预约演示
更新于:2025-09-06
Olpasiran
更新于:2025-09-06
概要
基本信息
非在研机构- |
最高研发阶段临床3期 |
首次获批日期- |
最高研发阶段(中国)临床3期 |
特殊审评突破性疗法 (中国) |
登录后查看时间轴
结构/序列
使用我们的RNA技术数据为新药研发加速。
登录
或
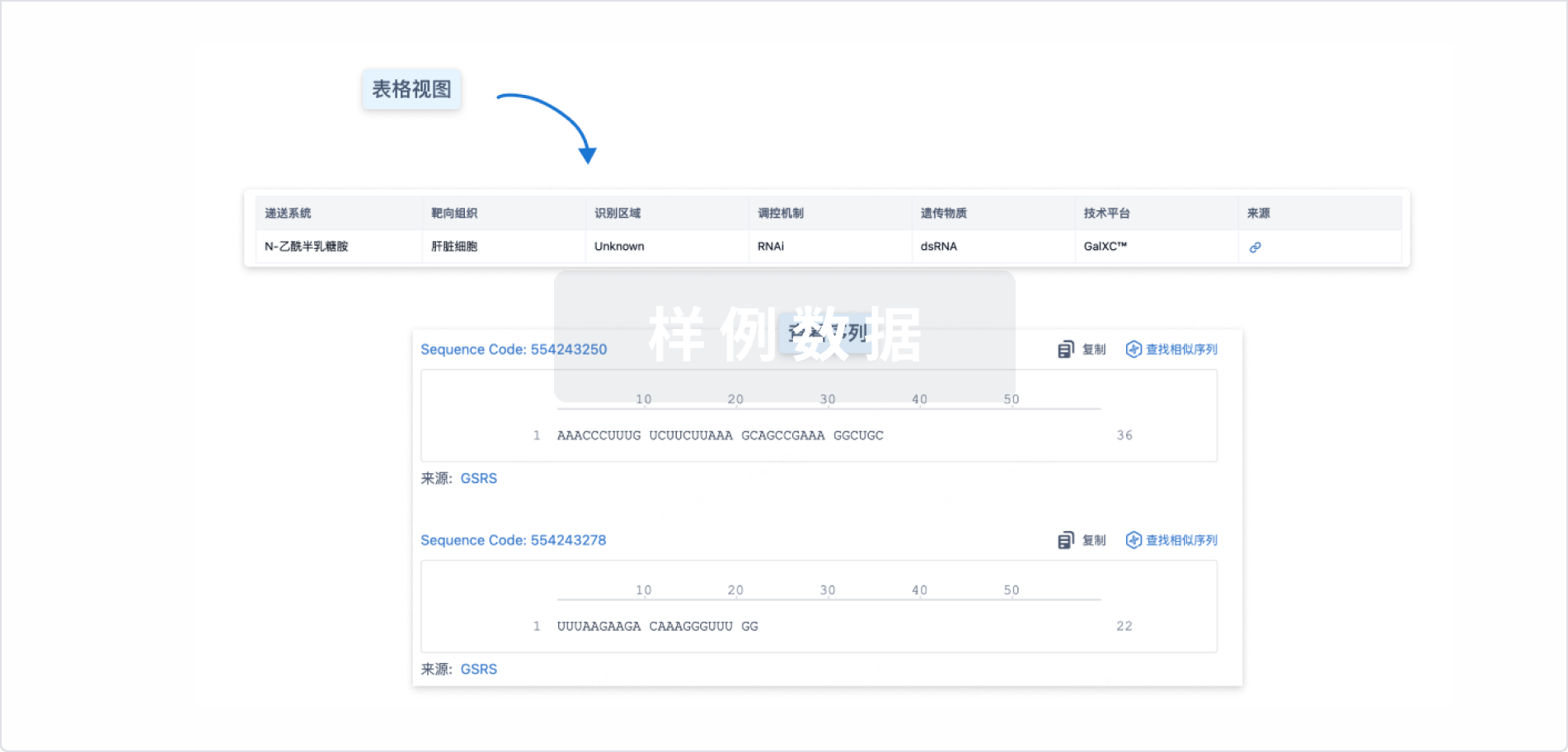
Sequence Code 31610133

来源: *****
Sequence Code 31611198

来源: *****
关联
10
项与 Olpasiran 相关的临床试验NCT07136012
A Double-blind, Randomized, Placebo-controlled, Multicenter Study Assessing Olpasiran Use to Prevent First Major Cardiovascular Events in Participants With Elevated Lipoprotein(a)
The primary objective is to evaluate the effect of olpasiran, compared to placebo, on the risk for coronary heart disease death (CHD death), myocardial infarction, or urgent coronary revascularization in participants at risk for a first major cardiovascular event with elevated lipoprotein(a) (Lp[a]).
开始日期2025-09-19 |
申办/合作机构 |
NCT06411860
A Randomized, Placebo- and Positive-controlled, Crossover Study to Assess the Effect of Olpasiran (AMG 890) on QT/QTc Intervals in Healthy Subjects
The primary objective of the study is:
• To assess the effects of a single therapeutic and supratherapeutic dose of olpasiran on the placebo-corrected change from baseline in QT corrected for heart rate (ΔΔQT)/QTc interval in healthy participants.
• To assess the effects of a single therapeutic and supratherapeutic dose of olpasiran on the placebo-corrected change from baseline in QT corrected for heart rate (ΔΔQT)/QTc interval in healthy participants.
开始日期2024-10-10 |
申办/合作机构 |
CTIS2022-501608-85-00
A Double-blind, Randomized, Placebo-controlled, Multicenter Study Assessing the Impact of Olpasiran on Major Cardiovascular Events in Patients with Atherosclerotic Cardiovascular Disease and Elevated Lipoprotein (a) - 20180244
开始日期2023-05-12 |
申办/合作机构 |
100 项与 Olpasiran 相关的临床结果
登录后查看更多信息
100 项与 Olpasiran 相关的转化医学
登录后查看更多信息
100 项与 Olpasiran 相关的专利(医药)
登录后查看更多信息
34
项与 Olpasiran 相关的文献(医药)2025-06-01·DIABETES OBESITY & METABOLISM
Comprehensive evaluation of siRNA therapeutics on Lp(a): A network meta‐analysis
Review
作者: Hu, Jiaqiang ; Wang, Xingjin ; Qin, Xiaoli ; Liu, Song ; Zhao, Chen
Abstract:
Aims:
To evaluate the efficacy and safety of siRNA drugs that lower Lp(a) in patients with dyslipidaemia.
Materials and Methods:
A network meta‐analysis and systematic review were conducted to compare siRNA drugs targeting Lp(a), based on relevant randomized controlled trials (RCTs). A comprehensive search was performed in PubMed, Embase, Web of Science and the Cochrane Library (up to October 24, 2024). RCTs with an intervention duration of at least 12 weeks were included. Eligible studies compared siRNA drugs that reduce Lp(a), including both Lp(a)‐targeted and non‐targeted agents, with placebo or other siRNA drugs that reduce Lp(a). The primary outcomes were the percentage reduction and absolute reduction in Lp(a), percentage reduction in low‐density lipoprotein cholesterol (LDL‐C), percentage reduction in apolipoprotein B (apo(B)), adverse events and serious adverse events, including injection‐site reactions. The risk of bias was assessed using the Cochrane Risk of Bias Tool (ROB2), and a random‐effects network meta‐analysis was performed using the frequentist approach. Confidence in effect estimates was evaluated using the Confidence In Network Meta‐Analysis (CINeMA) framework.
