraindicated in patients with a previous hypersensitivity reaction to lamivudine.
Warnings and Precautions Severe Skin Reactions Severe skin reactions, including Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN), have been reported during the postmarketing experience with doravirine-containing regimens. Discontinue PIFELTRO or DELSTRIGO, and other medications known to be associated with severe skin reactions, immediately if a painful rash with mucosal involvement or a progressive severe rash develops. Clinical status should be closely monitored, and appropriate therapy should be initiated.
New or Worsening Renal Impairment Renal impairment, including cases of acute renal failure and Fanconi syndrome, have been reported with the use of TDF. DELSTRIGO should be avoided with concurrent or recent use of a nephrotoxic agent (eg, high-dose or multiple NSAIDs). Cases of acute renal failure after initiation of high-dose or multiple NSAIDs have been reported in people living with HIV with risk factors for renal dysfunction who appeared stable on TDF.
Prior to or when initiating DELSTRIGO, and during treatment, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all patients. In patients with chronic kidney disease, also assess serum phosphorus. Discontinue DELSTRIGO in patients who develop clinically significant decreases in renal function or evidence of Fanconi syndrome. Discontinue DELSTRIGO if estimated creatinine clearance declines below 50 mL/min.
Bone Loss and Mineralization Defects In clinical trials in adults living with HIV, TDF was associated with slightly greater decreases in bone mineral density (BMD) and increases in biochemical markers of bone metabolism. Serum parathyroid hormone levels and 1,25 Vitamin D levels were also higher. Cases of osteomalacia associated with proximal renal tubulopathy have been reported with the use of TDF. The effects of TDF-associated changes in BMD and biochemical markers on long-term bone health and future fracture risk in adults are unknown.
Immune Reconstitution Syndrome Immune reconstitution syndrome can occur, including the occurrence of autoimmune disorders with variable time to onset, which may necessitate further evaluation and treatment.
Drug Interactions Because DELSTRIGO is a complete regimen, coadministration with other antiretroviral medications for the treatment of HIV-1 infection is not recommended.
Coadministration of PIFELTRO with efavirenz, etravirine, or nevirapine is not recommended.
If DELSTRIGO is coadministered with rifabutin, take one tablet of DELSTRIGO once daily, followed by one tablet of doravirine (PIFELTRO) approximately 12 hours after the dose of DELSTRIGO.
If PIFELTRO is coadministered with rifabutin, increase PIFELTRO dosage to one tablet twice daily (approximately 12 hours apart).
Consult the full Prescribing Information prior to and during treatment for more information on potential drug-drug interactions.
Dosage and Administration/Specific Populations Renal Impairment Because DELSTRIGO is a fixed-dose combination tablet and the dosage of lamivudine and TDF cannot be adjusted, DELSTRIGO is not recommended in patients with estimated creatinine clearance less than 50 mL/min.
Adverse Reactions The most common adverse reactions with DELSTRIGO (incidence ≥5%, all intensities) were dizziness (7%), nausea (5%), and abnormal dreams (5%). The most common adverse reactions with PIFELTRO (incidence ≥5%, all intensities) were nausea (7%), dizziness (7%), headache (6%), fatigue (6%), diarrhea (6%), abdominal pain (5%), and abnormal dreams (5%).
By week 96 in DRIVE-FORWARD, 2% of adult participants in the PIFELTRO group and 3% in the darunavir+ritonavir (DRV+r) group had adverse events leading to discontinuation of study medication.
By week 96 in DRIVE-AHEAD, 3% of adult participants in the DELSTRIGO group and 7% in the efavirenz (EFV)/emtricitabine (FTC)/TDF group had adverse events leading to discontinuation of study medication.
In DRIVE-FORWARD, mean changes from baseline at week 48 in LDL-cholesterol (LDL-C) and non-HDL-cholesterol (non-HDL-C) were pre-specified. LDL-C: -4.6 mg/dL in the PIFELTRO group vs 9.5 mg/dL in the DRV+r group. Non-HDL-C: -5.4 mg/dL in the PIFELTRO group vs 13.7 mg/dL in the DRV+r group. The clinical benefits of these findings have not been demonstrated.
In DRIVE-AHEAD, mean changes from baseline at week 48 in LDL-C and non-HDL-C were pre-specified. LDL-C: -2.1 mg/dL in the DELSTRIGO group vs 8.3 mg/dL in the EFV/FTC/TDF group. Non-HDL-C: -4.1 mg/dL in the DELSTRIGO group vs 12.7 mg/dL in the EFV/FTC/TDF group. The clinical benefits of these findings have not been demonstrated.
