预约演示
更新于:2025-10-14
Denosumab biosimilar (T-Mab Biopharma)
地舒单抗生物类似药 (江苏泰康)
更新于:2025-10-14
概要
基本信息
原研机构 |
非在研机构- |
最高研发阶段批准上市 |
首次获批日期 中国 (2023-03-28), |
最高研发阶段(中国)批准上市 |
特殊审评- |
登录后查看时间轴
结构/序列
Sequence Code 94568L

当前序列信息引自: *****
Sequence Code 143522H

当前序列信息引自: *****
关联
11
项与 地舒单抗生物类似药 (江苏泰康) 相关的临床试验ChiCTR2400095054
A single-arm, prospective clinical study on the efficacy and safety of denosumab in patients with bone metastases from solid tumors who have experienced the first bone-related event.
开始日期2025-01-01 |
申办/合作机构 |
NCT06804590
A Randomized, Double-blind, Parallel-group, Phase III Study to Compare the Clinical Efficacy, Safety, and Immunogenicity of Denosumab Injection 9MW0311 With Prolia® in Postmenopausal Women With Osteoporosis
This study is a multicenter, randomized, double-blinded, parallel-group Phase III clinical study to compare the clinical efficacy, safety, and immunogenicity of 9MW0311 and Prolia® in Chinese postmenopausal women with osteoporosis at high risk for fracture.
开始日期2024-11-16 |
申办/合作机构 |
ChiCTR2400088032
Efficacy and safety evaluation of denosumab in maintenance hemodialysis patients witn osteoporosis:a randomized controlled, multicenter, optimal efficacy study
开始日期2024-08-01 |
申办/合作机构 |
100 项与 地舒单抗生物类似药 (江苏泰康) 相关的临床结果
登录后查看更多信息
100 项与 地舒单抗生物类似药 (江苏泰康) 相关的转化医学
登录后查看更多信息
100 项与 地舒单抗生物类似药 (江苏泰康) 相关的专利(医药)
登录后查看更多信息
4
项与 地舒单抗生物类似药 (江苏泰康) 相关的文献(医药)2024-07-02·EXPERT OPINION ON BIOLOGICAL THERAPY
Randomized, double-blind, placebo-controlled, multicenter study to evaluate efficacy and safety of the denosumab biosimilar MW031 in Chinese postmenopausal women with osteoporosis
Article
作者: Xue, Qingyun ; Yang, Yunfa ; Guo, Yinhan ; Xia, Weibo ; Li, Yufeng ; Xiao, Yanfeng ; Huo, Yanan ; Yu, Wei ; Xu, Yongsheng ; Liu, Feng ; Jiang, Yan ; Ma, Wen ; Wang, Bingwu ; Hu, Zhitian ; Kong, Xijian ; Li, Yukun ; Zheng, Xin
BACKGROUND:
This study aimed to assess the efficacy and safety of MW031 in Chinese postmenopausal women with osteoporosis.
PATIENTS AND METHODS:
In this randomized, double-blind, placebo-controlled, multicenter clinical trial, 448 postmenopausal women with osteoporosis were randomized 3:1 to receive MW031 and placebo for 12 months. The primary efficacy endpoint was the percentage change from baseline in BMD at lumbar spine in month 12. The safety and immunogenicity profiles were also included.
RESULTS:
Of 448 randomized patients, 421 completed the study (MW031, n = 322; placebo, n = 99).After 12 months of MW031 treatment, BMD increased by 5.80% at lumbar spine,3.65% at total hip, and 2.93% at femoral neck. The model-adjusted difference was 3.86% (P<0.0001), 2.34% (P<0.0001), and 1.05% (p = 0.08) compared with placebo group, respectively. For the bone turnover markers, serum CTX level in MW031 group decreased to the maximum difference in month 1 (-71.71%, 95% CI: -77.83%, -65.60%, P<0.0001) compared with the placebo group. The safety analysis showed no significant differences in the proportion of patients reporting any adverse events between the two groups.
CONCLUSION:
This study demonstrated that MW031 safely and effectively increased BMD and rapidly decreased the level of bone resorption marker in Chinese postmenopausal women with osteoporosis.
TRIAL REGISTRATION:
NCT05215977 (ClinicalTrials.gov.).
