预约演示
更新于:2024-09-22
GPRC5D
更新于:2024-09-22
基本信息
相关靶点 |
关联
41
项与 GPRC5D 相关的药物作用机制 CD3刺激剂 [+3] |
在研机构 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2023-08-09 |
作用机制 BCMA调节剂 [+3] |
在研机构 |
原研机构 |
非在研适应症- |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期- |
靶点 |
作用机制 GPRC5D抑制剂 |
非在研适应症- |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期- |
70
项与 GPRC5D 相关的临床试验NCT06572605
A Phase 1b/2 Study of Talquetamab Plus Concomitant Priming Radiotherapy in Multiple Myeloma With Extramedullary Disease
This phase I/II trial tests the safety and effectiveness of extramedullary disease (EMD)-directed external beam radiation therapy (EBRT) in combination with talquetamab for the treatment of multiple myeloma patients with extramedullary disease. Extramedullary disease in multiple myeloma involves the infiltration of organs and soft tissues by malignant plasma cells and has proven difficult to treat. Radiation therapy uses high energy x-rays, particles, or radioactive seeds to kill cancer cells and shrink cancers. EBRT is a type of radiation therapy that delivers high-energy beams to the cancer from outside of the body. In this trial, the EBRT will be directed to a site of extramedullary disease. Talquetamab is a monoclonal antibody that may interfere with the ability of cancer cells to grow and spread. A monoclonal antibody is a type of protein that can bind to certain targets in the body, such as molecules that cause the body to make an immune response (antigens). Combining EMD-directed EBRT with talquetamab may be safe, tolerable, and/or effective in treating multiple myeloma patients with extramedullary disease.
开始日期2024-12-15 |
申办/合作机构  Hope Medical Center Hope Medical Center [+1] |
NCT06461988
An Open-Label, Non-Randomized, Multicenter, Phase II Study to Study the Efficacy of Talquetamab (JNJ-64407564) and Lenalidomide as Post Stem Cell Transplant Maintenance in Multiple Myeloma (OPTIMMAL)
Multiple myeloma (MM) is a heterogenous plasma cell malignancy characterized by clonal proliferation of plasma cells and organ damage. Autologous transplantation with high dose chemotherapy is the standard of care in frontline treatment of eligible patients with MM.
开始日期2024-11-01 |
申办/合作机构  Stanford University Stanford University [+1] |
NCT06550895
A Phase 2, Open-Label, Multicenter Study of Ciltacabtagene Autoleucel and Talquetamab for the Treatment of Participants With High-Risk Multiple Myeloma
The purpose of this study is to define the safety of Ciltacabtagene Autoleucel (Cilta-cel) and Talquetamab in participants with high-risk multiple myeloma (MM).
开始日期2024-09-23 |
100 项与 GPRC5D 相关的临床结果
登录后查看更多信息
100 项与 GPRC5D 相关的转化医学
登录后查看更多信息
0 项与 GPRC5D 相关的专利(医药)
登录后查看更多信息
125
项与 GPRC5D 相关的文献(医药)2024-10-01·Seminars in Oncology Nursing
Nursing Considerations for the Clinical Management of Adverse Events Associated with Talquetamab in Patients with Relapsed or Refractory Multiple Myeloma
Review
作者: Catamero, Donna ; Leahey, Sheryl ; Rogers, Stephanie ; Hannemann, Linda ; Gray, Kathleen ; Ray, Chloe ; O'Rourke, Lisa ; Shenoy, Samantha ; Hefner, Kayla ; Esler, Elaine ; Tolbert, Jaszianne ; Patel, Saurabh ; Purcell, Kiah ; Renaud, Thomas
OBJECTIVES:
Talquetamab is a newly approved bispecific antibody targeting the CD3 receptor on T cells and a receptor, G protein-coupled receptor family C group 5 member D (GPRC5D), highly expressed on multiple myeloma (MM) cells. In addition to immune therapy-related adverse events (AEs) associated with bispecific antibody therapies, talquetamab is associated with unique skin/nail and oral GPRC5D-related side effects that require additional supportive care. This review provides clinical management strategies for talquetamab based on oncology nurses' experience during the MonumenTAL-1 (NCT03399799/NCT04634552) clinical trial. The objective of this review is to raise awareness among nurses and patients to better understand and manage the side effects associated with talquetamab treatment in order to optimize patient outcomes.
