预约演示
更新于:2025-05-07
Affamed Therapeutics (Us), Inc.
更新于:2025-05-07
概览
标签
眼部疾病
其他疾病
神经系统疾病
小分子化药
生物药
Fab片段抗体
疾病领域得分
一眼洞穿机构专注的疾病领域
暂无数据
技术平台
公司药物应用最多的技术
暂无数据
靶点
公司最常开发的靶点
暂无数据
| 排名前五的药物类型 | 数量 |
|---|---|
| 小分子化药 | 2 |
| Fab片段抗体 | 1 |
| 生物类似药 | 1 |
| 生物药 | 1 |
| 排名前五的靶点 | 数量 |
|---|---|
| VEGF-A(血管内皮生长因子A) | 1 |
关联
4
项与 Affamed Therapeutics (Us), Inc. 相关的药物靶点- |
作用机制- |
在研适应症 |
非在研适应症- |
最高研发阶段申请上市 |
首次获批国家/地区- |
首次获批日期- |
靶点- |
作用机制 感觉感受器调节剂 |
非在研适应症- |
最高研发阶段临床3期 |
首次获批国家/地区- |
首次获批日期- |
靶点- |
作用机制- |
在研适应症 |
非在研适应症- |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期- |
100 项与 Affamed Therapeutics (Us), Inc. 相关的临床结果
登录后查看更多信息
0 项与 Affamed Therapeutics (Us), Inc. 相关的专利(医药)
登录后查看更多信息
4
项与 Affamed Therapeutics (Us), Inc. 相关的新闻(医药)2024-04-12
帕金森病(Parkinson's disease,简称PD)帕金森病是仅次于阿尔茨海默病(AD)的第二大神经退行性疾病,于1817年由James Parkinson医生最早描述其症状,并以他的名字命名。作为常见的中老年神经系统退行性疾病,临床表现为震颤、肌强直、姿势平衡障碍等运动症状和睡眠障碍、自主神经功能障碍、认知和精神障碍等非运动症状,中晚期阶段常伴有运动并发症,严重影响患者的生活质量。变,万物随阳气上升而萌芽生长。 “ 据统计,全球范围内约有1000多万名患者饱受帕金森病的困扰。而我国的帕金森病患者人数预计将从2005年的199万人上升到2030年的近500万人,几乎占到全球帕金森病患病人数的一半。”发病机制帕金森的主要病理变化发生在中脑黑质腹侧的致密部,该区含有大量多巴胺神经元,患者致密部神经元大量死亡,有的甚至丧失高达70%的神经元,导致多巴胺的缺乏以及动作、边缘系统等神经回路的故障。二十世纪60年代研究发现,多巴胺前体化合物左旋多巴对帕金森症有治疗效果,直到今天仍是治疗帕金森的主力药物。然而,多巴胺制剂只能缓解帕金森症状,治标不治本,且副作用较大。后续研究发现,大脑黑质的病变与α-突触核蛋白有关,这种蛋白异常积聚,形成纤维蛋白沉淀并最终导致神经细胞死亡。α-突触核蛋白是可溶性蛋白,导致其异常聚集通常伴随着免疫激活、神经炎症、线粒体功能障碍以及溶酶体和内体功能的改变。研究者发现,这些病理过程与基因密切相关。比如,具有PRKN基因突变和主要线粒体功能障碍的患者表现出一种受限的细胞丧失模式,主要局限在黑质纹状体系统中,没有典型散发性帕金森病中所见的广泛病理和非运动特征。相反,具有SNCA或GBA突变,且α-突触核蛋白病理明显的患者在早期表现出非运动特征,包括自主神经功能障碍和痴呆,可能反映了全身和大脑中的广泛病理。这些风险基因不一定与α-突触核蛋白的病理直接相关。目前已经确定七个基因作为单基因病因,其中四个导致晚发性、常染色体显性遗传疾病(即LRRK2、CHCHD2、VPS35和SNCA),三个导致早发性、常染色体隐性遗传疾病(即PARKIN、DJ1和PINK1)。第八个基因GBA的变异是帕金森病的最常见遗传风险因素。未来有望开发出针对专门靶点的治疗药物。主要治疗思路流程除多巴胺制剂之外,儿茶酚-氧位-甲基转移酶抑制剂以及单胺氧化酶B型抑制剂也是帕金森的常用药物。根据《2020年中国帕金森病治疗指南》,对早发型、晚发型帕金森病,根据主要症状的不同选择不同的治疗方案(图1)。图1.患者用药选择流程图参考来源:《2020年中国帕金森病治疗指南》;DAs:多巴胺受体激动剂;MAO-BI:单胺氧化酶B型抑制剂;COMTI:儿茶酚-O-甲级转移酶抑制剂2020年指南继续强调了多学科治疗: ①坚持综合治疗,应对帕金森病的运动症状和非运动症状采取全面综合治疗;②强调多学科治疗模式,药物治疗仍为首选,并结合手术、运动与康复、心理干预等多种治疗手段;同时,提倡在临床条件允许的情况下,组建神经内科、功能神经外科、神经心理、康复乃至社区全科医生等多学科的医生团队;③明确全程管理,目前的治疗仍以改善症状为主,不能阻止病情的发展,治疗不仅立足当前,而且需长期管理,以达到长期获益。常见药物与在研新药根据传统治疗分类和在中国获批的药品,帕金森治疗药物主要有以下几种。