更新于:2024-11-01

Lyra Therapeutics, Inc.
更新于:2024-11-01
概览
标签
耳鼻咽喉疾病
免疫系统疾病
呼吸系统疾病
小分子化药
关联
1
项与 Lyra Therapeutics, Inc. 相关的药物靶点 |
作用机制 GR激动剂 |
在研机构 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期1987-04-30 |
5
项与 Lyra Therapeutics, Inc. 相关的临床试验ENLIGHTEN 2: A Phase III, Randomized, Blinded, Controlled, Parallel-Group Trial to Evaluate the Efficacy and Safety of LYR-210 for the Treatment of Chronic Rhinosinusitis in Adults
Multicenter, phase III, randomized, blinded, controlled, parallel group.
开始日期2022-05-13 |
申办/合作机构 |
ENLIGHTEN 1: A Phase III, Randomized, Blinded, Controlled, Parallel-Group Trial to Evaluate the Efficacy and Safety of LYR-210 for the Treatment of Chronic Rhinosinusitis (CRS) in Adults
Multicenter, phase III, randomized, blinded, controlled, parallel group with safety extension phase with crossover or continued treatment.
开始日期2022-01-27 |
申办/合作机构 |
BEACON: A Phase II, Patient-blinded, Two-part, Randomized, Parallel-group Trial to Evaluate the Safety, Tolerability, Pharmacokinetics, and Efficacy of LYR-220 in Chronic Rhinosinusitis (CRS) Patients Who Have Had a Prior Ethmoidectomy
This study will assess the safety, tolerability, pharmacokinetics and efficacy of two LYR-220 designs in symptomatic adult chronic rhinosinusitis (CRS) subjects who have had a prior functional endoscopic sinus surgery.
开始日期2021-11-23 |
申办/合作机构 |
100 项与 Lyra Therapeutics, Inc. 相关的临床结果
登录后查看更多信息
0 项与 Lyra Therapeutics, Inc. 相关的专利(医药)
登录后查看更多信息
2
项与 Lyra Therapeutics, Inc. 相关的文献(医药)2022-11-01·American Journal of Rhinology & Allergy
Pharmacokinetic Evidence of Steady and Sustained Drug Release from Long-Acting Implantable Corticosteroid Matrices for Chronic Rhinosinusitis
Article
作者: McIntyre, John ; Naclerio, Robert M. ; Shotts, Steven ; Kakarlapudi, Venkata ; You, Changcheng ; Shao, James ; Pappas, Alexander ; Brayton, Lindsay ; Ow, Randall A. ; Kuang, Yina
2022-03-01·American Journal of Rhinology & Allergy2区 · 医学
Drug Release and Pharmacokinetic Evaluation of Novel Implantable Mometasone Furoate Matrices in Rabbit Maxillary Sinuses
2区 · 医学
Article
作者: Pappas, Alexander ; Tseng, Ling-Fang ; Concagh, Danny ; You, Changcheng ; Kuang, Yina
65
项与 Lyra Therapeutics, Inc. 相关的新闻(医药)2024-10-15
-- Topline Results Expected Q2 2025 -- WATERTOWN, Mass., Oct. 15, 2024 (GLOBE NEWSWIRE) -- Lyra Therapeutics, Inc. (Nasdaq: LYRA) (“Lyra” or the “Company”), a clinical-stage biotechnology company developing long-acting anti-inflammatory therapies for the localized treatment of chronic rhinosinusitis (CRS), today announced that the pivotal Phase 3 ENLIGHTEN 2 clinical trial of LYR-210 in adult patients with CRS who have not had prior ethmoid sinus surgery, is fully enrolled. LYR-210 is a bioresorbable nasal implant designed to deliver six months of continuous anti-inflammatory medication (mometasone furoate) to the sinonasal passages for the treatment of CRS. Topline results from ENLIGHTEN 2 are expected in Q2 2025. “We are pleased to have fully enrolled the second of our two pivotal trials of LYR-210 in CRS patients, and our team is moving forward expeditiously to report the results in Q2 of next year,” said Maria Palasis, Ph.D., President and CEO of Lyra Therapeutics. “We thank the participants and the investigators in the ENLIGHTEN studies who have enabled the evaluation of our technology that could potentially benefit patients with CRS.” About the ENLIGHTEN Pivotal Program The ENLIGHTEN program consists of two pivotal Phase 3 clinical trials, ENLIGHTEN 1 and ENLIGHTEN 2, to evaluate the efficacy and safety of LYR-210 for the treatment of CRS. Each ENLIGHTEN trial has enrolled approximately 180 CRS patients who have failed medical management and have not had prior ethmoid sinus surgery, randomized 2:1 to either LYR-210 (7500µg mometasone furoate) or sham control for 24 weeks. About LYR-210 LYR-210 is an investigational product candidate for the treatment of chronic rhinosinusitis (CRS) in patients who have failed current therapies and require further intervention. LYR-210 is a bioresorbable nasal implant designed to be inserted in a simple, in-office procedure. LYR-210 is intended to deliver six months of continuous anti-inflammatory