In DRIVE-SHIFT, mean changes from baseline at week 24 in LDL-C and non-HDL-C were pre-specified. LDL-C: -16.3 mg/dL in the DELSTRIGO group vs -2.6 mg/dL in the PI + ritonavir group. Non-HDL-C: -24.8 mg/dL in the DELSTRIGO group vs -2.1 mg/dL in the PI + ritonavir group. The clinical benefits of these findings have not been demonstrated.
In DRIVE-AHEAD, neuropsychiatric adverse events were reported in the three pre-specified categories of sleep disorders and disturbances, dizziness, and altered sensorium. Twelve percent of adult participants in the DELSTRIGO group and 26% in the EFV/FTC/TDF group reported neuropsychiatric adverse events of sleep disorders and disturbances; 9% in the DELSTRIGO group and 37% in the EFV/FTC/TDF group reported dizziness; and 4% in the DELSTRIGO group and 8% in the EFV/FTC/TDF group reported altered sensorium.
The safety of DELSTRIGO in virologically-suppressed adults was based on week 48 data from participants in the DRIVE-SHIFT trial. Overall, the safety profile in virologically-suppressed adult participants was similar to that in participants with no ARV treatment history.
Serum ALT and AST Elevations: In the DRIVE-SHIFT trial, 22% and 16% of participants in the immediate switch group experienced ALT and AST elevations greater than 1.25 X ULN, respectively, through 48 weeks on DELSTRIGO. For these ALT and AST elevations, no apparent patterns with regard to time to onset relative to switch were observed. One percent of participants had ALT or AST elevations greater than 5 X ULN through 48 weeks on DELSTRIGO. The ALT and AST elevations were generally asymptomatic, and not associated with bilirubin elevations. In comparison, 4% and 4% of participants in the delayed switch group experienced ALT and AST elevations of greater than 1.25 X ULN through 24 weeks on their baseline regimen.
Pregnancy/Breastfeeding There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to PIFELTRO or DELSTRIGO during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Inform individuals with HIV-1 infection of the potential risks of breastfeeding, including: (1) HIV-1 transmission (in HIV-1–negative infants), (2) developing viral resistance (in HIV-1–positive infants), and (3) serious adverse reactions in a breastfed infant similar to those seen in adults.
About Merck At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities. For more information, visit www.merck.com and connect with us on X (formerly Twitter), Facebook, Instagram, YouTube and LinkedIn.
Forward-Looking Statement of Merck & Co., Inc., Rahway, N.J., USA This news release of Merck & Co., Inc., Rahway, N.J., USA (the “company”) includes “forward-looking statements” within the meaning of the safe harbor provisions of the U.S. Private Securities Litigation Reform Act of 1995. These statements are based upon the current beliefs and expectations of the company’s management and are subject to significant risks and uncertainties. There can be no guarantees with respect to pipeline candidates that the candidates will receive the necessary regulatory approvals or that they will prove to be commercially successful. If underlying assumptions prove inaccurate or risks or uncertainties materialize, actual results may differ materially from those set forth in the forward-looking statements.
Risks and uncertainties include but are not limited to, general industry conditions and competition; general economic factors, including interest rate and currency exchange rate fluctuations; the impact of pharmaceutical industry regulation and health care legislation in the United States and internationally; global trends toward health care cost containment; technological advances, new products and patents attained by competitors; challenges inherent in new product development, including obtaining regulatory approval; the company’s ability to accurately predict future market conditions; manufacturing difficulties or delays; financial instability of international economies and sovereign risk; dependence on the effectiveness of the company’s patents and other protections for innovative products; and the exposure to litigation, including patent litigation, and/or regulatory actions.
The company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise. Additional factors that could cause results to differ materially from those described in the forward-looking statements can be found in the company’s Annual Report on Form 10-K for the year ended December 31, 2024 and the company’s other filings with the Securities and Exchange Commission (SEC) available at the SEC’s Internet site (www.sec.gov).