DATA SOURCES:
MonumenTAL-1 is a phase 1/2 clinical trial of talquetamab in patients with relapsed/refractory MM who are triple-class exposed. Details on overall response, safety, and AE incidence and occurrence were previously published. Management strategies for the T-cell-related and unique GPRC5D-related AEs were collected from oncology nurses from different study sites.
CONCLUSION:
Talquetamab has shown overall response rates of >71% in patients with relapsed/refractory MM in the MonumenTAL-1 study. AEs were low grade and predictable; few led to study discontinuation.
IMPLICATIONS FOR NURSING PRACTICE:
Oncology nurses have specialized knowledge of treatment administration monitoring based on their participation in the MonumenTAL-1 trial. This review provides information for nurses in both the academic and community settings on how to monitor, counsel, and support patients, which will in turn improve patients' quality of life and overall survival.
2024-10-01·JOURNAL OF MOLECULAR BIOLOGY
Structural Basis for the Recognition of GPRC5D by Talquetamab, a Bispecific Antibody for Multiple Myeloma
Article
作者: Park, Junhyeon ; Shin, Jinwoo ; Young Mo, Geun ; Jeong, Jihong ; Cho, Yunje
Multiple myeloma (MM) is a complex hematological malignancy characterized by abnormal antibody production from plasma cells. Despite advances in the treatment, many patients experience disease relapse or become refractory to treatment. G-protein-coupled receptor class C group 5 member D (GPRC5D), an orphan GPCR predominantly expressed in MM cells, is emerging as a promising target for MM immunotherapy. Talquetamab, a Food and Drug Administration-approved T-cell-directing bispecific antibody developed for treatment of MM, targets GPRC5D. Here, we elucidate the structure of GPRC5D complexed with the Fab fragment of talquetamab, using cryo-electron microscopy, providing the basis for recognition of GPRC5D by the bispecific antibody. GPRC5D forms a symmetric homodimer with the interface between transmembrane helix (TM) 4 of one protomer and TM4/5 of the other protomer. A single talquetamab Fab interacts with the GPRC5D dimer with its orientation toward the dimer interface. All six complementarity-determining regions of talquetamab engage with extracellular loops and TM3/5/7. In particular, the side-chain of an arginine residue from the antibody penetrates into a shallow pocket on the extracellular surface of GPRC5D. The structure offers insights for optimizing antibody design against GPRC5D for relapsed or refractory MM therapy.
2024-09-01·Lancet Haematology
Mechanisms of resistance against T-cell engaging bispecific antibodies in multiple myeloma: implications for novel treatment strategies
Review
作者: Mateos, Maria Victoria ; van de Donk, Niels W C J ; Chari, Ajai
Off-the-shelf T-cell-redirecting bispecific antibodies targeting BCMA, GPRC5D, and FcRH5 have high activity in multiple myeloma with a manageable toxicity profile. However, not all patients respond to bispecific antibodies and patients can develop bispecific antibody resistance after an initial response. Mechanisms that contribute to bispecific antibody resistance are multifactorial and include tumour-related factors, such as high tumour burden, expression of T-cell inhibitory ligands, and antigen loss. Resistance due to antigen escape can be prevented by simultaneously targeting two tumour-associated antigens with a trispecific antibody or a combination of two bispecific antibodies. There is also increasing evidence that primary resistance to bispecific antibodies is associated with impaired baseline T-cell function. Long-term exposure to bispecific antibodies with chronic T-cell stimulation further aggravates T-cell dysfunction, which could contribute to failure of disease control. Therapeutic interference with T-cell exhaustion by targeting inhibitory or costimulatory pathways could improve bispecific antibody-mediated antitumour activity. The immunosuppressive microenvironment also contributes to bispecific antibody resistance. CD38-targeting antibodies hold promise as combination partners for bispecific antibodies because of their potential to eliminate CD38+ immune suppressor cells. In conclusion, a better understanding of the mechanisms underlying the absence of disease response has provided novel insights to optimise T-cell activity and bispecific antibody efficacy in multiple myeloma.