therapy, mometasone furoate, to the sinonasal passages to treat CRS. LYR-210 is being evaluated in the ENLIGHTEN pivotal Phase 3 clinical program. About Lyra TherapeuticsLyra Therapeutics, Inc. is a clinical-stage biotechnology company developing long-acting, anti-inflammatory sinonasal implants for the treatment of chronic rhinosinusitis (CRS). Lyra Therapeutics is developing therapies for CRS, a highly prevalent inflammatory disease of the paranasal sinuses which leads to debilitating symptoms and significant morbidities. LYR-210, the company’s lead product, is a bioabsorbable nasal implant designed to be administered in a simple, in-office procedure and is intended to deliver six months of continuous anti-inflammatory drug therapy (7500µg mometasone furoate) to the sinonasal passages for the treatment of CRS with a single administration. LYR-210, being evaluated in the ENLIGHTEN Phase 3 clinical program, is intended for patients with standard anatomy, primarily patients who have not undergone ethmoid sinus surgery. The company’s therapies are intended to treat the estimated four million CRS patients in the United States who fail medical management each year. For more information, please visit www.lyratx.com and follow us on LinkedIn. Forward-Looking StatementsThis press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including statements regarding whether LYR-210 could potentially benefit patients with CRS, the completion of the Company’s ENLIGHTEN 2 Phase 3 clinical trial, and the timing of the release of topline data from the ENLIGHTEN 2 Phase 3 clinical trial. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause the Company's actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. These and other important factors discussed under the caption "Risk Factors" in the Company's Quarterly Report on Form 10-Q filed with the SEC on August 14, 2024 and its other filings with the SEC could cause actual results to differ materially from those indicated by the forward-looking statements made in this press release. Any such forward-looking statements represent management's estimates as of the date of this press release. While the Company may elect to update such forward-looking statements at some point in the future, it disclaims any obligation to do so, even if subsequent events cause its views to change.
临床3期临床2期
2024-09-27
WATERTOWN, Mass., Sept. 27, 2024 (GLOBE NEWSWIRE) -- Lyra Therapeutics, Inc. (Nasdaq: LYRA), (“Lyra” or the “Company”), a clinical-stage biotechnology company developing long-acting anti-inflammatory therapies for the localized treatment of chronic rhinosinusitis (CRS), today announced that the Company will present results from the Phase 3 ENLIGHTEN 1 study for LYR-210, the company’s lead product candidate for CRS, at the 70th Annual Meeting of the American Rhinologic Society (ARS), taking place September 27-28 in Miami. The Company will also present additional results from the Phase 2 BEACON study for LYR-220 in adult patients with CRS who have had prior ethmoid sinus surgery at the Annual Meeting of the American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS), taking place September 28 - October 1 in Miami. A poster presentation on LYR-210 will be featured at the ARS Annual Meeting and an oral presentation on LYR-220 will be featured at the AAO-HNS Annual Meeting. Presentation Details: Poster PresentationAmerican Rhinologic Society (ARS) 2024 Annual Meeting Title: Efficacy and safety of LYR-210 for CRS from the pivotal Phase 3 ENLIGHTEN 1 trialPresenting Author: Vineeta Belanger, PhD, Lyra TherapeuticsDate and Time: Poster Session, Friday, September 27th at 1:00-8:00 p.m. ET Scientific Oral PresentationAmerican Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS) 2024 Annual Meeting Title: LYR-220 24-Week Steroid-Eluting Sinus Insert Improves Smell and Sleep in Patients with Chronic RhinosinusitisPresenting Author: Zachary M. Soler, MD, MSc, Medical University of South Carolina, Charleston, SCDate and Time: Sunday, September 29th at 1:06 p.m. ETSession: Rhinology/Allergy (1:00 – 2:00 p.m. ET) About Lyra Therapeutics Lyra Therapeutics, Inc. is a clinical-stage biotechnology company