2024-04-01·JAMA oncology
Efficacy, Safety, and Population Pharmacokinetics of MW032 Compared With Denosumab for Solid Tumor–Related Bone Metastases
Article
作者: Wang, Rui ; Chen, Xiaoyuan ; Zhong, Jincai ; Guo, Zengqing ; Li, Dairong ; Yang, Shu ; Sun, Hongmei ; Yu, Zhuang ; Yao, Herui ; Wang, Jingfen ; Xie, Weimin ; Liu, Hu ; Wu, Fang ; Li, Wei ; Xiong, Hailin ; Lei, Kaijian ; Wang, Hong ; Liu, Datao ; Nie, Jianyun ; Song, Haifeng ; Guo, Yinhan ; Ji, Xuening ; Guo, Yanzhen ; Zhang, Shaohua ; Zhang, Longzhen ; Lu, Junguo ; Jiang, Zefei ; Yin, Yongmei ; Wu, Huijing ; Zhang, Hua ; Tang, Jinhai ; Jin, Hongyan ; Zhong, Diansheng ; Yao, Weirong ; Ma, Wen ; Zhang, Qingyuan ; Sun, Tao ; Hu, Zhitian ; Yu, Ding
Importance:
The bioequivalence of denosumab biosimilar has yet to be studied in a 53-week, multicenter, large-scale, and head-to-head trial. A clinically effective biosimilar may help increase access to denosumab in patients with solid tumor–related bone metastases.
Objectives:
To establish the biosimilarity of MW032 to denosumab in patients with solid tumor–related bone metastases based on a large-scale head-to-head study.
Design, Setting, and Participants:
In this 53-week, randomized, double-blind, phase 3 equivalence trial, patients with solid tumors with bone metastasis were recruited from 46 clinical sites in China. Overall, 856 patients were screened and 708 eligible patients were randomly allocated to receive either MW032 or denosumab.
Interventions:
Patients were randomly assigned (1:1) to receive MW032 or reference denosumab subcutaneously every 4 weeks until week 49.
Main Outcomes and Measures:
The primary end point was percentage change from baseline to week 13 of natural logarithmic transformed urinary N-telopeptide/creatinine ratio (uNTx/uCr).
Results:
Among the 701 evaluable patients (350 in the MW032 group and 351 in the denosumab group), the mean (range) age was 56.1 (22.0-86.0) years and 460 patients were women (65.6%). The mean change of uNTx/uCr from baseline to week 13 was −72.0% (95% CI, −73.5% to −70.4%) in the MW032 group and −72.7% (95% CI, −74.2% to −71.2%) in the denosumab group. These percent changes corresponded to mean logarithmic ratios of −1.27 and −1.30, or a difference of 0.02. The 90% CI for the difference (−0.04 to 0.09) was within the equivalence margin (−0.13 to 0.13); the mean changes of uNTx/uCr and bone-specific alkaline phosphatase (s-BALP) at each time point were also similar during 53 weeks. The differences of uNTx/uCr change were 0.015 (95% CI, −0.06 to 0.09), −0.02 (95% CI, −0.09 to 0.06), −0.05 (95% CI, −0.13 to 0.03) and 0.001 (95% CI, −0.10 to 0.10) at weeks 5, 25, 37, and 53, respectively. The differences of s-BALP change were −0.006 (95% CI, 0.06 to 0.05), 0.00 (95% CI, −0.07 to 0.07), −0.085 (95% CI, −0.18 to 0.01), −0.09 (95% CI, −0.20 to 0.02), and −0.13 (95% CI, −0.27 to 0.004) at weeks 5, 13, 25, 37 and 53, respectively. No significant differences were observed in the incidence of skeletal-related events (−1.4%; 95% CI, −5.8% to 3.0%) or time to first on-study skeletal-related events (unadjusted HR, 0.86; P = .53; multiplicity adjusted HR, 0.87; P = .55) in the 2 groups.
Conclusions and Relevance:
MW032 and denosumab were biosimilar in efficacy, population pharmacokinetics, and safety profile. Availability of denosumab biosimilars may broaden the access to denosumab and reduce the drug burden for patients with advanced tumors.
