预约演示
更新于:2025-05-07
Mild dementia
轻度痴呆
更新于:2025-05-07
基本信息
别名 Dementia, mild, NOS、Mild dementia、Mild dementia (diagnosis) + [2] |
简介- |
关联
6
项与 轻度痴呆 相关的药物靶点 |
作用机制 GLP-1R激动剂 |
在研机构 |
原研机构 |
非在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2017-12-05 |
作用机制 NMDA receptor调节剂 |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
作用机制 NMDA receptor调节剂 |
在研机构 |
原研机构 |
非在研适应症- |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
154
项与 轻度痴呆 相关的临床试验NCT06723015
Study of Tau PET Outcomes in Patients Receiving Anti-amyloid Immunotherapies
This is an imaging study of patients with mild Alzheimer's disease receiving amyloid-targeting therapy and under the care of neurologists at Mayo Clinic in Rochester, MN.
开始日期2025-08-01 |
申办/合作机构 |
NCT06264362
Developing a Nonpharmacological Pain Intervention for Community-dwelling Older Adults With Dementia
The purpose of this study is to develop an exercise treatment program for knee osteoarthritis that works well for people with memory problems. This study will include two parts. Participants can choose to participate in either or both parts.
In Part 1 (not a clinical trial) - The investigators are inviting the following people to participate in an interview:
1. Individuals aged 60 years or older who do not live in a nursing home and have both memory issues and knee pain.
2. Care partners of people with knee pain and memory issues. A care partner can be a family member or friend who helps regularly.
3. Clinicians (exercise physiologists or rehabilitation therapists like physical and occupational therapists) who have worked with older adults with memory issues and knee pain.
The goal of these interviews is to learn from the experiences of people with memory issues and knee pain and those who care for them. From these interviews, the investigators hope to design a non-drug, tailored physical activity program that will be interesting and helpful in managing knee pain for older adults with memory problems.
For people with memory problems and knee pain and their care partners: The investigators will ask some questions to determine eligibility to participate in the study.
Those who qualify for the study will be invited to participate in an interview that could be completed online or in person (Philadelphia, PA). The interview could last up to 90 minutes (1 and a half hours) and can be completed as one or multiple sessions.
For clinicians: Participants will engage in a 90-minute focus group with other clinicians where lunch will be served if in person. Those participating in an online focus group will receive a gift card equal to the value of lunch.
In Part 2 (a clinical trial) - The program developed in Part 1 will be tested. The program will be physical activity-based. Specific program details for part 2 will be provided as they emerge. Participants can choose to sign up for either or both parts. Signing up for Part 1 does not mean participants have to sign up for Part 2.
Please contact us for any questions.
In Part 1 (not a clinical trial) - The investigators are inviting the following people to participate in an interview:
1. Individuals aged 60 years or older who do not live in a nursing home and have both memory issues and knee pain.
2. Care partners of people with knee pain and memory issues. A care partner can be a family member or friend who helps regularly.
3. Clinicians (exercise physiologists or rehabilitation therapists like physical and occupational therapists) who have worked with older adults with memory issues and knee pain.
The goal of these interviews is to learn from the experiences of people with memory issues and knee pain and those who care for them. From these interviews, the investigators hope to design a non-drug, tailored physical activity program that will be interesting and helpful in managing knee pain for older adults with memory problems.
For people with memory problems and knee pain and their care partners: The investigators will ask some questions to determine eligibility to participate in the study.
Those who qualify for the study will be invited to participate in an interview that could be completed online or in person (Philadelphia, PA). The interview could last up to 90 minutes (1 and a half hours) and can be completed as one or multiple sessions.
For clinicians: Participants will engage in a 90-minute focus group with other clinicians where lunch will be served if in person. Those participating in an online focus group will receive a gift card equal to the value of lunch.
In Part 2 (a clinical trial) - The program developed in Part 1 will be tested. The program will be physical activity-based. Specific program details for part 2 will be provided as they emerge. Participants can choose to sign up for either or both parts. Signing up for Part 1 does not mean participants have to sign up for Part 2.
Please contact us for any questions.