复方左旋多巴:恩他卡朋双多巴、多巴丝肼多巴胺受体激动剂(DAs):溴隐亭、培高利特(已撤市)、吡贝地尔、甲磺酸-a-二氢麦角隐亭、普拉克索、罗匹尼罗和罗替高汀儿茶酚-O-甲级转移酶抑制剂(COMTI):托卡朋和恩他卡朋单胺氧化酶B型抑制剂(MAO-BI):雷沙吉兰和司来吉兰抗胆碱药:苯甲托品、苯海索以及苯海拉明去甲肾上腺素前体:屈昔多巴神经保护剂:单唾液酸四己糖神经节苷脂钠此外,还有左旋多巴和卡比多巴,其中卡比多巴需与左旋多巴联用。帕金森领域进展较慢,从2010年至今,美国FDA批准的能用于缓解帕金森症状的新分子实体仅4款:Acadia Pharma 的5-羟色胺2A受体反向激动剂酒石酸匹莫范色林、US WorldMeds的沙非酰胺、协和发酵麒麟的A2A受体拮抗剂伊曲茶碱以及第三代强效COMTI奥匹卡朋。奥匹卡朋已有复星医药引进中国,目前正在进行临床试验。在国内进行开发的帕金森药品见下表。来源:GBI SOURCE新兴技术干细胞衍生药物治疗帕金森成为一条新的探索帕金森治疗的路径。继拜耳BlueRock之后,睿健医药成为国际第二家、国内首家提交帕金森领域多能干细胞衍生药物临床申请的公司。2022年4月28日,国家药监局受理了睿健医药自主研发的帕金森管线人源多巴胺能前体细胞注射液NouvNeu001的IND申请。临床前数据显示,通过睿健医药自研的高效化学小分子诱导功能细胞再生技术,NouvNeu001 移植入体内后可高效转化为成熟多巴胺能神经元,分泌多巴胺递质,并与体内原有神经元形成神经连接,产生综合性的治疗功能,改善帕金森症状。这类前体细胞除了能够分泌多巴胺这类递质之外,还可进一步分泌多种蛋白及小核酸来改善帕金森病灶,为移植到脑部的细胞发挥作用提供良好的“土壤”环境。企业交易活动国内对帕金森病的研究对比国际略显落后,因此一些企业开始引进海外的产品和技术。2018年1月,万邦生化(母公司复星医药)与葡萄牙公司BIAL达成合作,在中国大陆独家销售代理其帕金森病治疗药物奥吡卡朋;2020年10月,日本药企Kissei Pharmaceutical宣布已与中国生物医药公司AffaMed Therapeutics就其帕金森在研新药KDT-3594达成许可协议,Affamed Therapeutics将获得在大中华区(包括中国大陆、香港、澳门和台湾地区)和东南亚六国(包括新加坡,马来西亚,泰国,印尼,越南和菲律宾)开发和商业化KDT-3594(DAs)的独家权利;2021年4月,赛神医药与礼来达合作,获得在大中华区开发和商业化针对帕金森病α-突触核蛋白靶向疗法的权利;2022年3月,上海中泽医药与美国公司 Digestome Therapeutics达成合作,获得后者DGX-001在大中华市场的独家权利。DGX-001是一种全球首创口服治疗药物,可用于治疗精神分裂症的阴性症状和认知障碍,以及帕金森病的非运动症状;2022年3月,翼思生物与德国史达德大药厂(STADA Arzneimittel AG)子公司Brtiannia Pharmaceuticals Ltd签署了一项许可协议,获得在中国大陆、香港和澳门开发和商业其皮下注射阿扑吗啡用于帕金森病的独家权利;2023年5月,畅溪制药与美国公司Acorda Therapeutics达成合作,引进后者INBRIJA(左旋多巴吸入粉雾剂)在大中华地区(中国大陆及港澳台地区)的权利。INBRIJA已在美国和欧盟获批上市,用于正在接受左旋多巴-多巴脱羧酶抑制剂治疗的成年帕金森病患者,其在关闭期(OFF episodes) 的间歇性治疗;2024年1月,斯坦福大学与先声药业达成合作研究协议,将共同推动一项神经系统领域的探索性研究,为帕金森患者研发创新疗法。帕金森发现至今,目前虽有多种可以缓解症状的疗法,但还没有一种能够延缓疾病进展或治愈疾病。未来,开发这一疾病更理想的治疗方法依然是医学领域的重要目标之一。\ | /★联 系 我 们投稿 | 发稿 | 媒体合作▶ sylvia.hua@generalbiologic.com数据库 | 咨询服务 | 资讯追踪▶ 点击左下“阅读原文”完成表单填写点击 阅读原文 立即订阅相关报告
核酸药物
2024-01-05
None
Welcome to this week's Chutes & Ladders, our roundup of hirings, firings and retirings throughout the industry. Please send the good word—or the bad—from your shop to Max Bayer or Gabrielle Masson, and we will feature it here at the end of each week.
Scorpion’s CEO, CMO out the door
Scorpion Therapeutics