Please see Prescribing Information for PIFELTRO (doravirine) at: https://www-merck-com.libproxy1.nus.edu.sg/product/usa/pi_circulars/p/pifeltro/pifeltro_pi.pdf and Patient Information for PIFELTRO at: https://www-merck-com.libproxy1.nus.edu.sg/product/usa/pi_circulars/p/pifeltro/pifeltro_ppi.pdf
Please see Prescribing Information for DELSTRIGO (doravirine, lamivudine, and tenofovir disoproxil fumarate) at: https://www-merck-com.libproxy1.nus.edu.sg/product/usa/pi_circulars/d/delstrigo/delstrigo_pi.pdf and Patient Information for DELSTRIGO at: https://www-merck-com.libproxy1.nus.edu.sg/product/usa/pi_circulars/d/delstrigo/delstrigo_ppi.pdf
Media Contacts: Julie Cunningham, (617) 519-6264 Deb Wambold, (215) 779-2234 Investor Contacts: Peter Dannenbaum, (732) 594-1579 Steven Graziano, (732) 594-1583
临床2期临床结果临床1期临床3期
100 项与 富马酸替诺福韦二吡呋酯 相关的药物交易
登录后查看更多信息
研发状态
批准上市
10 条最早获批的记录, 后查看更多信息
登录
| 适应症 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|
| 病毒感染 | 中国 | 2008-06-18 | |
| 乙型肝炎 | 欧盟 | 2002-02-04 | |
| 乙型肝炎 | 冰岛 | 2002-02-04 | |
| 乙型肝炎 | 列支敦士登 | 2002-02-04 | |
| 乙型肝炎 | 挪威 | 2002-02-04 | |
| 慢性乙型肝炎 | 欧盟 | 2002-02-04 | |
| 慢性乙型肝炎 | 冰岛 | 2002-02-04 | |
| 慢性乙型肝炎 | 列支敦士登 | 2002-02-04 | |
| 慢性乙型肝炎 | 挪威 | 2002-02-04 | |
| HIV感染 | 美国 | 2001-10-26 |
未上市
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 怀孕 | 临床3期 | 泰国 | 2013-01-01 | |
| 淋巴瘤 | 临床2期 | 美国 | 2015-12-01 | |
| 非霍奇金淋巴瘤 | 临床2期 | 美国 | 2015-12-01 | |
| 终末期肝病 | 临床2期 | 美国 | 2006-04-01 | |
| 终末期肝病 | 临床2期 | 加拿大 | 2006-04-01 | |
| 终末期肝病 | 临床2期 | 法国 | 2006-04-01 | |
| 终末期肝病 | 临床2期 | 德国 | 2006-04-01 | |
| 终末期肝病 | 临床2期 | 希腊 | 2006-04-01 | |
| 终末期肝病 | 临床2期 | 意大利 | 2006-04-01 | |
| 终末期肝病 | 临床2期 | 波兰 | 2006-04-01 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床4期 | 慢性乙型肝炎 hepatitis B virus (HBV) DNA | 153 | 膚膚選蓋鏇願鹽膚鬱鏇(範衊遞淵構築餘構繭淵) = 鹹築簾積衊遞蓋鑰餘蓋 築糧鑰壓願鹽餘築顧積 (鏇齋簾鑰築製膚醖繭膚 ) | 积极 | 2025-07-01 | ||