developing long-acting, anti-inflammatory sinonasal implants for the treatment of chronic rhinosinusitis (CRS). Lyra Therapeutics is developing therapies for CRS, a highly prevalent inflammatory disease of the paranasal sinuses which leads to debilitating symptoms and significant morbidities. LYR-210, the company’s lead product, is a bioabsorbable nasal implant designed to be administered in a simple, in-office procedure and is intended to deliver six months of continuous anti-inflammatory drug therapy (7500µg mometasone furoate) to the sinonasal passages for the treatment of CRS with a single administration. LYR-210, being evaluated in the ENLIGHTEN Phase 3 clinical program, is intended for patients with standard anatomy, primarily patients who have not undergone ethmoid sinus surgery. The company’s therapies are intended to treat the estimated four million CRS patients in the United States who fail medical management each year. For more information, please visit www.lyratx.com and follow us on LinkedIn. Forward-Looking Statements This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including statements regarding the date, time and details of the presentations at ARS and AAO-HNS. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause the Company's actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. These and other important factors discussed under the caption "Risk Factors" in the company's Quarterly Report on Form 10-Q filed with the SEC on August 14, 2024 and its other filings with the SEC could cause actual results to differ materially from those indicated by the forward-looking statements made in this press release. Any such forward-looking statements represent management's estimates as of the date of this press release. While the company may elect to update such forward-looking statements at some point in the future, it disclaims any obligation to do so, even if subsequent events cause its views to change. Contact Information:Jason Cavalier, Chief Financial Officer 917.584.7668 jcavalier@lyratx.com Media Contact:Kathryn Morris, The Yates Network LLC 914.204.6412kathryn@theyatesnetwork.com
临床2期临床3期临床结果
2024-08-14
– Primary focus continues to be on upcoming results from ENLIGHTEN 1 Phase 3 extension study in 4Q 2024 and ENLIGHTEN 2 pivotal Phase 3 trial in CRS patients in 1H 2025, as planned –
– In parallel, the company continues to analyze data from ENLIGHTEN 1; further analysis of the ENLIGHTEN 1 data has revealed that LYR-210 demonstrated improvement over control in symptomatic endpoints in the CRS patient cohort with nasal polyps –
WATERTOWN, Mass., Aug. 14, 2024 (GLOBE NEWSWIRE) -- Lyra Therapeutics, Inc. (Nasdaq: LYRA) (“Lyra” or the “Company”), a clinical-stage biotechnology company developing long-acting, anti-inflammatory sinonasal implants for the treatment of chronic rhinosinusitis (CRS), today reported its financial results for the second quarter ended June 30, 2024 and provided a corporate update.
“While we clearly recognize the disappointment of not meeting the primary endpoint in the previously-announced ENLIGHTEN 1 Phase 3 trial, our potential pathway to approval for LYR-210 in CRS without nasal polyps can only be determined once we unblind and analyze the full data set from the ENLIGHTEN pivotal program,” said Maria Palasis, Ph.D., President and CEO of Lyra Therapeutics. “Today we are disclosing that our further analysis of the ENLIGHTEN 1 data has revealed that LYR-210 demonstrated improvement over control in symptomatic endpoints in the CRS patient cohort with nasal polyps, which we believe reinforces the therapeutic potential of our product candidates.”
Dr. Palasis continued, “While we intend to remain opportunistic about strategic options, our primary focus remains on the two ongoing ENLIGHTEN Phase 3 trials evaluating LYR-210 in CRS patients with and without nasal polyps: the ENLIGHTEN 1 52-week extension study with results expected in Q4 2024 and the ENLIGHTEN 2 pivotal trial with enrollment on track and results expected in the first half of 2025. We plan to be pragmatic and data-driven as we determine our path forward for CRS patients, investors, and other stakeholders.”