It’s a new year and change is afoot at Scorpion Therapeutics, with both former CEO Axel Hoos, M.D., Ph.D., and CMO Michael Streit, M.D., hitting the exits.
Industry veteran Hoos has led the oncology company since July 2021, when he left GSK for Scorpion. At the time, Hoos had taken the reins from interim CEO Adam Friedman, M.D., Ph.D., who will now take back the top title, this time without the temporary designation. Friedman is part of Scorpion’s founding team and most recently served as the biotech’s president of corporate strategy and business development.
The Boston biotech is also replacing Streit, who was the first to fill the company’s chief medical officer position. Streit joined in 2022 from Sanofi, where he had most recently served as vice president and senior project head.
Now, Mark Chao, M.D., Ph.D., will take over as CMO. He joins Scorpion from TenSixteen Bio, where he served as co-founder and CEO.
But the changes don’t stop there. Scorpion has also promoted Erica Jackson, Ph.D., to chief discovery officer. She previously served as the company’s executive vice president of biology. Fierce Biotech
CRISPR hunts for new CMO
CRISPR Therapeutics
CRISPR Therapeutics Chief Medical Officer Phuong Khanh Morrow, M.D., is resigning—a decision that was announced six days after the company secured FDA approval for its Vertex-partnered gene therapy Casgevy.
The biotech has already kicked off the search for a new CMO, a spokesperson told Fierce Biotech.
“CRISPR Therapeutics has a seasoned leadership team with extensive experience managing clinical development for CRISPR’s programs and is well positioned to successfully manage the transition,” a spokesperson said via email.
Morrow, who has been with the company since May 2022, will officially depart Jan. 26. Her resignation was not the result of any disagreement with the gene-editing company, according to CRISPR Tx.
Before joining the Swiss-American biotech, Morrow spent more than 10 years in various leadership roles at Amgen. Fierce Biotech
Long-time Evotec CEO steps down
Evotec
Evotec’s CEO Werner Lanthaler has resigned from his role before his expected end of term, which was set for March 2026, citing personal reasons.