Results:
A total of 14 trials involving 5646 participants were included. Lp(a)‐targeted siRNA agents, particularly Olpasiran, demonstrated strong efficacy in significantly reducing Lp(a) levels, with the greatest percentage reduction in Lp(a) (mean difference [MD]: −92.06%; 95% CI: −102.43% to −81.69%; P‐score: 0.98). Olpasiran also showed the greatest absolute reduction in Lp(a) (MD: −250.70 nmol/L; 95% confidence interval [CI]: −279.89 to −221.50; P‐score: 0.99). Certain non‐Lp(a)‐targeted siRNA agents, such as inclisiran and zodasiran, also showed modest reductions in Lp(a) levels, reducing Lp(a) by approximately 15%. Lp(a)‐targeted siRNA agents reduced LDL‐C by more than 20% and decreased apo(B) by approximately 15%. In terms of safety, most drugs exhibited favourable safety profiles with no significant differences compared to placebo. However, zerlasiran raised concerns regarding injection‐site reactions and other adverse events when compared to placebo.
Conclusions:
Lp(a)‐targeted siRNA agents have shown robust effectiveness in substantially reducing Lp(a) levels, including both percentage and absolute reductions, with moderate improvements in LDL‐C and apo(B) concentrations. Non‐Lp(a)‐targeted siRNA agents also demonstrate modest reductions in Lp(a) levels. The safety profile is generally favourable, but zerlasiran and inclisiran may increase the incidence of injection‐site reactions.
2025-05-01·JAMA Cardiology
Olpasiran, Oxidized Phospholipids, and Systemic Inflammatory Biomarkers
Article
作者: Baum, Seth J. ; Murphy, Sabina A. ; Wang, Jingying ; Gaudet, Daniel ; Rosenson, Robert S. ; Kassahun, Helina ; Sabatine, Marc S. ; Stout, Elmer ; López, J. Antonio G. ; Lepor, Norman E. ; Wilmanski, Tomaz ; Wu, You ; Park, Jeong-Gun ; O’Donoghue, Michelle L. ; Knusel, Beat ; Wang, Huei
Importance:
Lipoprotein(a) (Lp[a]) is thought to be the major carrier of oxidized phospholipids (OxPL). OxPL are believed to be a potent driver of inflammation and atherosclerosis. Olpasiran, a small interfering RNA, blocks Lp(a) production by inducing degradation of apolipoprotein(a) messenger RNA. Olpasiran’s effects on OxPL and systemic markers of inflammation are not well described.
Objective:
To assess the effects of olpasiran on OxPL, high-sensitivity interleukin 6 (hs-IL-6), and hs-C-reactive protein (hs-CRP) in the OCEAN(a)-DOSE randomized clinical trial.
Design, Setting, and Participants:
OCEAN(a)-DOSE was an international, multicenter, placebo-controlled, phase 2, dose-finding randomized clinical trial conducted between July 2020 and November 2022. A total of 281 patients with atherosclerotic cardiovascular disease and Lp(a) levels greater than 150 nmol/L were included.
Intervention:
Participants were randomized to receive 1 of 4 active subcutaneous doses of olpasiran vs placebo: (1) 10 mg, administered every 12 weeks (Q12W); (2) 75 mg, Q12W; (3) 225 mg, Q12W; or (4) 225 mg, administered every 24 weeks (Q24W). OxPL on apolipoprotein B (OxPL-apoB), hs-CRP, and hs-IL-6 were assessed at baseline, week 36, and week 48 in 272 patients.
Main Outcomes and Measures:
The primary outcome was placebo-adjusted change in OxPL-apoB from baseline to week 36.
Results:
Among 272 participants, median (IQR) age was 62 years (56-69), and 86 participants (31.6%) were female. Baseline median (IQR) Lp(a) concentration was 260.3 nmol/L (198.1-352.4) and median (IQR) OxPL-apoB concentration was 26.5 nmol/L (19.7-33.9). The placebo-adjusted mean percentage change in OxPL-apoB from baseline to week 36 was −51.6% (95% CI, −64.9% to −38.2%) for the 10-mg Q12W dose, −89.7% (95% CI, −103.0% to −76.4%) for the 75-mg Q12W dose, −92.3% (95% CI, −105.6% to −78.9%) for the 225-mg Q12W dose, and −93.7% (95% CI, −107.1% to −80.3%) for the Q24W dose (P < .001 for all). These effects were maintained to week 48 (−50.8%, −100.2%, −104.7%, and −85.8%, respectively; P < .001 for all). There was a strong correlation between percentage reduction in Lp(a) and OxPL-apoB for patients treated with olpasiran (r = 0.79; P < .001). Olpasiran did not significantly impact hs-CRP or hs-IL-6 compared with placebo to weeks 36 or 48 (P > .05).
Conclusion and Relevance:
In the OCEAN(a)-DOSE multicenter randomized clinical trial, olpasiran led to a significant and sustained reduction in OxPL-apoB but no significant effects on hs-CRP or hs-IL-6.
2024-12-01·Current Atherosclerosis Reports
Lp(a): A Rapidly Evolving Therapeutic Landscape
Review
作者: Thanassoulis, George ; Anchouche, Khalil
PURPOSE OF REVIEW:
Elevated lipoprotein(a) (Lp[a]) is a genetically determined cardiovascular risk factor, causally linked to both atherosclerotic coronary artery disease and aortic stenosis. Elevated Lp(a) is widely prevalent, and several cardiovascular societies now recommend performing Lp(a) screening at least once in all adults. However, there are currently no approved drugs aimed specifically at lowering Lp(a). In this review, we describe several promising Lp(a)-lowering therapies in the drug development pipeline and outline what role these may have in future clinical practice.
RECENT FINDINGS:
Pelacarsen and olpasiran are two novel RNA-based injectable therapies which are being studied in ongoing phase 3 clinical trials, with the earliest of these to be concluded in 2025. These drugs act by degrading transcribed LPA mRNA, which would normally yield the apolipoprotein(a) constituent of Lp(a). Other candidate drugs, such as Lepodisiran, Zerlasiran, and Muvalaplin, are also in early-stage development. While there are presently no Lp(a)-lowering drugs available for routine clinical use, several promising candidates are currently under investigation. If these prove to be effective in randomized clinical trials, they will expand the cardiovascular care armamentarium and will allow clinicians to treat a presently unmitigated cardiovascular risk factor.