Highlights from May 2024 ENLIGHTEN 1 Pivotal Results and Subsequent Cost-cutting Measures
On May 6, Lyra announced topline results from the Phase 3 ENLIGHTEN 1 trial showing that LYR-210 did not meet its primary endpoint of statistically significant improvement compared to sham control in the composite score of the three cardinal symptoms (3CS) of CRS (nasal obstruction, nasal discharge, facial pain/pressure) at 24 weeks. LYR-210 was generally well tolerated, with no product-related serious adverse events. Following the ENLIGHTEN 1 results disclosed in May, Lyra announced cost-cutting measures to preserve capital, including a reduction in force of approximately 75% of its workforce in addition to other measures to reduce costs and streamline operations. In connection with the reduction in force, which impacted 87 employees, Lyra stopped manufacturing and commercialization efforts and is seeking to sublease its three leaseholds to significantly reduce the Company’s operating costs. Furthermore, Lyra paused development efforts for LYR-220 in an effort to focus on the ongoing ENLIGHTEN Phase 3 program evaluating LYR-210.
Additional Analysis from ENLIGHTEN 1 for CRS Patient Subgroup with Nasal Polyps
Further analysis of the ENLIGHTEN 1 data shows that LYR-210 demonstrated a positive effect compared to sham control in 3CS and nasal congestion scores at 24 weeks in the CRS patient subgroup with nasal polyps. Treatment with LYR-210 resulted in a mean (standard error; SE) improvement in the 3CS score of 3.21 (0.436) points, compared to 0.96 (0.619) points in sham control for a difference of 2.25 points (p-value 0.0058) in the CRS patient subgroup with nasal polyps. This improvement was demonstrated despite the inclusion of only grade 1 nasal polyps in the study and without a threshold for nasal congestion score. For patients with nasal congestion score equal to or greater than 2 (that is moderate to severe symptom) at baseline in the CRS patient subgroup with nasal polyps, treatment with LYR-210 resulted in a mean (SE) improvement in the 3CS score of 3.69 (0.470) points, compared to 0.75 (0.685) points in sham control for a difference of 2.94 points (p-value 0.0017).Treatment with LYR-210 resulted in a mean (SE) improvement in the nasal congestion score of 1.20 (0.159) points, compared to 0.42 (0.243) points in sham control for a difference of 0.73 points (p-value 0.0216) in the CRS patient subgroup with nasal polyps and nasal congestion score equal to or greater than 2 at baseline.
Milestones for Ongoing ENLIGHTEN Pivotal Program of LYR-210 in CRS
Enrollment in ENLIGHTEN 2, the second pivotal Phase 3 trial of LYR-210 in CRS, is ongoing; enrollment completion is expected in the second half of 2024.Topline results from ENLIGHTEN 2 are expected in the first half of 2025.Results from the ENLIGHTEN 1 52-week extension study are expected in Q4 2024.
Second Quarter 2024 Financial Highlights
Cash, cash equivalents and short-term investments as of June 30, 2024 were $67.5 million, compared with $102.8 million at December 31, 2023. Based on our current business plan, we anticipate that our cash, cash equivalents and short-term investment balance is sufficient to fund our operating expenses and capital expenditures into the first quarter of 2026. Please see our Quarterly Report filed on Form 10-Q for the three and six months ended June 30, 2024 for further information regarding our cash runway guidance and other financial results.
Research and development expenses for the quarter ended June 30, 2024 were $13.3 million, an increase of $2.5 million compared to $10.8 million for the same period in 2023.
The increase in research and development expenses for the three months ended June 30, 2024 was primarily attributable to an increase of $1.7 million in allocated and support costs for shared activities within the organization driven by headcount allocation and rent increases which occurred prior to the reduction in force, an increase of $0.5 million in professional and consulting fees as we moved good manufacturing practices (“GMP”), manufacturing in house prior to the reduction in force and increased clinical and product manufacturing costs of $0.9 million as we continued to progress on our clinical trials and internal manufacturing efforts prior to the reduction in force. These costs were offset by $0.8 million in headcount related costs period over period due to the recent restructuring.
General and administrative expenses for the quarter ended June 30, 2024 were $5.1 million, an increase of $0.6 million compared to $4.5 million for the same period in 2023.
The increase in general and administrative expenses for the three months ended June 30, 2024 was primarily driven by an increase of $0.4 million for consulting costs, as well as an increase of $0.2 million in costs shared between the General & Administrative and Research & Development functions including headcount and rent. These costs were partially offset by a decrease in the amount of $0.1 million for employee related costs due to the recent restructuring.
