Novartis prepares for FDA submission for its chronic hives treatment
2023-08-09
临床3期临床结果

Preview
来源: Pharmaceutical Technology
Phalguni Deswal
@Phalguni_GD
Preview
来源: Pharmaceutical Technology
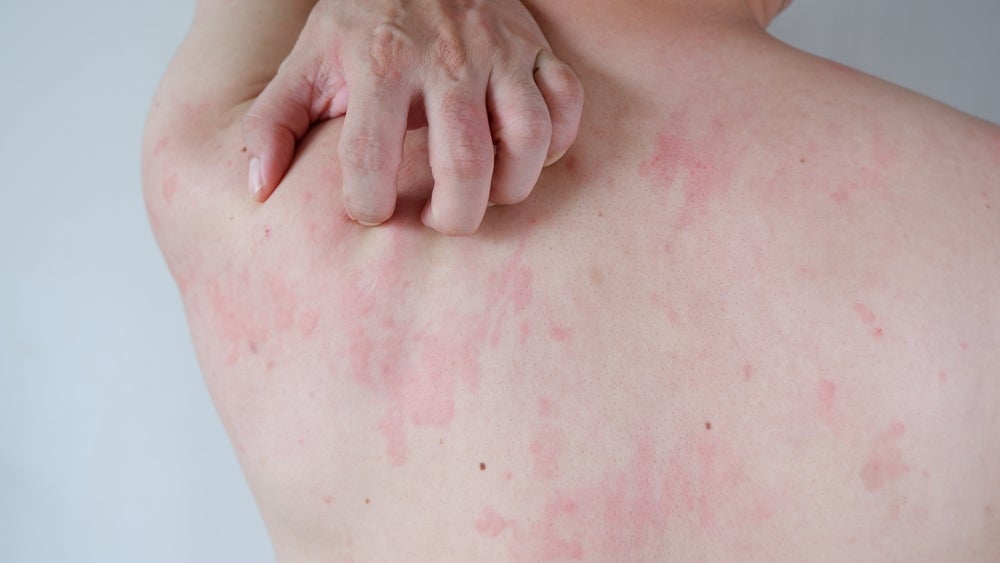
Remibrutinib started showing improvement in disease activity after 2 weeks of therapy. Image Credit: wisely / Shutterstock.
Novartis has announced that the two Phase III trials for remibrutinib have demonstrated clinical efficacy and met their primary endpoints in patients with chronic spontaneous urticaria.
Chronic spontaneous urticaria, also known as chronic hives, is a skin condition of unknown cause that causes red, raised, and itchy hives that are occasionally painful. These hives last for six weeks or more. As per Allergy UK, chronic spontaneous urticaria is not caused by allergens but by an immune response, and is managed symptomatically with anti-histamines, antipruritic, and anti-inflammatories.
Recommended Reports

Preview
来源: Pharmaceutical Technology
ReportsHospital Acquired Methicillin-Resistant Staphylococcus aureus (HA-MRSA) Infections Drugs in Devel... GlobalData

Preview
来源: Pharmaceutical Technology
ReportsLOA and PTSR Model - Zinpentraxin Alfa in Post-Essential Thrombocythemia Myelofibrosis (Post-ET MF) GlobalData
View allCompanies IntelligenceNovartis AGSanofiAmgen IncAllakos IncCSU, INC.View all
Remibrutinib is a selective Bruton’s tyrosine kinase (BTK) inhibitor. It acts by stopping histamine release by blocking the BTK cascade. The release of histamine is responsible for chronic spontaneous urticaria symptoms such as itching, hives, and swelling.
Remibrutinib started showing improvement in disease activity after two weeks of therapy. It also reported a favourable safety profile in multiple conditions apart from chronic spontaneous urticaria, including multiple sclerosis, hidradenitis suppurativa, and food allergy.
“These positive top-line results from the Phase III REMIX studies confirm that remibrutinib, a highly selective BTK inhibitor, has the potential to be a first-in-class, oral treatment for people living with CSU whose symptoms are refractory despite the use of antihistamines,” said Shreeram Aradhye, chief medical officer of Novartis in a press release.
Additional one-year follow-up data is planned for presentation at scientific conferences in 2024. Furthermore, Novartis is planning to apply for regulatory submissions in 2024.
Sanofi’s monoclonal antibody, Dupixent (dupilumab) showed a 63% decline in the severity of itch in Phase III trials within the paediatric population (aged six years or older). Other drugs in development for chronic spontaneous urticaria include Amgen’s Tezepelumab, Allakos’ lirentelimab, and Celldex’s barzovolimab.
更多内容,请访问原始网站
文中所述内容并不反映新药情报库及其所属公司任何意见及观点,如有版权侵扰或错误之处,请及时联系我们,我们会在24小时内配合处理。
靶点
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。