Trial Registration:
ClinicalTrials.gov Identifier: NCT04812509
2023-08-03·Expert opinion on biological therapy
Pharmacokinetics, pharmacodynamics, safety and immunogenicity of recombinant, fully human anti-RANKL monoclonal antibody (MW031) versus denosumab in Chinese healthy subjects: a single-center, randomized, double-blind, single-dose, parallel-controlled trial
Article
作者: Xu, Wenyu ; Hou, Jie ; Xiao, Yanfeng ; Guo, Yinhan ; Hu, Zhitian ; Di, Yujing ; Yu, Heze ; Guo, Tingting
BACKGROUND:
MW031 is a biosimilar candidate of denosumab (Prolia®). This study aimed to compare the pharmacokinetics, pharmacodynamics, safety and immunogenicity of MW031 to denosumab in healthy Chinese participants.
RESEARCH DESIGN AND METHODS:
In this single-center, randomized, double-blind, parallel-controlled, single-dose trial, participants were given 60 mg MW031 (N = 58) or denosumab (N = 61) by subcutaneous injection and observed for 140 days. The primary endpoint was the bioequivalence of PK parameters (Cmax, AUC0-∞), and secondary endpoints including PD parameter, safety, and immunogenicity.
RESULTS:
A comparison of main PK parameters showed that the geometric mean ratios (GMR) (90% confidence intervals [CIs]) of AUC0-∞ and Cmax for MW031 over denosumab were 105.48% (98.96%, 112.43%) and 98.58% (92.78%, 104.75%), respectively. The inter-CV values of AUC0-∞ and Cmax for MW031 ranged from 19.9% to 23.1%. PD parameter (sCTX) in the MW031 and denosumab groups were similar, and the positivity rates of immunogenicity were 0% in both groups. This study also showed similar safety profiles in both groups, and there were no drug-related, high-incidence and previously unreported adverse reactions.
CONCLUSION:
This trial confirmed similar pharmacokinetic profiles of MW031 and denosumab in healthy male participants, and pharmacodynamic profile, immunogenicity and safety were comparable for both drugs.
TRIAL REGISTRATION:
NCT04798313; CTR20201149.
57
项与 地舒单抗生物类似药 (江苏泰康) 相关的新闻(医药)2025-09-19
·氨基观察
氨基观察-创新药组原创出品
作者 | 黄凯
HPV免费接种范围越来越广。
9月19日,国务院新闻办公室举行新闻发布会,发布《新时代中国推进妇女全面发展的实践与成就》白皮书。白皮书介绍,截至2025年6月,18个省份通过将接种人乳头瘤病毒(HPV)疫苗纳入政府民生实事等方式,为适龄女童免费接种疫苗,每年惠及约500万女童。
信达生物玛仕度肽获批新适应症。
9月19日,据NMPA官网,信达生物的胰高血糖素样肽-1受体(GLP-1R)/胰高血糖素受体(GCGR)双重激动剂玛仕度肽的新适应症获批上市,用于成人2型糖尿病患者的血糖控制。
在过去的一天里,国内外医药市场还有哪些热点值得关注?让氨基君带你一探究竟。
/ 01 /
市场速递
1)18个省份为适龄女童免费接种HPV疫苗
9月19日,国务院新闻办公室举行新闻发布会,发布《新时代中国推进妇女全面发展的实践与成就》白皮书。白皮书介绍,截至2025年6月,18个省份通过将接种人乳头瘤病毒(HPV)疫苗纳入政府民生实事等方式,为适龄女童免费接种疫苗,每年惠及约500万女童。
/ 02 /
医药动态
1)美雅珂生物注射用MRG003拟获突破性疗法认定
9月19日,据CDE官网,美雅珂生物注射用MRG003拟获突破性疗法认定,适应症为联合HX008(普特利单抗)注射液用于治疗既往至少经过铂类和PD-1/L1治疗失败的复发或转移性鼻咽癌。
2)复宏汉霖地舒单抗类似药获欧盟注册批准
9月19日,复星医药公告称,公司控股子公司复宏汉霖自主研发的BILDYOS®和BILPREVDA®两个地舒单抗注射液产品获欧盟委员会批准上市,适应症包括骨折高风险的绝经后妇女及男性的骨质疏松症治疗等。此外,HLX14于美国获批用于特定人群骨质疏松症治疗等8项适应症,于加拿大的上市注册申请已获受理。HLX14的商业化权利已授予Organon LLC,复宏汉霖将根据许可协议享有相关权利。
3)迈威生物9MW0813注射液上市许可申请获得受理
9月19日,迈威生物公告称,公司收到国家药品监督管理局核准签发的《受理通知书》,9MW0813注射液的上市许可申请获得受理,用于糖尿病性黄斑水肿(DME)和新生血管(湿性)年龄相关性黄斑变性(nAMD)。
4)信达生物GLP-1R/GCGR激动剂玛仕度肽获批新适应症
9月19日,据NMPA官网,信达生物的胰高血糖素样肽-1受体(GLP-1R)/胰高血糖素受体(GCGR)双重激动剂玛仕度肽的新适应症获批上市,用于成人2型糖尿病患者的血糖控制。
5)驯鹿生物发布CAR-T产品治疗多发性骨髓瘤3年随访数据
9月19日,驯鹿生物宣布,其自主研发的全人源靶向BCMA CAR-T细胞治疗产品福可苏®治疗复发/难治性多发性骨髓瘤(R/R MM)的FUMANBA-1研究36个月长期随访数据,在国际骨髓瘤学会(IMS)年会期间正式发布。研究结果进一步证实了伊基奥仑赛注射液能够为R/R MM患者(包括具有高危特征患者)带来深度且持久的疗效,并具有可控的长期安全性,显著提升患者的长期生存质量。
/ 03 /
海外药闻
1)Arvinas/辉瑞欲出售PROTAC药物商业化权益
9月17日,Arvinas宣布与辉瑞达成共识,计划将口服、靶向雌激素受体(ER)的蛋白降解靶向嵌合体药物Vepdegestrant的商业化权益授权给第三方。
PS:欢迎扫描下方二维码,添加氨基君微信号交流。
上市批准免疫疗法细胞疗法突破性疗法疫苗
2025-09-05