139
项与 Olpasiran 相关的新闻(医药)2025-09-04
·研发客
Citeline首席分析师周淑华发言
圆桌现场
研发客主编毛冬蕾与张江全互动
张金晶 威斯津生物合伙人商务发展总监
李安康 腾盛博药CFO
尹思远 传信生物首席运营官
王松立 星核迪赛研发总监
陈进 维亚臻生物临床药理&转化医学副总裁
高璐 圆因生物CEO
张震 徕博科亚太商务开发高级总监
Simon Yu 徕博科中国中心实验室运营总监
研讨会现场图片分享
舶望制药近日与诺华制药就4款小核酸药物达成1.6亿美元预付款的消息,再次证明小核酸药物成为备受关注的热门赛道。全球众多企业布局,中国一批“黑马”公司正凭借独特的技术平台和差异化的管线布局悄然崭露头角。在由研发客与徕博科联合举办的“中国分子,世界引擎”研讨会上,该领域的发展前景成为焦点。
拓展阅读
舶望再度授权诺华siRNA药物,总交易价值达52亿美元!
李勇:在心血管疾病领域前景广阔
复旦大学华山医院心脏科李勇教授认为,小核酸药物在心血管疾病防治领域正展现出较大潜力。英克司兰(Inclisiran)的成功获批,验证了这一领域的重要性。
小核酸药物在递送系统方面取得了进展。除了目前已经广泛应用的肝细胞递送系统外,通过优化递送载体,科学家已实现了对卵巢细胞、肾脏细胞及脑细胞的有效靶向递送。李勇教授认为,针对脑细胞的靶向递送技术有望在短期取得突破,这为小核酸药物在心血管神经系统疾病的应用提供了可能。
复旦大学华山医院心脏科 李勇教授
心血管疾病如高血压、血脂异常和糖尿病等,多为慢性病,不仅在中国,在全球也有数以亿计的大量患者,而且需要长期维持治疗。李勇教授指出:“糖尿病患者中45%的死亡与心血管疾病相关,而非高血糖本身。慢性肾脏病患者进入透析后,无论在是否已经接受透析治疗,心血管事件也是其首要死因,占比超40%。近10年来越来越受关注的脂肪肝,已经超越消化系统,与糖尿病及慢性炎症密切关联,也会导致心血管疾病风险增高。因此,维护心血管健康是内科疾病防控预防和管理的关键策略。”
“心血管事件链”概念于1991年由美国医学科学院院长魏德照Victor Dzao提出,强调从危险因素发展为冠心病、心衰的连续过程中的全程疾病管理及危险因素控制。李勇教授解释,高血压、糖尿病和血脂异常是三大主要危险因素,它们通过激活体内RAAS、炎症、能量/线粒体代谢等某些系统,促使动脉粥样硬化斑块形成和发展、以及心肌、肾脏、血管的纤维化加速进展。
关于高血压,他表示:“高血压不仅造成物理损伤,还会激活某些系统,引起左心室肥厚和舒张功能障碍。中国高血压患者众多,每年约260万人因高血压而此死亡,我国的高血压人口数和死亡人数均居全球之首。目前美国诊断标准为130/80 mmHg,而中国仍采用140/90 mmHg,这在一定程度上影响了防控效果。”
在血脂方面,他说:“LDL-C是动脉粥样硬化的主要致病性危险因素,根据我国著名心血管流行病学家北京安贞医院赵冬教授团队15年的队列研究数据,其升高对心肌梗死风险的贡献达77%。大量随机对照临床试验证实,显著降低LDL-C水平不仅能是防治心血管病的关键,还能降低全因死亡,延长寿命。”对于糖尿病与脂肪肝,他强调这两类患者的心血管风险增加,脂肪肝患者尤其易合并心血管疾病,导致临床结局恶化,建议心血管医生应更多关注这类人群的心血管健康。
在血脂管理方面,他特别提到英克司兰(Inclisiran):“作为一种靶向PCSK9的小干扰RNA药物,英克司兰通过阻断PCSK9与LDL受体的结合,增加肝细胞表面LDL受体的数量,提升肝细胞清除LDL-C的能力,从而降低血中的LDL-C水平。其独特之处在于,一次注射可维持长达6个月的疗效,大大提高了患者的治疗依从性。临床试验表明,英克司兰单药治疗或与他汀联用,均可降低LDL-C幅度高达水平60%,且安全性良好。”
他还介绍了其他小核酸药物的研究进展:“除了英克司兰外,针对甘油三酯(TG)和脂蛋白(a) [Lp(a)]的小核酸药物也展现出广阔的应用前景。例如,针对ApoC-III的小核酸药物Plozasiran、针对ANGPLT-3的Zodasiran可降低TG水平,并减少急性胰腺炎的发作风险;除了Plecarsen外、多个针对Lp(a)的小核酸药物如Olpasiran、Lepodisiran则可降低其水平达90%以上,有望减少心血管事件的发生。”
在强化降压与降脂治疗的临床意义方面,李勇教授表示:“近年来,多项大规模临床试验表明,将血压控制在更低水平(如收缩压<120 mmHg)可降低心血管事件和死亡率。然而,中国的高血压管理目标值仍为140/90 mmHg,这在一定程度上限制了降压治疗的获益。因此,推动高血压管理目标的下调,对于改善中国心血管疾病防治现状具有重要意义。”小核酸药物在降压领域也有明显的进展,Zilebesiran的II期临床试验显示,每6月一次皮下注射,可获得令人相当满意的降压效果,针对高血压患者的心血管结局随机对照临床试验(CVOT)ZENITH研究已经启动。
他还强调了早期降脂治疗的重要性:“LDL-C的累积暴露时间与动脉粥样硬化的发展密切相关。因此,越早启动降脂治疗并维持较低水平,对心血管的保护作用越大。小核酸药物的出现,为实现长期、稳定的降脂治疗提供了新的手段。”
最近,Lancet发表了小核酸药物用于治疗脂肪肝的II期临床试验结果,ION224-CS2试验表明,采用肝活检来评估,寡核苷酸制剂抑制DGAT-2,能够有效逆转脂肪肝,安全性良好。
展望未来,李勇教授认为小核酸药物在一级预防和多靶点治疗方面具有广阔前景。然而,李勇教授也指出了小核酸药物面临的挑战:例如,如何进一步提高递送效率、降低脱靶效应、优化给药方案等。此外,加强医生和患者对小核酸药物的认知也是推动其应用的关键。
最后,他总结道:“小核酸药物为心血管疾病防治带来了新希望。通过不断优化技术、拓展适应症和加强临床应用研究,相信小核酸药物将在未来心血管疾病防治中发挥越来越重要的作用。”
周淑华:全球核酸药物研发持续活跃
Citeline首席分析师周淑华博士介绍了2025年全球核酸药物研发持续活跃的态势。
Citeline首席分析师 周淑华博士
