MHLW批准Solasia公司有机砷药物Darvias上市
2022-06-21
孤儿药临床结果
日前,日本Solasia Pharma公司宣布,日本厚生劳动省(MHLW)已批准有机砷药物Darvias(曾用名SP-02)上市,用于治疗复发或难治性外周T细胞淋巴瘤(PTCL)。新闻稿指出,这是这款创新疗法在全球范围内首次获批。
恶性淋巴瘤分为霍奇金淋巴瘤(HL)和非霍奇金淋巴瘤(NHL),非霍奇金淋巴瘤又分为B细胞淋巴瘤和T细胞淋巴瘤两大类。PTCL是T细胞淋巴瘤的一种亚型,相对其他亚型发病率较高,尚未确立PTCL的标准治疗,由于PTCL的预后较差且治疗困难,因此存在高度未满足医疗需求。
Darvias是一款具有抗癌活性的有机砷化合物。提到砷,大家可能都想到著名的毒药砒霜。有机砷曾被广泛应用于药物和农药,它可以用于治疗非洲锥虫病和阿米巴痢疾。Darvias的潜在作用机理包括扰乱线粒体功能,提高活性氧的产生,以及调节细胞内信号通路。除了PTCL,它的其它潜在适应症包括其它血液学癌症和实体瘤。它已经在美国和欧盟获得孤儿药资格。
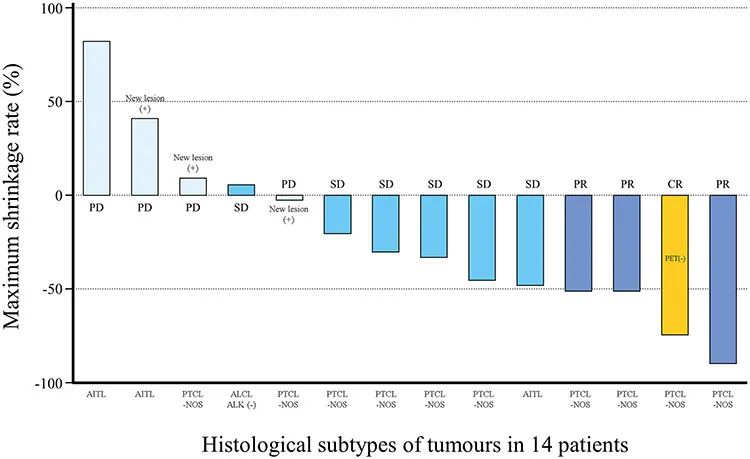
发表在Japanese Journal of Clinical Oncology的1期临床试验结果显示,在14名能够被评估的患者中,1名患者获得完全缓解,3名患者获得部分缓解,6名患者疾病稳定。
Preview
来源: 新浪医药新闻
接受Darvias治疗的患者肿瘤大小在1期临床试验中的变化(图片来源:参考资料[2])
这一批准是基于在多个亚洲国家进行的2期临床试验的结果。
参考资料:
[1] Solasia Pharma: DARVIAS® Injection 135mg (Generic Name: DARINAPARSIN /Development Code: SP-02) Approved in Japan. Retrieved June 20, 2022, from https://www.businesswire.com/news/home/20220620005030/en/
[2] Ogura et al., (2021). Phase I studies of darinaparsin in patients with relapsed or refractory peripheral T-cell lymphoma: a pooled analysis of two phase I studies conducted in Japan and Korea, Jpn J Clin Oncol, DOI: 10.1093/jjco/hyaa177
内容来源于网络,如有侵权,请联系删除。<br>
靶点
-热门报告
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。