开始日期2025-06-02 |
申办/合作机构  Drexel University Drexel University [+1] |
NCT06928298
Cognitive Screening Made Easy for PCPs - Administrative Supplement
This project will facilitate early detection of cognitive decline in older adults through development and implementation of an automated risk assessment and cognitive screening tool for use in primary care. By providing an automated tool developed specifically to address the needs of PCPs, it will be easier to screen for cognitive impairment, increasing the number of older adults who are screened and thus identified and treated.
开始日期2025-04-30 |
100 项与 轻度痴呆 相关的临床结果
登录后查看更多信息
100 项与 轻度痴呆 相关的转化医学
登录后查看更多信息
0 项与 轻度痴呆 相关的专利(医药)
登录后查看更多信息
1,285
项与 轻度痴呆 相关的文献(医药)2025-06-01·The American Journal of Geriatric Psychiatry
Developing Treatment Models for the Delivery of the Antiamyloid Therapy, Lecanemab: Considerations for Implementation of Lecanemab in Healthcare Systems
Article
作者: Johnson, Kim G ; Abramowitz, Amy ; Weber, Michael ; Roth, Heidi L
2025-05-01·The Journal of Prevention of Alzheimer's Disease
Donanemab: Appropriate use recommendations
Article
作者: Weiner, M ; Greenberg, S M ; Rabinovici, G D ; Atri, A ; Salloway, S ; Schindler, S E ; Apostolova, L G ; Aisen, P ; Hendrix, S B ; Selkoe, D J ; Cummings, J ; Petersen, R C
2025-05-01·Neurobiology of Sleep and Circadian Rhythms
Beyond sleep: Rest and activity rhythm as a marker of preclinical and mild dementia in older adults with less education
Article
作者: Satomi, Erika ; Ribeiro, Elyse ; Suemoto, Claudia Kimie ; Magaldi, Regina Miksian ; Busse, Alexandre Leopold ; Poyares, Dalva ; Apolinário, Daniel ; Genta, Pedro Rodrigues ; Piovezan, Ronaldo Delmonte ; Jacob-Filho, Wilson ; Vieira Gomes, Gisele Cristina
274
项与 轻度痴呆 相关的新闻(医药)2025-05-01
·医药速览