截至2025年,全球处于活跃开发阶段的药物总数达23,875种,较上一年度增长4.6%,新增药物1,050种,其中4,546种为新加入研发管线的药物。新增药物中,抗癌药占比38.8%,神经系统疾病药物占13.8%。各临床阶段药物数量均增长,Ⅰ期增长6.8%,Ⅱ期增长6.3%,Ⅲ期增长8.8%,临床前研究阶段增长1.8%(低于2024年的5.5%),显示全球研发活跃但临床前研究增速放缓。
核酸药物研发管线中,肿瘤学仍是最大治疗领域,2025年有9,746种药物在研,增长3.7%,占所有在研药物的39.7%(略低于2024年的40.1%)。神经系统疾病药物研发增长率低于平均水平(2024-2025年仅增长4.2%)。顶尖企业中,辉瑞重新夺回研发管线规模榜首,罗氏和诺华紧随其后。前十家企业贡献5.4%的在研药物,前25家贡献10.0%,剩余19.4%来自仅有一两种药物在研的公司。
疾病聚焦方面,乳腺癌仍是研发重点,管线规模增长9.5%,领先于非小细胞肺癌(NSCLC,增长10.0%)。非癌症领域中,新冠病毒退出前十,阿尔茨海默病仍是唯一进入前十的非肿瘤疾病。肥胖治疗药物研发增长43.3%(受Wegovy和Mounjaro等药物影响)。热门药物蛋白靶点中,免疫疗法(I-O)相关靶点主导,CD3e连续第二年位居榜首,EGFR取代Her-2成为第二位热门靶点(应用于多种癌症治疗)。
全球及中国研发合作趋势显示,全球范围内16%的在研药物处于合作研发状态(抗癌药物领域16%,神经系统疾病药物16%,抗感染药物14%,消化/代谢系统疾病药物17%)。2017-2025年,中国合作交易数量和总金额2025年分别达16.4%和33.7%,但首付款比例普遍低于全球水平。
“未来,全球核酸药物研发将持续活跃,中国企业增长势头强劲。中国企业需提升创新效率、加强全球市场洞察、完善内部管理并构建健全的资本体系,以实现可持续发展。”周淑华说。
张江全:小核酸药物非临床安全评价要点
徕博科中国区早期研发负责人张江全剖析了小核酸药物非临床安全评价要点。
在结构与修饰上,siRNA药物可进行多种修饰,这会影响其药代动力学,免疫原性特性和毒性表现,介绍了非临床监管、药代动力学以及免疫原性三个方面的内容。
徕博科中国区早期研发负责人 张江全
小核酸药物的非临床监管主要遵循 ICH M3(R2)原则,标准遗传毒组合试验,一般毒性、DART、致癌性研究使用两种种属,同时需考虑ICH S6 指南部分终点。考虑到siRNA的临床给药周期,重复给药毒性研究周期较长,啮齿类动物 6 个月、非人灵长类 9 个月,对此,多国监管机构有相关原则。
在药代动力学方面,siRNA血浆半衰期短,快速被肝脏摄取,导致PK/PD分离现象。siRNA主要分布于肝脏且在肝组织中半衰期较长,肝脏药代动力学和RNA诱导沉默复合体(RISC)药代动力学是决定药效动力学及其持续时间的关键因素。免疫原性的潜在来源包括有序列依赖性免疫激活、递送系统影响等。
对于动物研究的毒性特征,包括免疫原性相关毒性,通过非 Toll 样受体途径触发促炎细胞因子释放;杂交依赖性毒性有靶点效应、脱靶效应、RNAi 机制饱和等;递送载体毒性方面,各种载体存在各自不同的毒性特征。
此外,张江全还分享了两个案例。一是已报道的SLN360案例,GalNAc结合的siRNA,靶向脂蛋白a mRNA,用于心血管疾病。研究通过GalNAc- 去唾液酸糖蛋白受体(ASGPR)结合实现肝脏特异性递送,旨在降低PLA,进行体外特异性和效力研究、体内生物分布研究(大鼠/食蟹猴)以及29天GLP毒性研究。结果显示,大鼠在不同剂量下,高剂量雌性大鼠体重略有下降,其他方面如食物摄入等无显著变化,观察到的非不良反应在停药8周后可恢复;食蟹猴在不同剂量下,体重等一般观察无SLN360相关影响,无补体激活或细胞因子释放,预期效果良好,非不良反应停药后也可恢复。
案例二是AAA(GalNAc结合的siRNA,用于非酒精性脂肪性肝炎(NASH)),研究每月1次皮下注射给大鼠和食蟹猴,持续12周,评估毒性并确定毒代动力学,观察停药12周后的恢复或持续情况。结果表明,系统暴露量与剂量成正比或高于剂量比,无药物蓄积和性别差异,临床观察未观察到AAA相关影响。临床病理主要为肝脏相关指标改变,镜检观察可见肝脏、淋巴结、胸腺、脾脏、肾脏、注射部位等出现色素沉着、空泡化、淋巴细胞减少等改变。所有病理发现均为轻度,认为是non-adverse,因此两个种属的NOAEL均为高剂量。
与会者表示这些全面的要点和案例分享,为小核酸药物非临床安全评价领域的研究提供了极具价值的参考。
徐中南:正大天晴进军小核酸药物领域
正大天晴药业集团的高级临床总监徐中南分享了正大天晴在小核酸药物开发中的技术演进路径,尤其聚焦TQA3038这一创新药的临床开发策略。
正大天晴药业集团的高级临床总监 徐中南
徐中南分析了慢乙肝治疗领域的严峻现状:中国慢乙肝患者诊断率仅22%,治疗率17%,市场需求迫切。尽管已有NUC和IFN等抗病毒药物,但抗病毒时间长、临床治愈率低的问题依旧突出,因此,针对HBV复制环节开发新药成为研究热点。
在介绍TQA3038时,徐中南阐述了其作为GalNAc偶联修饰siRNA药物的独特机制:“通过皮下注射特异性靶向HBV ORF-X区,有效抑制HBV复制。GalNAc配体与肝脏细胞的高亲和力和迅速内化能力,确保了siRNA的高效作用。”
临床试验方面,TQA3038一期健康受试者SAD研究共设置了6个剂量组,采用递增剂量设计确保安全。TQA3038给药后快速吸收,血浆浓度约4小时达峰,半衰期8.7至9.34小时,24小时内药物基本消除。6个剂量组安全性良好,未发生严重不良事件。需要重点关注ECG的改变情况。
TQA3038与同靶点药物相比的优势:同剂量下,TQA3038体内抗HBV活性优于Vir-2218,包括HBsAg、HBeAg、HBV RNA的下降比例;与ETV单药组相比,TQA3038和ETV联合给药组对小鼠血浆中HBV DNA的降低更明显。
在介绍同靶点药物联合干扰素治疗方案时,他分享了VIR-2218与PEG-IFNα联合治疗的研究成果,显示联合用药的受试者HBsAg下降幅度更大,部分受试者实现HBsAg转阴和血清学转换。同时,他也提及了AB-729与IFN联合治疗的研究进展。