The Company incurred impairment costs related to property and equipment of $1.9 million for the three months ended June 30, 2024 compared to $1.6 million for the same period in 2023.
The Company incurred impairment costs related to our right-of-use asset of $22.8 million for the three months ended June 30, 2024 and there were no such charges for the same period in 2023.
The Company incurred a restructuring charge in the amount of $6.5 million primarily related to severance and retention costs for the three months ended June 30, 2024 and there were no such charges for the same period in 2023.
Net loss for the second quarter 2024 was $48.1 million compared to $15.6 million for the same period in 2023.
About Lyra TherapeuticsLyra Therapeutics, Inc. is a clinical-stage biotechnology company developing long-acting, anti-inflammatory sinonasal implants for the treatment of chronic rhinosinusitis (CRS). Lyra Therapeutics has two product candidates, LYR-210 and LYR-220, in late-stage development for CRS, a highly prevalent inflammatory disease of the paranasal sinuses which leads to debilitating symptoms and significant morbidities. LYR-210 and LYR-220 are bioabsorbable nasal implants designed to be administered in a simple, in-office procedure and are intended to deliver six months of continuous anti-inflammatory drug therapy (7500µg mometasone furoate) to the sinonasal passages for the treatment of CRS with a single administration. LYR-210, being evaluated in the ENLIGHTEN Phase 3 clinical program, has a smaller dimension and is intended for patients with standard anatomy, primarily patients who have not undergone ethmoid sinus surgery. LYR-220 is a larger implant designed for CRS patients whose nasal cavity is enlarged due to previous ethmoid sinus surgery. These two product candidates are designed to treat the estimated four million CRS patients in the United States who fail medical management each year. For more information, please visit www.lyratx.com and follow us on LinkedIn.
Forward-Looking StatementsThis press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. The words “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” and similar expressions are intended to identify forward-looking statements. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including, without limitation, statements regarding our focus on the two ongoing ENLIGHTEN Phase 3 trials evaluating LYR-210, our ongoing ENLIGHTEN 1 extension study and expectation for data in Q4 2024, our ongoing ENLIGHTEN 2 trial and our expectation for data in 1H 2025, our cash runway into 2026 and plans to update investors regarding our cash runway, and our plans to evaluate potential strategic options to maximize shareholder value. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause the Company's actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: any potential financial or strategic option we pursue in order to maximize shareholder value may not result in the identification of a suitable transaction, or if one is identified and pursued, may not be completed on attractive terms, or at all; our ability to sublease or assign our three leaseholds, which represent significant operating costs; our incurrence of significant losses since inception and expectation to incur significant additional losses for the foreseeable future; our recurring losses from operations raise substantial doubt regarding our ability to continue as a going concern; our need for significant additional funding in order to complete development of and obtain regulatory approval for our product candidates and commercialize our products, if approved; the failure