欧洲时间4月15日,仑卡奈单抗正式获得欧盟批准上市,用于治疗经临床诊断为轻度认知障碍及轻度痴呆的早期阿尔茨海默病(AD)成人患者,且为载脂蛋白E ε4(ApoE ε4)非携带者或杂合子,并经确认存在β-淀粉样蛋白病理。此次获批,使仑卡奈单抗成为首款在欧盟获批用于治疗符合条件的早期阿尔茨海默病(early AD)患者的、针对该疾病潜在病因的药物。而20天前,欧洲药品管理局(EMA)人用药品委员会(CHMP)却以 “疗效不足以抵消安全风险” 为由,拒绝批准多奈单抗在欧盟上市。在很多人眼里,仑卡奈单抗和多奈单抗大同小异,都是针对Aβ的药物,这让EMA的拒批看起来非常无理,但实际上二者在证据的充分性和获益程度上,表现大相径庭。漫漫审批路EMA向来是全球最为保守的药物审批部门,仑卡奈单抗在欧盟的批准也是一波三折。EMA是先拒绝了仑卡奈单抗的上市申请,后经补充递交数据再次申请后,才由拒绝转为建议批准。期间多奈单抗吸取了仑卡奈单抗第一次闯关失败被拒的经验,直接放弃挑战ARIA发生率较高的ApoE ε4携带人群。但EMA依然认为,即便对于非ApoE4携带者这一小规模人群,多奈单抗的有限疗效收益仍不足以抵消其引发致命性ARIA的风险。EMA在对TRAILBLAZER – ALZ 2研究的数据进行进一步分析后,发现其对于ApoE ε4非携带者的安全性表现依然不尽如人意,ARIA事件总体发生率达到了24%,比安慰剂组的12%高出一倍,与Clarity AD研究中仑卡奈单抗在总人群中的ARIA发生率相当。那既然仑卡奈单抗没有获批全人群适应症,多奈单抗自然无法获批ApoE ε4非携带者的适应症。更为致命的是,礼来方面虽然提出了重新审查的要求,但他们似乎很难提供更多过硬的证据了。多奈单抗的治疗目标为清除大脑中的淀粉样蛋白斑块,当斑块水平评估达标后即可停药。但这个特点导致其患者的平均暴露时间差别很大,有一些患者可能在12个月内实现停药,有一些患者则可能治疗时长大大超过18个月,疗程并不明确。这导致其在长期疗效数据上的披露进度远落后于仑卡奈单抗。在EMA的意见中也指出,对于多奈单抗治疗ApoE ε4非携带早期AD患者,目前尚无长期有效性数据支持。而且,即便是现有的疗效数据,多奈单抗的获益也很并不明显。从整体数据来看,接受多奈单抗治疗的患者 iADRs 评分恶化了 10 分,而接受安慰剂治疗的患者恶化了 13 分。这表明多奈单抗虽然在一定程度上使患者的评分恶化程度稍低,但改善幅度并不显著,仅仅只有 3 分的差异。而在ApoE ε4非携带者中,接受多奈单抗治疗的患者 iADRs 评分恶化14分,接受安慰剂治疗的患者恶化 16分 。也就是说,在这一特定人群中,多奈单抗与安慰剂的差值仅有2 分。这样的疗效收益绝对无法抵消安全性上的风险。靶点带来安全性差异淀粉样蛋白相关性影像异常(ARIA)是所有抗Aβ药物治疗都可能产生的副作用,因为Aβ不仅可沉积于脑实质,也可沉积于脑血管壁,导致血管壁的结构和功能异常,增加了血管的脆性和易损性。但脑实质与血管壁上的Aβ并不是同样的Aβ,血管壁上沉积的主要是Aβ40,而Aβ42才是主要参与Aβ级联反应的聚合体。所以理想的阿尔茨海默病抗体药物应该尽可能减少与Aβ40的结合度,增加与Aβ42的结合度。而多奈单抗与Aβ40的结合力度比仑卡奈单抗高,这导致理论上多奈单抗的安全性比仑卡奈单抗更差。临床研究结果表明,多奈单抗的ARIA-E和ARIA-H发生率都比仑卡奈单抗高出一倍左右, 而且多奈单抗的临床试验中还出现了3例导致受试者死亡的严重ARIA事件。欧盟对抗Aβ药物态度的 “冰火两重天”,远不是安全性问题可以概括,需要综合评价药物的风险与治疗获益。由于AD 治疗需要“早期干预、长期管理” ,所以药物既要有长期疗效证据,也要能保证长期用药的安全,二者缺一不可,不能做一锤子买卖。推文用于传递知识,如因版权等有疑问,请于本文刊发30日内联系医药速览。原创内容未经授权,禁止转载至其他平台。有问题可发邮件至yong_wang@pku.edu.cn获取更多信息。©2021 医药速览 保留所有权利往期链接“小小疫苗”养成记 | 医药公司管线盘点 人人学懂免疫学| 人人学懂免疫学(语音版)综述文章解读 | 文献略读 | 医学科普|医药前沿笔记PROTAC技术| 抗体药物| 抗体药物偶联-ADC核酸疫苗 | CAR技术| 化学生物学温馨提示医药速览公众号目前已经有近12个交流群(好学,有趣且奔波于医药圈人才聚集于此)。进群加作者微信(yiyaoxueshu666)或者扫描公众号二维码添加作者,备注“姓名/昵称-企业/高校-具体研究领域/专业”,此群仅为科研交流群,非诚勿扰。简单操作即可星标⭐️医药速览,第一时间收到我们的推送①点击标题下方“医药速览” ②至右上角“...” ③点击“设为星标