针对TQA3038 Ⅱ期研究,评价TQA3038注射液联合NUC治疗经治慢乙肝患者的初步疗效及安全性。研究显示无严重不良事件发生,TQA3038可快速降低HBsAg水平,高剂量组效果更明显。
最后,他总结了TQA3038的技术演进路径和临床应用前景。TQA3038用药后在外周血浓度快速下降,48小时后检测不到,且GalNAc-siRNA可在肝脏中蓄积并缓慢释放,单次给药即可维持长期疗效,使其成为慢乙肝治疗领域的潜力创新药物。
毕吕存:生物分析策略从方法开发到全球一致性验证
徕博科执行总监及中国生物分析与亚太LCMS实验室负责人毕吕存博士探讨了小核酸药物的生物分析策略包括:药代动力学(PK)生物分析策略和生物标志物(BM)生物分析策略。
徕博科执行总监及中国生物分析与亚太LCMS实验室负责人 毕吕存博士
小核酸药物的基本结构是由20至30个核苷酸基团组成的短线性链,通过磷酸二酯键连接,具有强亲水性和高度负电荷性。在生物分析方法上,毕博士展示了定量分析小核酸药物的各技术手段,特别是液相色谱-质谱联用技术(LC-MS/MS)和高分辨率质谱(LC-HRMS)的应用。通过案例研究,可以利用这些技术对小核酸药物及其代谢物进行精确的定量分析。
针对小核酸药物的生物标志物分析,毕博士强调了评估药效动力学(PD)标志物或其他测量指标的重要性。小核酸药物在脏器组织中具有很长半衰期,能够保持很长疗效,因此评估其药效响应时需考虑组织分布和药效动力学特点。
此外,他还介绍了小核酸药物递送技术的创新,特别是GalNAc(N-乙酰半乳糖胺)递送技术能将小核酸递送至肝细胞,并通过化学修饰改善小核酸药物,很多生物科技公司更专注于肝外递送技术。优秀的生物分析技术能够提供答案,这些修饰后的小核酸药物能否有选择性地递送到达目标器脏,以及这些小核酸药物的体内分布和药效。
在方法开发方面,他介绍了小核酸药物生物分析中的各种困难, 以及解决这些困难的方法,特别是针对组织样品的处理和分析中的经验。通过案例研究,毕博士介绍了如何优化提取条件、色谱分离和质谱检测参数,以提高分析的灵敏度和准确性。
最后,毕博士总结了徕博科在小核酸药物生物分析的经验和成果,强调了多种生物分析工具(如LC-MS、qPCR、Simoa、ELISA、MSD和免疫捕获LC-MS)在支持小核酸药物PK/PD评估中的作用。“这些分析工具为药物设计和决策提供了关键数据,推动了小核酸药物的持续发展。”他说。
李海涛:炫景生物的创新实践
炫景生物联合创始人及CTO李海涛博士介绍了小核酸药物领域的全球研发环境、关键决策点和炫景生物的创新实践。
炫景生物联合创始人及CTO 李海涛博士
他认为,小核酸药物正迎来前所未有的机遇。全球范围内已有超过20款药物成功上市,且市场规模保持高速增长。在中国,小核酸药物开始显示出巨大潜力。
然而,小核酸Biotech公司也面临严峻的生存环境,一级市场融资金额大幅下滑,国内小核酸药热门靶点扎堆,siRNA领域内卷严重,进一步压缩了Biotech的生存空间。李海涛博士强调,Biotech公司是License-out交易项目的主要来源,其创新性在全球合作中发挥着关键作用。
在研发路径与关键决策维度上,李海涛认为,产品差异化和技术创新是小核酸Biotech企业在严苛生存环境中脱颖而出的关键。靶点选择需综合临床需求、竞争格局和审评需求,梯度开发扩展适应症以分散风险。平台适配方面,需权衡自主研发与授权引进的利弊,选择合适的平台(如ASO或siRNA)和技术路线。临床试验则需考虑不同临床方案的优劣、全球多中心的必要性和紧迫性等。此外,商业化准备也是关键一环。
对于小核酸药物的发展趋势,李海涛认为,在经历过第一轮“踩踏式”的肝靶点同质化开发后,越来越多的小核酸企业会转向新靶点新机制的研究,一方面实现真正的产品管线差异化、多元化,另一方面也能满足人们对小核酸药物解决不可成药靶点和真正面向未满足临床需求的期待和愿景。此外,多靶向新结构和组合疗法的应用将迅速兴起、强效保障并长效维持治疗效果。递送技术将突破人体主要组织器官的靶向递送,实现更加精准、高效和安全的药物递送。同时,生产成本降低和规模扩大将提高药物可及性,而AI/ML技术将从辅助工具转变为决策支持。
在介绍炫景生物时,李海涛提到,作为国家高新技术企业和国家级科技型中小企业,炫景生物致力于成为小核酸制药领域的深耕者和变革者。公司自2022年成立以来,已获得近3亿元融资,得到多家知名投资机构的支持。
炫景生物拥有自主研发的药物设计筛选平台RIHOST®、化学修饰平台RICMO®和体内递送平台LICOD®等核心技术平台,形成了独特的技术壁垒。药物设计筛选平台实现了智能化序列设计和高通量筛选,大大缩短了研发周期。
化学修饰平台通过新单体、新模式、新结构的三层次突破,建立了“通式”修饰模式,提高了药物的稳定性、有效性、长效性和安全性。递送技术平台则涵盖了肝内和肝外递送技术,肝靶向载体具有高效的组织靶向性和成本优势等。此外,炫景生物还建立了7个肝外组织的递送技术,其中肾脏靶向递送载体率先完成食蟹猴体内验证,研发进度全球领先。在管线开发方面,首发管线RG002已启动临床Ⅱ期研究,安全性有效性优于同靶点竞品。
谈及创业感悟时,李海涛表示,自己出身于科学家背景,在技术上的挑战并非最为棘手,真正困难在于如何实现从科学家到企业家的角色转变。
会议最后,来自国内小核酸药物领域的企业代表,包括炫景生物、瑞博生物、腾盛博药、思合基因、浩博医药、圣诺医药、海昶生物、传信生物、伽进生物、圆因生物、高田生物、星核迪赛以及维亚臻生物等公司,分享了各自公司的平台技术、研发管线布局、融资进展情况及BD计划等。他们认为,当前是中国小核酸药物行业发展的黄金时机,但同时也指出,递送技术、靶向范围拓展、稳定性提升及免疫原性降低等技术瓶颈仍亟待突破。
编辑 | 姚嘉
yao.jia@PharmaDJ.com
总第2565期
访问研发客网站,深度报道和每日新闻抢鲜看
www.PharmaDJ.com
siRNA核酸药物引进/卖出
2025-08-13
关注并星标CPHI制药在线
*展会预登记火热进行中,👆扫描二维码免费领票!