1,646
项与 GPRC5D 相关的新闻(医药)2024-09-22
·医药笔记
▎Armstrong
2024年9月20日,礼来在Clinicaltrials.gov注册了LY3537031治疗减重的一期临床试验。
根据礼来此前公开的信息,LY3537031为一款GLP-1R/GIPR双靶点激动剂,此前开展过降糖一期临床试验。此次开展的一期临床针对减重适应症,计划入组230例受试者,预计2025年7月完成。
LY3537031仍采用皮下注射给药,礼来先后有3款GLP-1/GIP双靶点激动剂进入临床阶段,除替尔泊肽外其他2款LY3537031、LY3493269(皮下或口服剂型)均开发二型糖尿病适应症,但未披露过临床数据,一期临床完成后未见继续开发。此次LY3537031重启临床,并转向减重适应症,意味着第2款GLP-1/GIP重新进入活跃开发阶段。
总结
最近一段时间,可以看到礼来全面推进多款双靶点/多靶点GLP-1类管线的临床开发,除了已经上市的替尔泊肽(高剂量版本在推进重度肥胖BMI≥35人群的二期临床),三靶点的Retatrutide,玛仕度肽也进入二期临床,并在前几天披露了16mg减重一期临床的数据,第2款GLP-1/GIP也重新启动治疗减重的临床试验。
Armstrong技术全梳理系列
GPRC5D靶点全梳理;
CD40靶点全梳理;
CD47靶点全梳理;
补体靶向药物技术全梳理;
补体药物:眼科治疗的重要方向;
Claudin 6靶点全梳理;
Claudin 18.2靶点全梳理;
靶点冷暖,行业自知;
中国大分子新药研发格局;
被炮轰的“me too”;
佐剂百年史;
胰岛素百年传奇;
CUSBEA:风雨四十载;
中国新药研发的焦虑;
中国生物医药企业的研发竞争;
中国双抗竞争格局;
中国ADC竞争格局;
中国双抗技术全梳理;
中国ADC技术全梳理;
Ambrx技术全梳理;
Vir Biotech技术全梳理;
Immune-Onc技术全梳理;
亘喜生物技术全梳理;
康哲药业技术全梳理;
科济药业技术全梳理;
恺佧生物技术全梳理;
同宜医药技术全梳理;
百奥赛图技术全梳理;
腾盛博药技术全梳理;
创胜集团技术全梳理;
永泰生物技术全梳理;
中国抗体技术全梳理;
德琪医药技术全梳理;
德琪医药技术全梳理2.0;
和铂医药技术全梳理;
荣昌生物技术全梳理;
再鼎医药技术全梳理;
药明生物技术全梳理;
恒瑞医药技术全梳理;
豪森药业技术全梳理;
正大天晴技术全梳理;
吉凯基因技术全梳理;
基石药业技术全梳理;
百济神州技术全梳理;
百济神州技术全梳理第2版;
信达生物技术全梳理;
信达生物技术全梳理第2版;
中山康方技术全梳理;
复宏汉霖技术全梳理;
先声药业技术全梳理;