Lanthaler, who has led the German biotech since 2009, will serve as a strategic advisor to Evotec’s board, which has launched a search for a permanent CEO.
Until a successor is chosen, board member Mario Polywka will take on the role of interim CEO. Ploywka had served as Evotec’s chief operating officer from 2006 to 2018. Release
> LianBio CEO Yizhe Wang, Ph.D., is headed for the exits in pursuit of new opportunities. Chief Investment Officer Adam Stone will be the interim chief while the search for a permanent replacement begins. Release
> Ex-Biogen CEO Michel Vounatsos has joined the board of Quris-AI. Former Merck regional president Yossi Ben Amram was also named president of the company. Release
> Vicore Pharma has hired Bertil Lindmark, Ph.D., M.D., to serve as chief medical officer, taking the reins from Rohit Batta. Lindmark joins from Galecto, where he also held the CMO role, and has previous experience as AstraZeneca’s global vice president of clinical development for respiratory and inflammation. Release
> Applied Therapeutics has named Constantine Chinoporos as its new chief operating officer and chief business officer. He’s been a strategic advisor for Apollo Therapeutics since February 2023 after past c-suite roles at other biotechs. Disclosure
> Innate Pharma CEO Mondher Mahjoubi has resigned to pursue a new gig at a larger pharmaceutical company, the European biotech announced. Chairman and co-founder Hervé Brailly will be the interim leader while the search for a replacement gets underway. Release
> SpliceBio will bring on Aniz Girach, M.D., as chief medical officer. Girach was previously CMO of fellow gene editing biotech ProQR until March 2023. Release
> New year, new gig for Nathan Dowden, who’s been promoted at Entrada from chief operating officer to president and COO. He’s been with the company since 2019 after working at Rubius. Release
> Relmada CMO Cedric O’Gorman, M.D., is leaving the company after one year. He previously was the chief medical officer at Alpha Cognition. Release
> After Zevra’s recent acquisition of Acer, Adrian Quartel, M.D., will become the new chief medical officer. He was the CMO of Acer and was the chief medical officer of Adamis before that. Release
> ARIAD Pharmaceuticals founder Harvey Berger, M.D., has been hired as the new CEO of Kojin Therapeutics. Kojin is backed by the likes of Leaps By Bayer and AbbVie’s venture arm. Release
> Included in Radionetics Oncology’s latest financing announcement was news that the biotech has brought on Paul Grayson as CEO. He was most recently president and CEO of fellow oncology company Tentarix Biotherapeutics. Release
> Mineralys Therapeutics has tapped Minji Kim, Ph.D., to serve as chief business officer. Before Mineralys, she held the same role at Affamed Therapeutics and has past experience at Jounce Therapeutics, Curis and Hoffmann-La Roche. Release
> ProMIS Neurosciences is bringing on Neil Warma as interim CEO to replace Gail Farfel, Ph.D. Warma has been a director on the company’s board since 2022. Release
> Brii is bringing aboard former HemoShear CSO Brian Alvin Johns, Ph.D., for the same role. Before HemoShear, he was VP of discovery at Viiv and GSK. Release
> Heike Keilhack, Ph.D., is joining ROME Therapeutics as chief scientific officer. She previously was CSO and oversaw R&D of two now-clinical-stage PARP inhibitors at Ribon Therapeutics. Release
> Diagnostics vet Brian Kim is the new CEO of genomics company Mission Bio, the company announced. He previously was CEO of Fortis Life Science and held other leadership roles at Biosearch Technologies and PerkinElmer. Release