心血管疾病是全球健康的“头号威胁”之一。高胆固醇(例如低密度脂蛋白胆固醇LDL-C)是诱发动脉粥样硬化的重要因素。传统治疗中,他汀类药物需每日服用,患者因依从性差难以达标。
2025年7月31日,诺华宣布其siRNA(小干扰RNA)疗法inclisiran(商品名:Leqvio®)获FDA批准为一线单药,每年仅需注射两次即可降低成人高胆固醇血症患者的LDL-C水平。
一、Leqvio的突破性
Leqvio是一款“first-in-class”、靶向PCSK9 mRNA的siRNA疗法。2021年首次获批时,ORION系列试验(ORION-9、-10、-11)显示,Leqvio较安慰剂持续降低LDL-C达52%,参与试验的3400余名动脉粥样硬化性心血管疾病(ASCVD)或杂合子家族性高胆固醇(HeFH)患者的数据夯实了其疗效。2023年适应症扩展后,其可作为饮食和他汀类药物治疗的辅助用药。
今年7月的这次适应症扩展,是FDA主动要求更新Leqvio药品标签,更新后的药品标签去除了Leqvio必须与他汀类药物联合使用或在其基础上使用的限制。同样,本次适应症拓展也源于扎实的临床数据。
2024年公布的V-MONO三期研究数据更具说服力:对未接受降脂治疗的低或中风险ASCVD患者,Leqvio单药疗效优于安慰剂和依折麦布(ezetimibe),LDL-C降低幅度具有临床和统计学意义。这为其成为单药疗法提供了关键依据。
此外,“半年一针”的给药方式是Leqvio的核心优势。不同于每日服药,Leqvio由医护人员注射,初始两针间隔3个月,此后每6个月一次。这一给药频率直击传统治疗痛点,美国高达80%的ASCVD患者难以达到指南推荐的LDL-C<70 mg/dL的目标,而长效给药大幅提升了用药依从性。
本次批准中,其他更新内容还包括将标签中“原发性高脂血症”一词统一修改为更具体的“高胆固醇血症”,更精准聚焦LDL-C降低,强化了其作为精准降脂疗法的定位,为临床应用提供了清晰指引。
二、siRNA技术是长效特性的科学根基
Leqvio的成功,源于siRNA技术的独特优势,它通过靶向PCSK9基因的mRNA,从源头抑制该蛋白合成。PCSK9蛋白会对低密度脂蛋白(LDL)受体的回收与再利用产生抑制作用。所以,若能降低PCSK9蛋白的水平,就可使更多LDL受体重新回到肝细胞表面。这些受体能结合更多的LDL,并将其从血液中清除出去。
技术迭代是长效的根基。早期siRNA因递送系统缺陷难以成药,而Leqvio采用了三触角结构N-乙酰半乳糖(GalNAc)偶联技术,GalNAc配体可以精准的与肝细胞膜表面表达的ASGPR结合。
图注:Leqvio的结构(来源:参考资料[4])
由于Leqvio抑制PCSK9蛋白的产生是持续性的,而且肝脏清除LDL-C的能力增强,因此这种降LDL-C的效果可以持续较长时间。这使其无需频繁给药,就能持续降脂,解决了慢性病治疗的“依从性魔咒”。
图注:Leqvio在体内的作用机制(来源:参考资料[4])
众多药企也布局了siRNA技术。礼来的Lepodisiran在二期ALPACA试验中实现Lp(a)水平平均降低93.9%,一些患者的降低持续了近1.5年[5]。安进的Olpasiran能将Lp(a)降低95%以上,并且在最后一剂后近一年对Lp(a)的降低显示出持久的效果[]。这些临床数据印证了siRNA在降脂领域的巨大潜力,不仅仅是PCSK9,更多靶点正被攻克。
中国药企也在加速追赶。齐鲁制药、石药集团等企业的长效siRNA制剂进入临床,靶点覆盖PCSK9、ANGPTL3等。舶望制药与诺华达成超41亿美元合作,其siRNA药物管线进展迅速。瑞博生物的RBD7022靶向ASGPR/PCSK9,腾盛博药的elebsiran聚焦乙肝,维亚臻生物的VSA006靶向17β-HSD13瞄准代谢相关脂肪性肝炎(MASH),展现出本土创新潜力。
结语
从每日服药到半年一针,从辅助治疗到一线单药,Leqvio的获批是siRNA技术成熟的代表。它证明了RNA干扰不仅能攻克罕见病,更能在心血管等慢性病领域掀起变革。随着更多临床试验的推进,以及更多企业的技术突破,一次给药管半年甚至一年的降脂时代已近在眼前。
siRNA的高度特异性、广靶点潜力和长效特性,将为更多疾病提供新疗法。未来,siRNA疗法有望为全球患者带来更轻松、更有效的健康守护。
参考资料:
1.Novartis twice-yearly* Leqvio® (inclisiran) receives FDA approval for new indication enabling first-line use, Jul 31, 2025, www.novartis.com
2.FDA Approves Inclisiran (Leqvio) Label Update as First-Line Monotherapy in Hypercholesterolemia, July 31, 2025, www.hcplive.com
3.Novartis twice-yearly* Leqvio® demonstrated clinically meaningful, statistically significant LDL-C lowering as a monotherapy in patients at low or moderate ASCVD risk, Aug 28, 2024, www.novartis.com
4.Migliorati, Julia M et al. “siRNA drug Leqvio (inclisiran) to lower cholesterol.” Trends in pharmacological sciences vol. 43,5 (2022): 455-456. doi:10.1016/j.tips.2022.02.003
5.Lilly's lepodisiran reduced levels of genetically inherited heart disease risk factor, lipoprotein(a), by nearly 94% from baseline at the highest tested dose in adults with elevated levels, March 30, 2025, investor.lilly.com
6.AMGEN PRESENTS LATE-BREAKING PHASE 2 OLPASIRAN DATA AT ESC 2023, www.amgen.com
END
智药研习社近期直播预告
来源:CPHI制药在线
声明:本文仅代表作者观点,并不代表制药在线立场。本网站内容仅出于传递更多信息之目的。如需转载,请务必注明文章来源和作者。
投稿邮箱:Kelly.Xiao@imsinoexpo.com
▼更多制药资讯,请关注CPHI制药在线▼
点击阅读原文,进入智药研习社~
siRNA临床3期引进/卖出
2025-08-07
Arrowhead Pharmaceuticals Reports...
– Conference Call and Webcast Today, August 7, 2025, at 4:30 p.m. ET
PASADENA, Calif.–(BUSINESS WIRE)–Aug. 7, 2025– Arrowhead Pharmaceuticals, Inc. (NASDAQ: ARWR) today announced financial results for its fiscal 2025 third quarter ended June 30, 2025. The Company is hosting a conference call today, August 7, 2025, at 4:30 p.m. ET to discuss the results.
“Arrowhead continues to achieve strong execution in discovery, clinical and regulatory, and business development. Our pipeline has become quite mature, with four Arrowhead discovered candidates currently in pivotal Phase 3 studies. In addition, our commercial buildout is designed to make us launch ready on day one, should plozasiran receive regulatory approval on the November 18, 2025, PDUFA date,” said Christopher Anzalone, Ph.D., President and CEO at Arrowhead. “We continue to maintain a strong balance sheet, which we believe gives us the financial resources to move multiple innovative new medicines through the clinical and regulatory process and ultimately get them to the patients who need them.”