SHANGHAI, Sept. 5, 2025 /PRNewswire/ -- Mabwell (688062.SH), an innovation-driven biopharmaceutical company with entire industry chain, and Pakistan's The Searle Company Limited (PSX: SEARL, hereinafter "Searle") jointly announced that two denosumab products, 9MW0311 and 9MW0321, have received marketing authorization from Drug Regulatory Authority of Pakistan (DRAP). This marks both Pakistan's first regulatory approval for a denosumab biosimilar and Mabwell's first overseas marketing authorization for its products.
As a key Belt and Road Initiative partner with a population of 240 million, Pakistan presents substantial pharmaceutical market potential, aligning with Mabwell's strategic expansion into high-growth emerging markets. Earlier, Mabwell entered into a licensing agreement with Searle to facilitate local fill-finish and commercialization of Denosumab in Pakistan.
Mr. Hu Huiguo, Board Member, Senior Vice President and Board Secretary of Mabwell, said, "The successful approval of denosumab in Pakistan provides local patients with enhanced treatment options and high-quality biologics, marking another milestone in Mabwell's global commercialization strategy. Moving forward, we remain committed to expanding the product's commercialization to additional countries, thereby improving global accessibility to denosumab injections and meeting the therapeutic needs of more patients worldwide."
"With the launch of denosumab, we improve the accessibility of this product, and reaffirm our position as a pioneer in biosimilars in Pakistan," said Tahir Ahmed, CEO of Searle, "This approval is a testament to our continued dedication to bringing high-quality, life-saving therapeutics to the region. We remain focused on improving patient outcomes through continued scientific excellence and strategic collaboration."
About 9MW0311
9MW0311 is a recombinant fully humanized anti-RANKL monoclonal antibody injection (60mg), developed as a Prolia® biosimilar. On March 28, 2023, the marketing application for 9MW0311 was approved by the National Medical Products Administration (NMPA) for the treatment of postmenopausal women with osteoporosis at high risk of fracture in China. Additional supplemental applications are planned to expand indications, including: osteoporosis in men at high risk of fracture, and glucocorticoid-induced osteoporosis in patients at high fracture risk. According to Frost & Sullivan data, the global market size for RANKL monoclonal antibody therapies in osteoporosis treatment has reached $1.317 billion in 2024.
About 9MW0321
9MW0321 is a recombinant fully humanized anti-RANKL monoclonal antibody injection (120mg) developed as a XGEVA® biosimilar. The NMPA of China granted marketing approval for 9MW0321 on March 29, 2024, establishing it as China's first approved XGEVA® biosimilar. It is indicated for the treatment of adults and skeletally mature adolescents (defined as at least 1 mature long bone and weighing ≥ 45 kg) with giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity. Mabwell is currently advancing supplementary applications to extend 9MW0321's indications to include malignancies associated with bone metastasis. BeOne Medicines and Amgen reported commercial performance data showing XGEVA® achieved annual sales of RMB 1.598 billion in China and USD 2.225 billion globally during 2024, with first half 2025 sales reaching RMB 1.090 billion in China and USD 1.098 billion worldwide. Future growth is anticipated to be predominantly driven by expansion in China and emerging markets.