> SV Health Investors has appointed Nikola Trbovic as managing partner and Jamil Beg as partner. The two will help steer the firm’s biotech portfolio, which includes companies like Bicycle and Nimbus. Release
> CymaBay Therapeutics and former Chief Commercial Officer Lewis Stuart mutually agreed that Stuart would leave the company on Dec. 29. CymaBay’s VP Liver Franchise Lead Ben Kozub will assume leadership of commercial organization. Release
> Alaunos Therapeutics has tapped Dale Curtis Hogue, Jr., to serve as director of the immuno-oncology company. Hogue most recently founded Dune Lake Capital and previously worked as a senior analyst at Discovery Capital Management. Release
> Viatris has chosen Philippe Martin to lead the company’s research and development as chief R&D officer. Martin has clocked in past experience at Celgene and Schering-Plough (acquired by Merck). Release
> Noriyuki Kasahara, M.D., Ph.D., is moving from 4DMT’s board to its C-suite, being named chief scientific officer. Kasahara is a brain surgeon who previously worked at UCSF. Release
> Scribe Therapeutics’ next chief scientific officer and head of research and development will be Aarif Khakoo, M.D., who joins the CRISPR genetic medicines company from Calico Life Sciences. Also joining the biotech is Maria Mirotsou, Ph.D., as vice president of discovery biology. Release
> Eterna Therapeutics has picked Sanjeev Luther as its next CEO, as of Jan. 1. He succeeds Matt Angel, Ph.D. Luther has spent the past 30 years at various roles across biopharma, most recently as CEO at Cornerstone Pharmaceuticals. Release
> Henrik Luessen is the new chief business officer for German biotech Eleva, which has a moss-based platform that develops complex proteins for treatment of diseases. Release
> Ashvattha Therapeutics has named Mehdi Paborji, Ph.D., as senior vice president of technical operations to oversee CMC and manufacturing for the biotech’s nanomedicines. Release
> Deka Biosciences has tapped Stanley Frankel, M.D., to serve as senior clinical advisor, interim chief medical officer and scientific advisory board member. The hematologist-oncologist joins Deka with past experience as SVP, global drug development for cell therapy for BMS following the 2019 acquisition of Celgene. Release
> HERVOLUTION Therapeutics has chosen J. Robert Coleman, Ph.D., to lead the biotech as CEO. Before joining HERVOLUTION, Coleman co-founded Codagenix and served as the company’s CEO for 12 years. Release
> ImPact Biotech has appointed clinical radiologist Eyal Morag, M.D., to be the company’s chief medical officer. Morag joins from Microbot Medical, where he was also CMO, and currently serves in scientific and medical advisory roles at Aidoc Medical, Vortex Imaging and HighRad. Release

高管变更上市批准
2024-01-04
RADNOR, Pa., Jan. 04, 2024 (GLOBE NEWSWIRE) -- Mineralys Therapeutics, Inc. (Nasdaq: MLYS), a clinical-stage biopharmaceutical company focused on developing medicines to target hypertension, chronic kidney disease (CKD) and other diseases driven by abnormally elevated aldosterone, today announced the appointment of Minji Kim, Ph.D. as Chief Business Officer. Adam Levy will remain in his role as Chief Financial Officer. “With our expanding operations, we have decided to split the functions of Chief Financial Officer and Chief Business Officer into two roles, and we are excited to have Minji join our team. She brings a solid track record of generating value for multiple companies by identifying and executing strategic opportunities,” stated Jon Congleton, Chief Executive Officer of