上市批准临床结果
2025-04-27
·小药说药
-01-引言阿尔茨海默病(Alzheimer′s disease,AD)是发生于老年期和老年前期的神经系统退行性疾病,起病隐袭且病程呈进行性发展,临床表现以获得性认知功能损害为核心症状,涉及记忆、学习、定向、理解、判断、计算、语言和视空间等认知域,不同程度地影响日常生活能力及社会职业功能,最终进展为全面性痴呆。根据GBD数据,2019年全球AD及相关痴呆的患病人数达到5162万,总体患病率为 667.2/10万,年龄标化患病率为 682.5/10万。同年,我国 AD 及相关痴呆总人数为1314 万, 大约占全球AD患者总人数的25.5%,我国AD及相关痴呆总体患病率为924.1/10万,年龄标化患病率为788.3/ 10万。目前我国国家药品监督管理局批准用于治疗 AD的一线药物包括胆碱酯酶抑制剂,如多奈哌齐、 卡巴拉汀等 ,以及N‑甲 基‑D‑门冬氨酸(NMDA)受体拮抗剂美金刚。2019年国家药品监督管理局有条件批准了甘露特钠胶囊上市注册申请,用于治疗轻度至中度AD。2024 年 1 月仑卡奈单抗(lecanemab)注射液获国家药品监督管理局批准上市,用于治疗由 AD引起的 MCI和轻度痴呆。然而,这些方法多半是对症治疗,近一半的患者对现有药物治疗的效果不满,其次是药物价格贵。因此,亟须针对这些问题和痛点进一步改善我国AD患者的治疗现状。主动免疫疗法,即疫苗接种,是针对淀粉样β(Aβ)的阿尔茨海默病(AD)研究最彻底的治疗方法之一。在20多年前, AN1792疫苗在临床进行了研究,虽然因为毒性问题没有继续开发,然而人们也从中看到了疫苗预防AD的潜力。AD的疫苗预防可能改变阿尔茨海默症目前的困境。-02-一、AD的发病机制目前,AD研究的主流观点认为,AD的发病机制是Aβ与Tau蛋白的协同作用结果:Aβ的毒性级联反应Aβ由淀粉样前体蛋白(APP)裂解产生,其可溶性寡聚体(AβOs)是神经毒性的“真凶”。它们攻击突触功能,触发炎症,并诱导Tau蛋白过度磷酸化,形成NFTs。研究表明,可溶性AβOs比斑块与认知衰退的相关性更强。Tau蛋白的雪崩效应Tau蛋白过度磷酸化后脱离微管,形成NFTs,破坏神经元运输系统。这种病理过程存在“交叉播种效应”:AβOs加速Tau病变扩散,而NFTs恶化神经元死亡。炎症与免疫失衡小胶质细胞和星形胶质细胞的慢性炎症反应进一步破坏脑内稳态,形成“病理恶性循环”。-03- 二、疫苗:AD预防的免疫堡垒传统药物治疗聚焦症状缓解,疫苗则直击疾病上游,提供“治未病”的可能。其核心策略包括:1. 精准打击Aβ毒性寡聚体AD疫苗通过诱导抗体中和AβOs,阻止其破坏突触并触发炎症。动物模型显示,靶向AβOs的疫苗可显著延缓认知衰退。2. Th2极化理想疫苗需诱导抗炎性Th2免疫反应,避免促炎性Th1反应加重脑损伤。佐剂选择是关键:传统铝佐剂(Alum)可能引发炎症副作用,而新型Th2偏向佐剂(如TLR9激动剂)可能解决上述问题。3. 被动免疫的局限性单克隆抗体(如Lecanemab、Donanemab和Aducanumab)虽可快速清除Aβ,但需频繁注射且成本高昂。主动免疫疫苗通过激发自身免疫记忆,提供长期保护,更适合预防性应用。-04-三、AD疫苗的开发现状AN-1792是第一个人类Aβ主动免疫疗法,由合成全长人类Aβ42和佐剂QS-21组成。AN-1792的1期临床研究包括372名轻度至中度AD患者。第1阶段试验的结果表明,该疫苗可诱导对Aβ42的抗体反应,并可清除大脑中的斑块。然而,在II期试验中,由于患者发展为脑膜炎,临床试验暂停。尽管AN-1792临床试验有副作用,但该研究的结果鼓励开发新的主动抗Aβ免疫治疗方法。 CAD106疫苗由诺华公司研发,通过诱导抗体产生来减少β淀粉样斑块。该疫苗由截短的β-淀粉样蛋白片段(Aβ1-6)组成,以避免T细胞活化。在第一阶段试验的52周内没有出现脑膜炎的迹象。