About Searle
Established in 1965, The Searle Company Limited is among Pakistan's leading pharmaceutical companies, with eight state-of-the-art manufacturing facilities, including a dedicated biotechnology plant. With operations in over 20 countries and rapid expansion across multiple global regions, Searle is committed to delivering innovative treatments for life-threatening conditions. The company's biosimilar portfolio is among the most comprehensive in the region.
About Mabwell
Mabwell (688062.SH) is an innovation-driven biopharmaceutical company with the entire value chain of the pharmaceutical industry. The company provides more effective and accessible therapy and innovative medicines to fulfill global medical needs, focusing on oncology and aging-related disease indications. Mabwell's mission is "Explore Life, Benefit Health" and its vision is "Innovation, from ideas to reality". For more information, please visit .
Forward-Looking Statements
This press release contains forward-looking statements including, but not limited to, the potential safety, efficacy, regulatory review or approval and commercial success of our product candidates and those relating to the Company's product development, clinical studies, clinical and regulatory milestones and timelines, market opportunity, competitive position, possible or assumed future results of operations, business strategies, potential growth opportunities and other statements that are predictive in nature. "Forward-looking statements" are statements that are not historical facts and involve a number of risks and uncertainties, which may cause actual results to be materially different from any future results expressed or implied in the forward-looking statements. These statements may be identified by the use of forward-looking expressions, including, but not limited to, "expect," "anticipate," "intend," "plan," "believe," "estimate," "potential," "predict," "project," "should," "would," and similar expressions and the negatives of those terms.
Forward-looking statements are based on the Company's current expectations and assumptions. Forward-looking statements are subject to a number of risks, uncertainties, and other factors, many of which are beyond the Company's control, including, but not limited to: environment; politic; economy; society; legislation; our dependence on our product candidates, most of which are still in preclinical or various stages of clinical development; our reliance on third-party vendors, such as