Mineralys. “We are pleased to start 2024 with our full executive team in place as we execute against our late-stage clinical development of lorundrostat for the treatment of aldosterone dependent conditions such as hypertension and chronic kidney disease.” “With several clinical milestones for lorundrostat expected over the next 12-18 months, I am excited to join Mineralys. Given the current trajectory of the ongoing pivotal clinical development program, lorundrostat has great potential to address unmet needs in patients suffering from diseases driven by abnormally elevated aldosterone,” stated Dr. Kim. Dr. Kim brings more than two decades of experience in business development, strategic leadership, and scientific research. During her career, she has worked with biotech companies in the U.S. and overseas across broad therapeutic and technical areas. Prior to joining Mineralys, she held the role of Chief Business Officer at Affamed Therapeutics and General Manager at Affamed Digital, and brought multiple products into their pipeline. Before Affamed, Dr. Kim was Head of Business Development and Alliance Management at Jounce Therapeutics. She led the execution of pharma partnerships and over $200 million in non-dilutive funding for the company. Prior to Jounce, she held the role of Vice President of Corporate and Business Development at Curis, Inc. During her tenure at Curis, she led strategic transactions that had a fundamental impact on the future sustainability of the company, tripling the value of the company and pivoting its R&D direction. Previously, Dr. Kim served on the Global Oncology Business Development and Licensing group at Hoffmann-La Roche. She began her career as an instructor in neurology at Harvard Medical School. Currently, Dr. Kim is an independent board member of SK Biopharmaceuticals, a global commercial-stage life science company. She obtained her Ph.D. from Seoul National University in South Korea and her MBA from Yale School of Management. About LorundrostatLorundrostat is a proprietary, orally administered, highly selective aldosterone synthase inhibitor being developed for the treatment of uncontrolled and resistant hypertension and CKD. Lorundrostat was designed to reduce aldosterone levels by inhibiting CYP11B2, the enzyme responsible for its production. Lorundrostat has 374-fold selectivity for aldosterone-synthase inhibition versus cortisol-synthase inhibition in vitro, an observed half-life of 10-12 hours and demonstrated approximately a 70% reduction in plasma aldosterone concentration in hypertensive subjects. About Mineralys TherapeuticsMineralys Therapeutics is a clinical-stage biopharmaceutical company focused on developing medicines to target hypertension, CKD and other diseases driven by abnormally elevated aldosterone. Its initial product candidate, lorundrostat, is a proprietary, orally administered, highly selective aldosterone synthase inhibitor that Mineralys Therapeutics is developing for cardiorenal conditions affected by abnormally elevated aldosterone, including hypertension and CKD. Mineralys is based in Radnor, Pennsylvania, and was founded by Catalys Pacific. For more information, please visit https://mineralystx.com. Follow Mineralys on LinkedIn and Twitter. Forward-Looking StatementsMineralys Therapeutics cautions you that statements contained in this press release regarding matters that are not historical facts are forward-looking statements. The forward-looking statements are based on our current beliefs and expectations and include, but are not limited to, statements regarding: the potential