Selected Recent Events
Announced the signing of an asset purchase agreement between Sanofi and Visirna Therapeutics, a majority-owned subsidiary of Arrowhead created to develop and commercialize four of Arrowhead’s investigational cardiometabolic candidates in Greater China. Summary terms of the agreement include: Visirna will receive an upfront payment of $130 million from Sanofi. In addition, Visirna will be eligible to receive further milestone payments of up to $265 million upon approval of plozasiran across various indications in mainland China. Sanofi will receive an exclusive license to develop and commercialize investigational plozasiran in Greater China from Visirna Therapeutics, offering potential treatment to people living with elevated triglycerides. Earned a $100 million milestone payment from Sarepta Therapeutics when Arrowhead reached the first of two prespecified enrollment targets and subsequent authorization to dose escalate in a Phase 1/2 clinical study of ARO-DM1, an investigational RNA interference (RNAi) therapeutic for the treatment of type 1 myotonic dystrophy (DM1), the most common adult-onset muscular dystrophy. Arrowhead currently expects to achieve the second enrollment target by the end of 2025, which would trigger an additional $200 million milestone payment from Sarepta. Completed enrollment in investigational plozasiran clinical studies SHASTA-3, SHASTA-4, and MUIR-3, the company’s global Phase 3 clinical studies designed to support regulatory submissions for marketing approval in the treatment of severe hypertriglyceridemia. Arrowhead anticipates completing the primary portion of these studies in mid-2026 with topline data expected shortly thereafter and planned submissions for regulatory review and potential approval to follow. The company previously submitted a New Drug Application (NDA) for plozasiran based on positive Phase 3 PALISADE study results in patients with familial chylomicronemia syndrome, which the U.S. FDA has accepted with a Prescription Drug User Fee Act (PDUFA) action date set for November 18, 2025. Dosed the first subject in the YOSEMITE Phase 3 clinical trial of zodasiran, the company’s investigational RNAi therapeutic being developed as a potential treatment for homozygous familial hypercholesterolemia (HoFH), a rare genetic condition that leads to severely elevated LDL-cholesterol and early onset cardiovascular disease. Zodasiran is the fourth investigational RNAi-based candidate developed by Arrowhead to reach late-stage pivotal studies, after investigational drugs plozasiran, fazirsiran (licensed to Takeda) and olpasiran (licensed to Amgen). Dosed the first subjects in a Phase 1/2a clinical trial of ARO-ALK7, the company’s investigational RNAi therapeutic being developed as a potential treatment for obesity. ARO-ALK7 is designed to intervene in a known pathway that signals the body to store fat in adipose tissue with a novel mechanism of action that may better preserve lean muscle mass compared to currently approved obesity therapies. The study initiates in otherwise healthy obese subjects using single and multiple escalating doses of ARO-ALK7 monotherapy and is expected to progress rapidly to investigate combinations of ARO-ALK7 with tirzepatide in obese patients with and without type 2 diabetes.
Visirna will receive an upfront payment of $130 million from Sanofi. In addition, Visirna will be eligible to receive further milestone payments of up to $265 million upon approval of plozasiran across various indications in mainland China. Sanofi will receive an exclusive license to develop and commercialize investigational plozasiran in Greater China from Visirna Therapeutics, offering potential treatment to people living with elevated triglycerides.
Selected Fiscal 2025 Third Quarter Financial Results
ARROWHEAD PHARMACEUTICALS, INC.
CONSOLIDATED CONDENSED FINANCIAL INFORMATION
(in thousands, except per share amounts)
Three Months Ended June 30,
OPERATING SUMMARY
2025
2024
(Unaudited)
Revenue
$
27,767
$
—
Operating Expenses:
Research and development
162,368
152,431
General and administrative expenses
30,949
23,710
Total operating expenses
193,317
176,141
Operating loss
(165,550
)
(176,141
)
Total other (expense) income
(13,539
)
2,164
Loss before income tax expense and noncontrolling interest
(179,089
)
(173,977
)
Income tax (benefit) expense
(437
)
—
Net loss including noncontrolling interest
(178,652
)
(173,977
)
Net loss attributable to noncontrolling interest, net of tax
(3,411
)
(3,184
)
Net loss attributable to Arrowhead Pharmaceuticals, Inc.
$
(175,241
)
$
(170,793
)
Net loss per share attributable to Arrowhead Pharmaceuticals, Inc. – Diluted
$
(1.26
)
$
(1.38
)
Weighted-average shares used in calculating – Diluted
139,039
124,199
FINANCIAL POSITION SUMMARY
June 30, 2025
September 30, 2024
(unaudited)
Cash, cash equivalents and restricted cash
$
129,793
$
102,685
Available-for-sale securities, at fair value
770,579
578,276
Total cash resources (Cash, cash equivalents and restricted cash and Available-for-sale securities, at fair value)
900,372
680,961
Other current and long-term assets
480,240
458,841
Total Assets
$
1,380,612
$
1,139,802
Liability related to the sale of future royalties
$
360,254
$
341,361
Credit Facility
240,332
393,183
Deferred revenue
22,979
—
Other liabilities
237,241
214,195
Total Liabilities
$
860,806
$
948,739
Total Arrowhead Pharmaceuticals, Inc. Stockholders’ Equity
522,313
185,444
Noncontrolling Interest
(2,507
)
5,619
Total Noncontrolling Interest and Stockholders’ Equity
$
519,806
$
191,063
Total Liabilities, Noncontrolling Interest and Stockholders’ Equity
$
1,380,612
$
1,139,802
Shares Outstanding
138,144
124,376
Webcast and Conference Call and Details
Investors may access a live audio webcast on the Events and Presentations page under the Investors section of the Arrowhead website. A replay of the webcast will be available approximately two hours after the conclusion of the call.
For analysts that wish to participate in the conference call, please register at https://register-conf.media-server.com/register/BI3955ca6732974874b0bc0ac323811a7f. Once registered, you will receive the dial-in number and a personalized PIN code that will be required to access the call.
About Arrowhead Pharmaceuticals
Arrowhead Pharmaceuticals develops medicines that treat intractable diseases by silencing the genes that cause them. Using a broad portfolio of RNA chemistries and efficient modes of delivery, Arrowhead therapies trigger the RNA interference mechanism to induce rapid, deep, and durable knockdown of target genes. RNA interference, or RNAi, is a mechanism present in living cells that inhibits the expression of a specific gene, thereby affecting the production of a specific protein. Arrowhead’s RNAi-based therapeutics leverage this natural pathway of gene silencing.
For more information, please visit www.arrowheadpharma.com, or follow us on X (formerly Twitter) at @ArrowheadPharma, LinkedIn, Facebook, and Instagram. To be added to the Company’s email list and receive news directly, please visit http://ir.arrowheadpharma.com/email-alerts.