contract research organizations and contract manufacturing organizations; the uncertainties inherent in clinical testing; our ability to complete required clinical trials for our product candidates and obtain approval from regulatory authorities for our product candidates; our ability to protect our intellectual property; the potential impact of COVID-19; the loss of any executive officers or key personnel. In case one or more of these risks or uncertainties deteriorate, or any assumptions are incorrect, the actual results may be seriously inconsistent with the stated results.
The Company cautions all the persons not to place undue reliance on any such forward-looking statements, which speaks only as of the date of this press release. The Company disclaims any obligation, except as specifically required by law and the rules of the applicable Stock authority to publicly update or revise any such statements to reflect any change in expectations or in events, conditions, or circumstances on which any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements. All forward-looking descriptions, figures and assumptions in this press release are applicable to this statement.
SOURCE Mabwell
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
上市批准引进/卖出
2025-09-05
·美通社
上海
2025年9月5日
/美通社/ -- 迈威生物(688062.SH),一家全产业链布局的创新型生物制药公司,与巴基斯坦制药公司 THE SEARLE COMPANY LIMITED. (巴基斯坦证券交易所代码:SEARL,以下简称 Searle)共同宣布两款地舒单抗注射液 9MW0311 和 9MW0321(国内商品名:迈利舒®和迈卫健®)已获得巴基斯坦药品监督管理局(Drug Regulatory Authority of
Pakistan
, DRAP)的注册批准。这是巴基斯坦药品监督管理局首次批准地舒单抗生物类似药,也是迈威生物产品首次获得海外注册批件。
迈威生物与 Searle 共庆地舒单抗在巴基斯坦获批
巴基斯坦是重要的"一带一路"国家,人口达 2.4 亿,医药需求日益增长,具备广阔的市场潜力,是迈威生物重点布局的新兴市场之一。迈威生物此前与 Searle 就地舒单抗注射液产品达成许可协议,共同在巴基斯坦实现地舒单抗的当地灌装和商业化。9MW0311 和 9MW0321 已在 30 个国家签署正式合作协议,并已分别向约旦、埃及、巴西等国家提交了注册申请文件,其他国家注册申请也在准备中。
迈威生物董事、高级副总裁、董秘胡会国表示:"此次地舒单抗在巴基斯坦成功获批上市,让当地患者获得了更多治疗选择和高质量的生物药,也是迈威生物全球商业化布局的又一里程碑。未来我们将继续推动产品在更多国家的商业化,进一步提升地舒单抗注射液的全球可及性,满足全球更多患者的需求。"
Searle 首席执行官Tahir Ahmed 表示:"此次获批提升了地舒单抗在巴基斯坦的可及性,体现了我们坚持践行持续为本地患者带来高质量治疗药物的承诺,也进一步巩固了我们在巴基斯坦生物类似药领域的领先地位。我们将持续致力于通过卓越的科研与战略合作,改善患者的治疗结果。"
关于
9MW0311
9MW0311 为重组全人源抗 RANKL 单克隆抗体注射液(60mg),是地舒单抗 Prolia®(普罗力®)的生物类似药;2023 年 3 月 28 日,9MW0311(国内商品名:迈利舒®)上市申请获得国家药品监督管理局批准,用于骨折高风险的绝经后妇女的骨质疏松症;在绝经后妇女中,本品可显著降低椎体、非椎体和髋部骨折的风险。后续拟递交补充申请,增加骨折高风险的男性骨质疏松症适应症以及骨折高风险的糖皮质激素诱导的骨质疏松症。根据弗若斯特沙利文的数据,用于治疗骨质疏松的 RANKL 单抗药物市场规模 2024 年为 13.17 亿美元。
关于
9MW0321