therapeutic benefits of lorundrostat; the Company’s expectation that aldosterone synthase inhibitors with an SGLT2 inhibitor may provide additive clinical benefits to patients; the Company’s expectation that the Advance-HTN and the planned Phase 3 clinical trial of lorundrostat may serve as pivotal trials in any submission of a new drug application (NDA) to the United States Food and Drug Administration (FDA); the Company’s ability to evaluate lorundrostat as a potential treatment for CKD or uncontrolled hypertension; the planned future clinical development of lorundrostat and the timing thereof; and the expected timing of commencement and enrollment of patients in clinical trials and topline results from clinical trials. Actual results may differ from those set forth in this press release due to the risks and uncertainties inherent in our business, including, without limitation: our future performance is dependent entirely on the success of lorundrostat; potential delays in the commencement, enrollment and completion of clinical trials and nonclinical studies; later developments with the FDA may be inconsistent with the feedback from the completed end of Phase 2 meeting, including whether the proposed pivotal program will support registration of lorundrostat which is a review issue with the FDA upon submission of an NDA; our dependence on third parties in connection with manufacturing, research and clinical and nonclinical testing; unexpected adverse side effects or inadequate efficacy of lorundrostat that may limit its development, regulatory approval and/or commercialization; unfavorable results from clinical trials and nonclinical studies; results of prior clinical trials and studies of lorundrostat are not necessarily predictive of future results; our ability to maintain undisrupted business operations due to any pandemic or future public health concerns; regulatory developments in the United States and foreign countries; our reliance on our exclusive license with Mitsubishi Tanabe Pharma to provide us with intellectual property rights to develop and commercialize lorundrostat; and other risks described in our filings with the Securities and Exchange Commission (SEC), including under the heading “Risk Factors” in our annual report on Form 10-K, and any subsequent filings with the SEC. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof, and we undertake no obligation to update such statements to reflect events that occur or circumstances that exist after the date hereof. All forward-looking statements are qualified in their entirety by this cautionary statement, which is made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Contact:Investor Relationsinvestorrelations@mineralystx.com Media RelationsTom WeibleElixir Health Public RelationsPhone: (1) 515-707-9678Email: tweible@elixirhealthpr.com
临床3期高管变更
100 项与 Affamed Therapeutics (Us), Inc. 相关的药物交易
登录后查看更多信息
100 项与 Affamed Therapeutics (Us), Inc. 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2025年08月26日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
临床1期
1
1
临床2期
临床3期
1
1
申请上市
登录后查看更多信息
当前项目
| 药物(靶点) | 适应症 | 全球最高研发状态 |
|---|---|---|
IOLs (AffaMed Therapeutics) | 白内障 更多 | 申请上市 |
Aloradine | 恐怖症 更多 | 临床3期 |
AM008 | 青光眼 更多 | 临床2期 |
雷珠单抗生物类似药(Samsung Bioepis) ( VEGF-A ) | 湿性年龄相关性黄斑变性 更多 | 临床1期 |
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
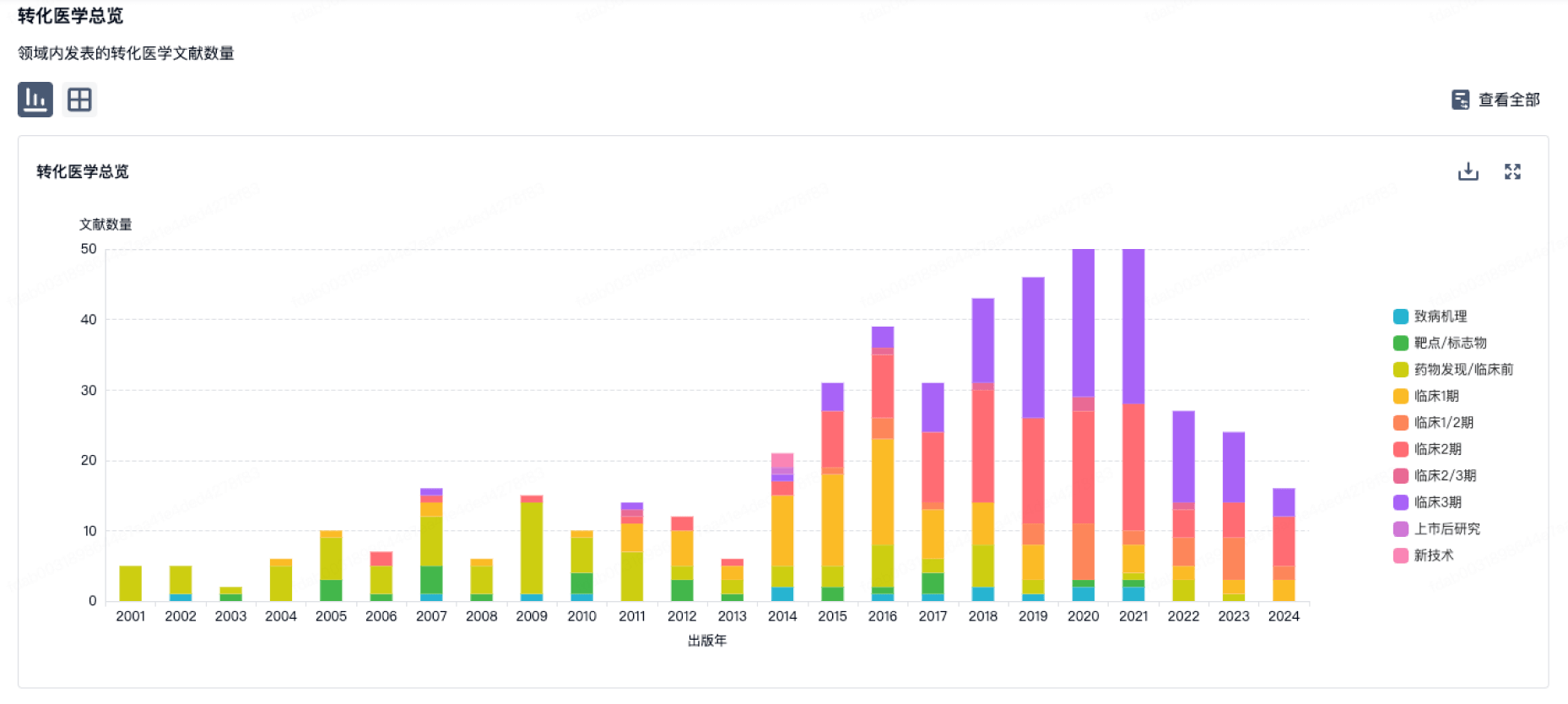
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或
投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或
融资
发掘融资趋势以验证和推进您的投资机会。
登录
或
Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