君实生物技术全梳理;
嘉和生物技术全梳理;
志道生物技术全梳理;
道尔生物技术全梳理;
尚健生物技术全梳理;
康宁杰瑞技术全梳理;
科望医药技术全梳理;
科望医药技术全梳理2.0;
岸迈生物技术全梳理;
礼进生物技术全梳理;
康桥资本技术全梳理;
余国良的抗体药布局;
荃信生物技术全梳理;
安源医药技术全梳理;
三生国健技术全梳理;
仁会生物技术全梳理;
乐普生物技术全梳理;
同润生物技术全梳理;
宜明昂科技术全梳理;
派格生物技术全梳理;
迈威生物技术全梳理;
Momenta技术全梳理;
NGM技术全梳理;
普米斯生物技术全梳理;
普米斯生物技术全梳理2.0;
三叶草生物技术全梳理;
贝达药业抗体药全梳理;
泽璟制药抗体药全梳理;
恒瑞医药抗体药全梳理;
齐鲁制药抗体药全梳理;
石药集团抗体药全梳理;
豪森药业抗体药全梳理;
华海药业抗体药全梳理;
科伦药业抗体药全梳理;
百奥泰技术全梳理;
凡恩世技术全梳理。
临床2期临床1期抗体药物偶联物
2024-09-20
·医药笔记
▎Armstrong
2024年9月20日,诺和诺德公布CB1R激动剂Monlunabant治疗减重的二期临床最新数据,经过16周治疗,Monlunabant组减重7.1公斤(6.5%),安慰剂组减重0.7公斤(0.6%),经过安慰剂校正减重6.0%。
当天诺和诺德股价大跌5.5%,市值缩水300亿美元。
Monlunabant为诺和诺德11亿美元收购Inversago获得,为诺和诺德重要的减重储备管线之一。
Monlunabant在临床前表现出优异的减重效果。
今年3月披露的数据显示Monlunabant治疗4周减重3.5公斤(3.2%),安慰剂校正后减重3.8%。
总结
Monlunabant的数据明显未达到此前预期,连累首发管线同样为CB1R激动剂的Corbus Pharmaceuticals股价大跌62%,市值仅剩下2.3亿美元。海量资源投入下,减重新靶点纷纷涌现,但探索之路仍旧漫长。
Armstrong技术全梳理系列
GPRC5D靶点全梳理;
CD40靶点全梳理;
CD47靶点全梳理;
补体靶向药物技术全梳理;
补体药物:眼科治疗的重要方向;
Claudin 6靶点全梳理;
Claudin 18.2靶点全梳理;
靶点冷暖,行业自知;
中国大分子新药研发格局;
被炮轰的“me too”;
佐剂百年史;
胰岛素百年传奇;
CUSBEA:风雨四十载;
中国新药研发的焦虑;
中国生物医药企业的研发竞争;
中国双抗竞争格局;
中国ADC竞争格局;
中国双抗技术全梳理;
中国ADC技术全梳理;
Ambrx技术全梳理;
Vir Biotech技术全梳理;
Immune-Onc技术全梳理;
亘喜生物技术全梳理;