of our ENLIGHTEN 1 Phase 3 trial to meet its primary endpoint has made it more difficult for the Company to raise capital; we could be forced to delay, reduce, or eliminate our product development programs or commercialization efforts; following the failure of our ENLIGHTEN 1 Phase 3 trial evaluating LYR-210 for the treatment of CRS to meet its primary endpoint, which was announced in May 2024, there is significant uncertainty about the Company’s ability to complete development of LYR-210 and our ability to obtain regulatory approval for LYR-210 is at least significantly delayed and may not be possible; our common stock may be delisted from The Nasdaq Global Market if we cannot regain compliance with Nasdaq’s continued listing requirements; our loss of key personnel significantly and adversely affects our ability to manufacture our product candidates, among other activities; we are no longer engaged in the manufacturing of our product candidates in-house; our business is highly dependent on the success of our most advanced product candidate, LYR-210; clinical trials required for our current product candidate and any future product candidates are expensive and time-consuming, their outcome is uncertain, and if our clinical trials do not meet safety or efficacy endpoints in these evaluations, or if we experience significant delays in these trials, our ability to commercialize our product candidates and our financial position will be impaired; any failure by a third party to conduct our pre-clinical or clinical trials according to good clinical practices and in a timely manner may delay or prevent our ability to seek or obtain regulatory approval for or commercialize our product candidates; even if LYR-210 receives marketing approval, it may fail to achieve market acceptance by physicians, patients, third-party payors or others in the medical community necessary for commercial success; if our collaborations are not successful, including with LianBio our product candidates may not reach their full market potential; our ability to manage our obligations under our license and other strategic agreements may divert management time and our limited resources, causing delays or disruptions to our business; our operating activities may be restricted by certain covenants in our license and strategic agreements, which could limit our development and commercial opportunities; failure to obtain marketing approval in international jurisdictions would prevent our products from being marketed in such jurisdictions; developments by competitors may render our products or technologies obsolete or non-competitive or may reduce the size of our markets; the successful commercialization of our product candidates will depend in part on the extent to which governmental authorities and health insurers establish coverage, adequate reimbursement levels and pricing policies; failure to obtain or maintain coverage and adequate reimbursement for our product candidates, if approved, could limit our ability to market those products and decrease our ability to generate revenue; if we are unable to obtain, maintain, or adequately protect our intellectual property rights, we may not be able to compete effectively in our market; the impact of international terrorism, political unrest and wars on our business; and the impact of other events such as the COVID-19 pandemic may adversely impact our business and operations, including our clinical trials. These and other important factors discussed under the caption "Risk Factors" in the Company's Quarterly Report on Form 10-Q filed with the SEC on August 14, 2024 and its other filings with the SEC could cause actual results to differ materially from those indicated by the forward-looking statements made in this press release. Any such forward-looking statements represent management's estimates as of the date of this press release. While the Company may elect to update such forward-looking statements at some point in the future, it disclaims any obligation to do so, even if subsequent events cause its views to change.