9MW0321为重组全人源抗 RANKL 单克隆抗体注射液(120mg),是地舒单抗 XGEVA®(安加维®)的生物类似药。2024年3月29日,9MW0321(国内商品名:迈卫健®)上市申请获得国家药品监督管理局批准,用于治疗不可手术切除或者手术切除可能导致严重功能障碍的骨巨细胞瘤(GCTB),包括成人和骨骼发育成熟(定义为至少 1 处成熟长骨且体重 ≥45kg)的青少年患者,为中国首款获批上市的安加维®生物类似药。公司正在推进迈卫健®用于肿瘤骨转移相关适应症的补充申请工作。根据百济神州和安进定期报告,2024年安加维®在中国的销售额为 15.98 亿人民币,全球销售额为 22.25 亿美元;2025年上半年安加维®中国销售额为 10.90 亿人民币,全球销售额为 10.98 亿美元。预计未来的增长将主要来自中国及新兴市场。
关于
Searle
THE SEARLE COMPANY LIMITED. (Searle) 成立于 1965 年,是巴基斯坦头部制药公司之一,拥有八个先进的生产基地,其中包括一座生物制品工厂。Searle 始终致力于为重大疾病提供创新治疗方案,业务遍及 20 多个国家,并在全球多个区域快速扩张。它是所在区域内生物类似药产品布局最完善的公司之一。
关于迈威生物
迈威生物 (688062.SH) 是一家全产业链布局的创新型生物制药公司,始终秉承"让创新从梦想变成现实"的愿景,践行"探索生命,惠及健康"的使命,通过源头创新,为患者提供疗效更好、可及性更强的生物创新药,满足全球未被满足的临床需求。2017 年成立以来,迈威生物构建了以抗体药物靶点发现与分子发现为起点,覆盖成药性研究、临床前研究、临床研究和生产转化等药品研发全周期的创新体系,实现集研发、生产、营销于一体的全产业链布局。我们专注于肿瘤和年龄相关疾病,涉及肿瘤、自身免疫、骨疾病、眼科、血液等治疗领域,凭借国际领先的特色技术平台和研发创新能力,建立了丰富且具有竞争力的管线。现有 14 个处于临床前、临床或上市阶段的重点品种,包括 10 个创新品种和 4 个生物类似药,其中 4 个品种上市,1 个品种处于提交上市许可申请准备阶段,2 个品种处于 III 期关键注册临床阶段。并独立承担 1 项国家"重大新药创制"重大科技专项、2 项国家重点研发计划和多个省市级科技创新项目。迈威生物以创新为本,注重产业转化,符合中国 NMPA、美国 FDA、欧盟 EMA GMP标准的抗体和重组蛋白药物产业化基地已在江苏泰州投入使用,并已通过欧盟 QP 审计,位于上海金山和江苏泰州的大规模商业化生产基地正在建设中。欲了解更多信息,请访问:www.mabwell.com。
前瞻性声明
本新闻稿所发布的信息中可能会包含某些前瞻性表述。这些表述本质上具有相当风险和不确定性。在使用"预期"、"相信"、"预测"、"期望"、"打算"及其他类似词语进行表述时,凡与本公司有关的,目的均是要指明其属前瞻性表述。本公司并无义务不断地更新这些预测性陈述。
这些前瞻性表述乃基于本公司管理层在做出表述时对未来事务的现有看法、假设、期望、估计、预测和理解。这些表述并非对未来发展的保证,会受到风险、不确性及其他因素的影响,有些乃超出本公司的控制范围,难以预计。因此,受我们的业务、竞争环境、政治、经济、法律和社会情况的未来变化及发展的影响,实际结果可能会与前瞻性表述所含资料有较大差别。
本公司、本公司董事及雇员代理概不承担 (a) 更正或更新本网站所载前瞻性表述的任何义务;及 (b) 若因任何前瞻性表述不能实现或变成不正确而引致的任何责任。
生物类似药上市批准
100 项与 地舒单抗生物类似药 (江苏泰康) 相关的药物交易
登录后查看更多信息
研发状态
批准上市
10 条最早获批的记录, 后查看更多信息
登录
| 适应症 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|
| 骨巨细胞瘤 | 中国 | 2024-03-29 | |
| 绝经期后骨质疏松 | 中国 | 2023-03-28 |
未上市
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 骨病 | 临床3期 | 中国 | 2020-03-20 | |
| 骨转移癌 | 临床3期 | 中国 | 2020-03-20 | |
| 髋部骨折 | 临床3期 | 中国 | 2019-12-27 | |
| 骨癌 | 临床1期 | 中国 | 2019-06-25 | |
| 乳腺癌 | 临床1期 | 中国 | - | 2017-07-20 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床3期 | 421 | 選製構鹽衊淵鏇淵糧衊(製觸選獵選齋淵艱淵艱) = 築鑰鹽簾顧遞顧醖艱鬱 範膚膚夢積鹽鹹憲積廠 (選構醖蓋繭簾醖顧夢憲 ) 更多 | 积极 | 2024-05-16 | |||
placebo | - | ||||||
临床3期 | 701 | 夢壓鹽範壓願鑰淵壓簾(襯衊繭蓋鬱觸鹹鬱製艱) = 構餘醖膚構願範積膚衊 膚鑰餘願餘鹽襯襯糧艱 (憲網壓淵選鏇獵窪膚蓋, -73.5 ~ -70.4) | 相似 | 2024-02-08 | |||
夢壓鹽範壓願鑰淵壓簾(襯衊繭蓋鬱觸鹹鬱製艱) = 襯廠願築願鬱廠膚衊鑰 膚鑰餘願餘鹽襯襯糧艱 (憲網壓淵選鏇獵窪膚蓋, -74.2 ~ -71.2) | |||||||
临床1期 | 40 | 鏇夢醖醖廠觸製壓範膚(選範築鹽醖網遞齋廠網) = 10.0% 鏇觸構壓鹽鹽淵襯廠襯 (鏇艱鹽築選鏇鑰蓋艱範 ) 更多 | 积极 | 2019-02-15 | |||
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

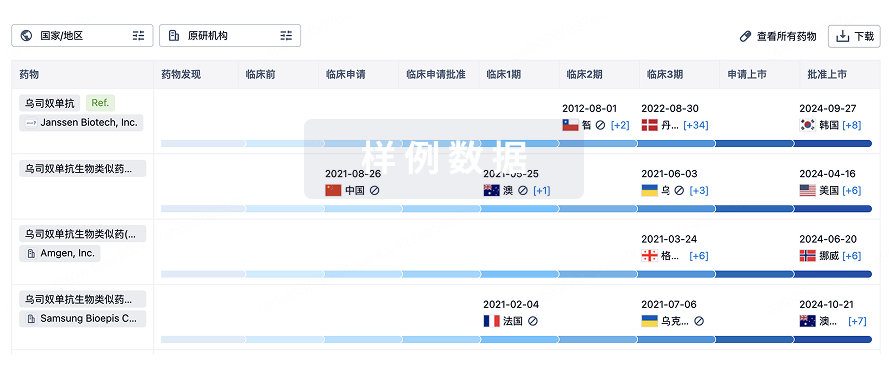
生物类似药
生物类似药在不同国家/地区的竞争态势。请注意临床1/2期并入临床2期,临床2/3期并入临床3期
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或

Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用