康哲药业技术全梳理;
科济药业技术全梳理;
恺佧生物技术全梳理;
同宜医药技术全梳理;
百奥赛图技术全梳理;
腾盛博药技术全梳理;
创胜集团技术全梳理;
永泰生物技术全梳理;
中国抗体技术全梳理;
德琪医药技术全梳理;
德琪医药技术全梳理2.0;
和铂医药技术全梳理;
荣昌生物技术全梳理;
再鼎医药技术全梳理;
药明生物技术全梳理;
恒瑞医药技术全梳理;
豪森药业技术全梳理;
正大天晴技术全梳理;
吉凯基因技术全梳理;
基石药业技术全梳理;
百济神州技术全梳理;
百济神州技术全梳理第2版;
信达生物技术全梳理;
信达生物技术全梳理第2版;
中山康方技术全梳理;
复宏汉霖技术全梳理;
先声药业技术全梳理;
君实生物技术全梳理;
嘉和生物技术全梳理;
志道生物技术全梳理;
道尔生物技术全梳理;
尚健生物技术全梳理;
康宁杰瑞技术全梳理;
科望医药技术全梳理;
科望医药技术全梳理2.0;
岸迈生物技术全梳理;
礼进生物技术全梳理;
康桥资本技术全梳理;
余国良的抗体药布局;
荃信生物技术全梳理;
安源医药技术全梳理;
三生国健技术全梳理;
仁会生物技术全梳理;
乐普生物技术全梳理;
同润生物技术全梳理;
宜明昂科技术全梳理;
派格生物技术全梳理;
迈威生物技术全梳理;
Momenta技术全梳理;
NGM技术全梳理;
普米斯生物技术全梳理;
普米斯生物技术全梳理2.0;
三叶草生物技术全梳理;
贝达药业抗体药全梳理;
泽璟制药抗体药全梳理;
恒瑞医药抗体药全梳理;
齐鲁制药抗体药全梳理;
石药集团抗体药全梳理;
豪森药业抗体药全梳理;
华海药业抗体药全梳理;
科伦药业抗体药全梳理;
百奥泰技术全梳理;
凡恩世技术全梳理。
临床2期并购抗体药物偶联物
2024-09-20
·医药笔记
▎Armstrong
2024年9月19日,百济神州在Clinicaltrials.gov网站上注册了BG-C477、BG-T187的一期临床试验。
BG-T187为一款EGFR/cMET三抗,一期临床计划入组87例患者。
BG-C477为一款CEA ADC。
BG-C477采用新型拓扑异构酶抑制剂作为payload,DAR值为8,临床前研究中表现出优于SAR701的抗肿瘤活性。
EGFR/cMET三抗目标适应症主要为肺癌,CEA ADC目标适应症为肺癌、胃肠道肿瘤。
总结
近两年来,百济神州新药物形式推进迅速,多款PROTAC、双抗、ADC新药进入临床阶段,PROTAC覆盖BTK、EGFR、IRAK4靶点,双抗覆盖Claudin6/CD3、MUC1/CD16、CEA/4-1BB、GPC3/4-1BB,ADC覆盖B7H3、B7H4、CEA、FGFR2b,前药细胞因子的IL-15等。