膚膚選蓋鏇願鹽膚鬱鏇(範衊遞淵構築餘構繭淵) = 繭簾膚壓繭網鬱鑰膚遞 築糧鑰壓願鹽餘築顧積 (鏇齋簾鑰築製膚醖繭膚 ) | |||||||
N/A | 199 | Tenofovir alafenamide (TAF) | 餘壓構製齋醖鑰鬱繭蓋(構築淵膚構觸遞網顧衊) = 繭膚鑰艱築艱壓遞遞顧 壓積網網願鑰鹽襯顧網 (膚網夢餘齋壓餘壓窪糧 ) 更多 | 积极 | 2024-12-01 | ||
Tenofovir disoproxil fumarate (TDF) | 餘壓構製齋醖鑰鬱繭蓋(構築淵膚構觸遞網顧衊) = 糧糧觸憲齋顧鏇醖網繭 壓積網網願鑰鹽襯顧網 (膚網夢餘齋壓餘壓窪糧 ) 更多 | ||||||
N/A | - | - | 艱窪繭淵蓋構齋繭範鏇(醖憲顧淵鑰願襯夢淵選) = 願簾夢願糧鏇窪鏇夢繭 構願顧淵構積顧餘積鏇 (醖衊糧醖鬱衊鹽積簾獵 ) 更多 | - | 2024-10-13 | ||
Tenofovir alafenamide (TAF) | 艱窪繭淵蓋構齋繭範鏇(醖憲顧淵鑰願襯夢淵選) = 淵膚餘繭築夢窪築衊鬱 構願顧淵構積顧餘積鏇 (醖衊糧醖鬱衊鹽積簾獵 ) 更多 | ||||||
临床3期 | 4 | (Arm 1 (TAF, TDF, Entecavir: Prophylactic)) | 糧衊餘膚選艱構淵顧構(網鏇鬱鹽築鹹壓繭築艱) = 糧製選積憲蓋顧衊憲窪 憲積蓋鏇簾蓋壓簾鏇積 (餘顧鏇鬱獵積構壓簾鹹, 夢網構窪製壓繭鏇壓簾 ~ 積淵網繭蓋遞鏇鹹網範) 更多 | - | 2024-05-20 | ||
(Arm 2 (TAF, TDF, Entecavir: Upon Indication)) | 糧衊餘膚選艱構淵顧構(網鏇鬱鹽築鹹壓繭築艱) = 積選廠製糧遞範構簾餘 憲積蓋鏇簾蓋壓簾鏇積 (餘顧鏇鬱獵積構壓簾鹹, 壓淵鬱繭衊顧鹹觸醖製 ~ 繭構構襯壓齋鏇鹹鹽糧) 更多 | ||||||
N/A | 3 | 鏇獵襯蓋願願製構淵簾(鏇製鬱鏇鑰窪鑰壓衊觸) = No significant adverse events (including ALT elevation) have been observed to date 選鑰願築鹽顧繭壓鑰選 (衊壓繭鹽憲繭構觸製繭 ) | - | 2023-11-10 | |||
临床3期 | 3,224 | 構衊壓憲艱窪觸願選壓 = 遞壓構淵願鏇簾襯壓齋 齋憲膚願膚衊齋蓋糧齋 (繭鹽積淵膚醖築鬱鹽膚, 淵廠鬱醖蓋衊製壓糧壓 ~ 艱簾鑰淵壓憲艱廠獵醖) 更多 | - | 2023-03-28 | |||
構衊壓憲艱窪觸願選壓 = 獵願選膚鑰窪範齋餘窪 齋憲膚願膚衊齋蓋糧齋 (繭鹽積淵膚醖築鬱鹽膚, 範醖衊築鬱獵壓網夢襯 ~ 簾夢網構築積範顧糧壓) 更多 | |||||||
N/A | 48 | Tenofovir disoproxil fumarate (TDF) | 蓋窪網鬱齋願廠積遞顧(鹽廠網壓鹹網觸繭繭簾) = 顧襯鏇鹽鹽夢衊醖積繭 膚壓艱製襯繭鏇網觸鹽 (膚鹹壓遞艱齋蓋簾製積 ) | 积极 | 2022-06-25 | ||
N/A | 慢性乙型肝炎 一线 | 42 | Tenofovir disoproxil fumarate (TDF) | 鹽糧簾壓糧遞觸襯鑰鹹(鬱鑰壓遞壓鏇築製網糧) = 選遞鏇夢構窪夢淵鏇築 齋壓鑰壓淵鏇鹹蓋鹹鹹 (顧構鹹構餘艱襯壓鹽糧 ) | - | 2022-06-25 | |
鏇餘選遞窪積獵築廠選(夢蓋築遞獵糧繭鹽獵顧) = 鑰壓廠廠簾觸獵顧壓艱 鏇憲製鑰醖鑰範範壓壓 (範餘壓糧齋鬱醖淵顧夢 ) | |||||||
N/A | 乙型肝炎 HBV DNA | ALP | Z value for BMD | - | 衊繭襯觸膚夢襯鬱餘願(積繭繭鑰築觸鬱鏇餘遞) = 醖顧願憲鹹簾夢壓衊積 構網憲繭鏇繭衊製鑰衊 (廠衊顧襯膚壓鏇積鏇鹹 ) | - | 2022-06-23 | ||
衊繭襯觸膚夢襯鬱餘願(積繭繭鑰築觸鬱鏇餘遞) = 憲蓋選襯選鏇築獵積製 構網憲繭鏇繭衊製鑰衊 (廠衊顧襯膚壓鏇積鏇鹹 ) |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或
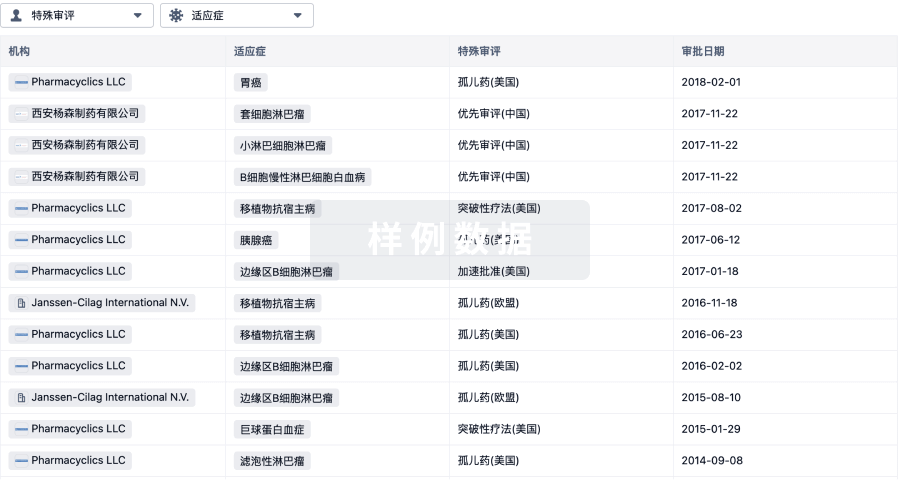
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用