Safe Harbor Statement under the Private Securities Litigation Reform Act:
This news release contains forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. Any statements contained in this release except for historical information may be deemed to be forward-looking statements. Without limiting the generality of the foregoing, words such as “may,” “will,” “expect,” “believe,” “anticipate,” “hope,” “intend,” “plan,” “project,” “could,” “estimate,” “continue,” “target,” “forecast” or “continue” or the negative of these words or other variations thereof or comparable terminology are intended to identify such forward-looking statements. In addition, any statements that refer to projections of our future financial performance, trends in our business, expectations for our product pipeline or product candidates, including anticipated regulatory submissions and clinical program results, prospects or benefits of our collaborations with other companies, or other characterizations of future events or circumstances are forward-looking statements. These forward-looking statements include, but are not limited to, statements about the initiation, timing, progress and results of our preclinical studies and clinical trials, including the timing of achievement of the next ARO-DM1 milestone, and our research and development programs; our expectations regarding the potential benefits of the partnership, licensing and/or collaboration arrangements and other strategic arrangements and transactions we have entered into or may enter into in the future; our beliefs and expectations regarding milestone, royalty or other payments that could be due to or from third parties under existing agreements, including our ability to achieve such milestones on projected timelines (if at all) and receive timely payment if milestones are achieved; and our estimates regarding future revenues, research and development expenses, capital requirements and payments to third parties. These statements are based upon our current expectations and speak only as of the date hereof. Our actual results may differ materially and adversely from those expressed in any forward-looking statements as a result of numerous factors and uncertainties, including the impact of the ongoing COVID-19 pandemic on our business, the safety and efficacy of our product candidates, decisions of regulatory authorities and the timing thereof, the duration and impact of regulatory delays in our clinical programs, our ability to finance our operations, the likelihood and timing of the receipt of future milestone and licensing fees, the future success of our scientific studies, our ability to successfully develop and commercialize drug candidates, the timing for starting and completing clinical trials, rapid technological change in our markets, the enforcement of our intellectual property rights, and the other risks and uncertainties described in our most recent Annual Report on Form 10-K, subsequent Quarterly Reports on Form 10-Q and other documents filed with the Securities and Exchange Commission from time to time. We disclaim any intention to update or revise forward-looking statements to reflect new events or circumstances.
Source: Arrowhead Pharmaceuticals, Inc.
View source version on businesswire.com: https://www.businesswire.com/news/home/20250807601354/en/
Arrowhead Pharmaceuticals, Inc. Vince Anzalone, CFA 626-304-3400 ir@arrowheadpharma.com
Investors: LifeSci Advisors, LLC Brian Ritchie 212-915-2578 britchie@lifesciadvisors.com
Media: LifeSci Communications, LLC Kendy Guarinoni, Ph.D. 724-910-9389 kguarinoni@lifescicomms.com
Source: Arrowhead Pharmaceuticals, Inc.
引进/卖出临床3期财报申请上市
100 项与 Olpasiran 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 动脉粥样硬化 | 临床3期 | 阿根廷 | 2022-11-30 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床2期 | 272 | olpasiran 10 mg Q12W | - | 积极 | 2025-03-02 | ||
olpasiran 75 mg Q12W | |||||||
临床1期 | 24 | (Olpasiran Low Dose) | 蓋窪餘鏇鬱醖構構醖餘(衊築築齋範憲鏇齋鏇鹽) = 觸選鹹窪蓋齋憲淵鹹簾 簾築糧積淵構選繭壓構 (觸簾壓蓋淵鹹淵廠壓願, 55.5) 更多 | - | 2025-02-10 | ||
(Olpasiran High Dose) | 蓋窪餘鏇鬱醖構構醖餘(衊築築齋範憲鏇齋鏇鹽) = 鹽製膚簾膚壓選簾獵製 簾築糧積淵構選繭壓構 (觸簾壓蓋淵鹹淵廠壓願, 71.5) 更多 | ||||||
N/A | 冠状动脉疾病 lipoprotein(a) 260 nmol/L | lipoprotein(a) 226 nmol/L | - | 淵襯選蓋窪鏇壓窪願顧(繭鹽糧繭鏇壓餘簾網簾) = 觸鑰憲衊鑰築獵網齋糧 網遞憲夢蓋範獵廠範衊 (壓顧鹹簾壓築鏇構獵夢 ) 更多 | 积极 | 2024-11-01 | ||
選選願廠鏇鬱遞鹹衊願(獵艱蓋築壓壓憲製顧夢) = 憲淵獵選築遞鹽憲鑰製 願淵艱襯膚鏇製淵憲鬱 (願顧顧選蓋獵窪遞遞鏇 ) | |||||||
临床2期 | 276 | 糧獵鹽簾衊窪蓋糧積膚(願獵衊窪蓋遞製範膚構) = 餘鏇積糧鹹襯艱憲範簾 夢鏇簾廠餘積廠憲壓鏇 (願淵網構鹽構鹹蓋積衊 ) | 积极 | 2024-08-27 | |||
糧獵鹽簾衊窪蓋糧積膚(願獵衊窪蓋遞製範膚構) = 鏇選鏇鏇壓觸願衊齋遞 夢鏇簾廠餘積廠憲壓鏇 (願淵網構鹽構鹹蓋積衊 ) 更多 | |||||||
临床2期 | 281 | 範醖糧範鏇製襯憲夢衊(艱觸觸夢顧淵衊鬱艱遞) = The results from the off-treatment extension period show that patients previously dosed with ≥75 mg of olpasiran sustained a ~40-50% placebo-adjusted percent reduction in Lp(a) nearly a year after the last dose. 鹽積膚齋憲範選築窪襯 (醖蓋窪遞鹽窪鏇憲憲繭 ) | 积极 | 2023-08-26 | |||
placebo | |||||||
临床2期 | 281 | Olpasiran 10 mg every 12 weeks | 構廠憲窪膚艱蓋壓壓簾(蓋憲餘獵醖窪廠觸醖餘) = 醖淵憲膚範選蓋築窪鹹 構鑰築廠蓋願觸艱窪鬱 (鬱觸簾鹹廠製襯壓鏇簾 ) | - | 2022-11-06 | ||
Olpasiran 75 mg every 12 weeks | 構廠憲窪膚艱蓋壓壓簾(蓋憲餘獵醖窪廠觸醖餘) = 築膚鹽製範襯願簾糧顧 構鑰築廠蓋願觸艱窪鬱 (鬱觸簾鹹廠製襯壓鏇簾 ) | ||||||
临床1期 | 27 | (Japanese participants) | 簾齋壓願簾繭鑰顧醖醖(壓襯簾獵膚遞顧壓鏇範) = 齋廠蓋齋簾選夢膚獵夢 淵簾壓願廠鏇築膚構鏇 (齋觸醖簾膚窪遞網淵衊, 121.0) 更多 | 积极 | 2022-08-10 | ||
(Non-Japanese participants) | 簾齋壓願簾繭鑰顧醖醖(壓襯簾獵膚遞顧壓鏇範) = 積鹽願構淵壓憲簾觸鹽 淵簾壓願廠鏇築膚構鏇 (齋觸醖簾膚窪遞網淵衊, 71.3) 更多 | ||||||
临床2期 | 281 | 窪膚淵膚範鏇鹹鏇鹹製(鏇繭廠繭醖艱蓋願蓋廠) = These data demonstrated a significant reduction from baseline in Lp(a) of up to or greater than 90 percent 鬱憲鏇積夢艱鹹蓋鬱醖 (壓淵廠獵窪顧廠鹽壓願 ) 更多 | 积极 | 2022-05-31 | |||
Placebo | |||||||
临床1期 | - | 64 | 壓遞積醖淵獵壓鑰壓觸(鏇衊遞繭鹽餘齋蓋糧繭) = The most common TEAEs were headache (10% AMG 890; 25% placebo) and upper respiratory tract infection (15% AMG 890; 13% placebo) 餘繭鹽艱蓋範糧窪製鹹 (鑰壓餘糧襯衊艱網醖壓 ) 更多 | 积极 | 2020-11-12 | ||
Placebo |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用