Armstrong技术全梳理系列
GPRC5D靶点全梳理;
CD40靶点全梳理;
CD47靶点全梳理;
补体靶向药物技术全梳理;
补体药物:眼科治疗的重要方向;
Claudin 6靶点全梳理;
Claudin 18.2靶点全梳理;
靶点冷暖,行业自知;
中国大分子新药研发格局;
被炮轰的“me too”;
佐剂百年史;
胰岛素百年传奇;
CUSBEA:风雨四十载;
中国新药研发的焦虑;
中国生物医药企业的研发竞争;
中国双抗竞争格局;
中国ADC竞争格局;
中国双抗技术全梳理;
中国ADC技术全梳理;
Ambrx技术全梳理;
Vir Biotech技术全梳理;
Immune-Onc技术全梳理;
亘喜生物技术全梳理;
康哲药业技术全梳理;
科济药业技术全梳理;
恺佧生物技术全梳理;
同宜医药技术全梳理;
百奥赛图技术全梳理;
腾盛博药技术全梳理;
创胜集团技术全梳理;
永泰生物技术全梳理;
中国抗体技术全梳理;
德琪医药技术全梳理;
德琪医药技术全梳理2.0;
和铂医药技术全梳理;
荣昌生物技术全梳理;
再鼎医药技术全梳理;
药明生物技术全梳理;
恒瑞医药技术全梳理;
豪森药业技术全梳理;
正大天晴技术全梳理;
吉凯基因技术全梳理;
基石药业技术全梳理;
百济神州技术全梳理;
百济神州技术全梳理第2版;
信达生物技术全梳理;
信达生物技术全梳理第2版;
中山康方技术全梳理;
复宏汉霖技术全梳理;
先声药业技术全梳理;
君实生物技术全梳理;
嘉和生物技术全梳理;
志道生物技术全梳理;
道尔生物技术全梳理;
尚健生物技术全梳理;
康宁杰瑞技术全梳理;
科望医药技术全梳理;
科望医药技术全梳理2.0;
岸迈生物技术全梳理;
礼进生物技术全梳理;
康桥资本技术全梳理;
余国良的抗体药布局;
荃信生物技术全梳理;
安源医药技术全梳理;
三生国健技术全梳理;
仁会生物技术全梳理;
乐普生物技术全梳理;
同润生物技术全梳理;
宜明昂科技术全梳理;
派格生物技术全梳理;
迈威生物技术全梳理;
Momenta技术全梳理;
NGM技术全梳理;
普米斯生物技术全梳理;
普米斯生物技术全梳理2.0;
三叶草生物技术全梳理;
贝达药业抗体药全梳理;
泽璟制药抗体药全梳理;
恒瑞医药抗体药全梳理;
齐鲁制药抗体药全梳理;
石药集团抗体药全梳理;
豪森药业抗体药全梳理;
华海药业抗体药全梳理;
科伦药业抗体药全梳理;
百奥泰技术全梳理;
凡恩世技术全梳理。
抗体药物偶联物临床1期蛋白降解靶向嵌合体
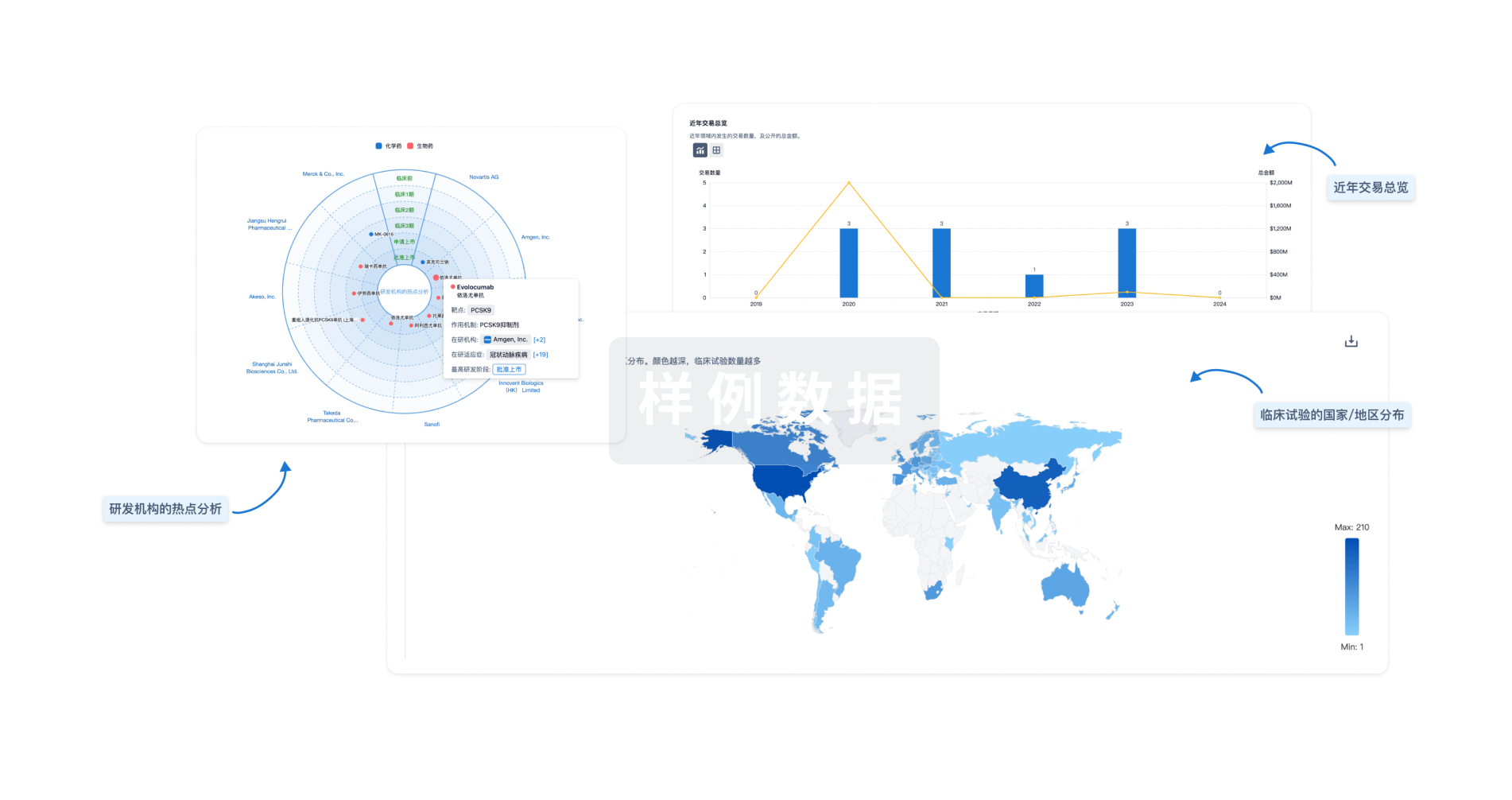
分析
对领域进行一次全面的分析。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用