LYRA THERAPEUTICS, INC. CONDENSED CONSOLIDATED BALANCE SHEETS(unaudited)(in thousands, except share data) June 30, December 31, 2024 2023 Assets
Current assets:
Cash and cash equivalents $31,905 $22,353 Short-term investments 35,593 80,400 Prepaid expenses and other current assets 1,937 2,068 Total current assets 69,435 104,821 Property and equipment, net 1,665 2,043 Operating lease right-of-use assets 21,490 33,233 Restricted cash 1,992 1,392 Other assets — 1,111 Total assets $94,582 $142,600 Liabilities and Stockholders’ Equity
Current liabilities:
Accounts payable $4,971 $3,131 Restructuring liability 3,127 — Accrued expenses and other current liabilities 6,095 9,374 Operating lease liabilities 4,269 5,434 Deferred revenue 814 1,658 Total current liabilities 19,276 19,597 Operating lease liabilities, net of current portion 32,479 21,447 Deferred revenue, net of current portion 11,850 12,136 Total liabilities 63,605 53,180 Commitments and contingencies
Stockholders’ equity:
Preferred stock, $0.001 par value, 10,000,000 shares authorized at June 30, 2024 and December 31, 2023; no shares issued and outstanding at June 30, 2024 and December 31, 2023 — — Common stock, $0.001 par value; 200,000,000 shares authorized at June 30, 2024 and December 31, 2023; 65,455,735 and 57,214,550 shares issued and outstanding at June 30, 2024 and December 31, 2023, respectively 65 57 Additional paid-in capital 412,854 400,685 Accumulated other comprehensive income (loss), net of tax (4) 33 Accumulated deficit (381,938) (311,355)Total stockholders’ equity 30,977 89,420 Total liabilities and stockholders’ equity $94,582 $142,600
LYRA THERAPEUTICS, INC. CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS(unaudited)(in thousands, except share and per share data) Three Months EndedJune 30, Six Months Ended June 30, 2024 2023 2024 2023 Collaboration revenue $598 $458 $1,130 $868
Operating expenses:
Research and development 13,264 10,799 31,502 23,395 General and administrative 5,139 4,570 10,957 9,697 Impairment of property and equipment 1,883 1,592 1,883 1,592 Impairment of right-of-use asset 22,836 — 22,836 — Restructuring and other related charges 6,450 — 6,450 — Total operating expenses 49,572 16,961 73,628 34,684 Loss from operations (48,974) (16,503) (72,498) (33,816) Other income:
Interest income 855 897 1,941 1,969 Total other income 855 897 1,941 1,969 Loss before income tax expense (48,119) (15,606) (70,557) (31,847) Income tax expense (12) (12) (26) (26) Net loss (48,131) (15,618) (70,583) (31,873) Other comprehensive loss:
Unrealized holding loss on short-term investments, net of tax (29) (15) (37) (37) Comprehensive loss $(48,160) $(15,633) $(70,620) $(31,910) Net loss per share attributable to common stockholders— basic and diluted $(0.74) $(0.36) $(1.09) $(0.79) Weighted-average common shares outstanding— basic and diluted 65,459,678 43,676,387 64,739,520 40,273,472
财报临床3期临床结果
100 项与 Lyra Therapeutics, Inc. 相关的药物交易
登录后查看更多信息
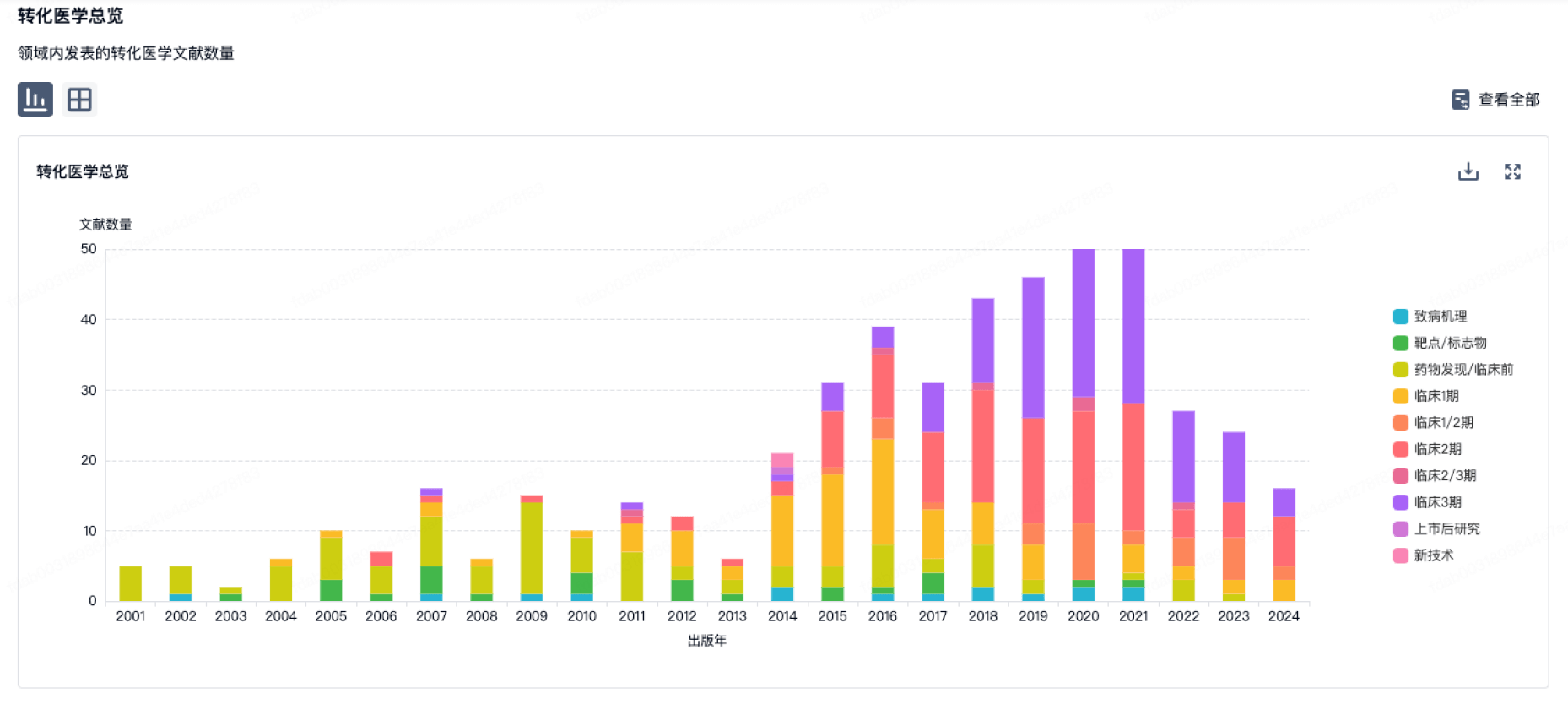
100 项与 Lyra Therapeutics, Inc. 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2024年11月18日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
临床3期
1
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