然而由于缺乏疗效,目前已停止开发。ABvac40疫苗由Araclon Biotech开发,其结构针对Aβ40的C末端,包含与KLH偶联的Aβ33–40。ABvac40 2023年公布了II期临床结果,达到了主要终点,与安慰剂相比,ABvac40治疗可将疾病进展的速度减慢38%。ACI-24疫苗由AC Immune开发,包含Aβ1-15表位,不包括T细胞表位,以避免T细胞反应。ACI‐24的 II期临床试验由于低免疫原性撤回,正在继续优化;新分子ACI-24.060被武田以里程碑总价22亿美元引进。UB-311疫苗由United Biomedical开发,含有Aβ1-14表位,并与辅助T细胞肽表位融合,利用CpG作为佐剂。肽混合物将诱导B细胞反应,同时避免T细胞的炎症反应。2023年发表的2期研究结果显示,UB-311治疗达到了主要终点,该疗法具有良好安全性、耐受性,并产生稳健的免疫应答。-05-四、AD疫苗研发开发的挑战虽然AD疫苗展现出缓解AD进展的前景,但AD疫苗研发仍面临多重“拦路虎”:1. 免疫衰老(Immunosenescence):老年人群免疫应答低下,疫苗需克服T细胞功能衰退和慢性炎症背景。2. 血脑屏障(BBB)阻碍98%的大分子抗体无法穿透BBB,新型的递送技术是破局方向。3. 患者异质性:AD亚型复杂,需基于生物标志物(如Aβ-PET、脑脊液pTau)细分人群,实现精准免疫。4. 临床试验设计困境:AD进程缓慢,需超长随访(10年以上)和替代终点(如Aβ清除率)加速验证。5. 安全性红线:避免诱发自身免疫性脑膜炎(如AN1792)或微出血,是疫苗设计的核心原则。-06-五、未来方向:多靶点联合与技术创新1. 靶向“Aβ-Tau轴”复合疫苗:同时激活针对AβOs和异常Tau的免疫应答,阻断病理级联。2. 新型递送系统:鼻内疫苗、DC细胞疫苗或外泌体载体可能增强脑内抗体渗透。3. 佐剂革命:开发纯Th2极化佐剂,平衡免疫激活与安全性。4. 人工智能助力设计:AI预测抗原表位和抗体结合构象,缩短疫苗开发周期。-07-结语AD是一种严重影响人类健康的多因素异质性神经退行性疾病,目前还没有能够治愈的药物,已获得批准的药物也只能减缓疾病的进展。因此,AD最好在发生严重认知障碍之前进行早期干预,AD预防有潜力可能实现延迟或预防AD痴呆的治疗目标。AD疫苗研发虽荆棘密布,但随着对病理机制的解码和技术迭代,未来十年或有革命性疫苗问世。“疫苗或许不能逆转时光,但能让我们优雅地老去。”愿未来AD将不再成为困扰我们的严重疾病。参考资料:1. The Importance of Vaccines in Preventing Impending Alzheimer's Epidemic. Rev Recent Clin Trials.2025 Apr 14.2. The informed road map to prevention of Alzheimer Disease: A call to arms. Mol Neurodegener.2021 Jul 21;16(1):49.公众号内回复“ADC”或扫描下方图片中的二维码免费下载《抗体偶联药物:从基础到临床》的PDF格式电子书!公众号已建立“小药说药专业交流群”微信行业交流群以及读者交流群,扫描下方小编二维码加入,入行业群请主动告知姓名、工作单位和职务。
免疫疗法临床结果疫苗临床2期
2025-04-25
全球批准1.Biogen公司Lecanemab获EMA批准上市,用于治疗经临床诊断为轻度认知障碍及轻度痴呆的早期阿尔茨海默病(AD)成人患者,且为载脂蛋白E ε4(ApoE ε4*)非携带者或杂合子,并经确认存在β-淀粉样蛋白病理;2.GSK公司Belantamab mafodotin获MHRA批准上市,用于治疗多发性骨髓瘤成年患者;3.Kowa Company公司Pemafibrate获NMPA批准上市,用于治疗血脂异常;4.康方生物Ebdarokimab获NMPA批准上市,用于治疗中重度斑块状银屑病成人患者;全球申报1.Regeneron公司Aflibercept获FDA消极反馈,用于治疗湿性年龄相关性黄斑变性(wAMD)和糖尿病性黄斑水肿(DME);2.Scilex公司Colchicine获FDA孤儿药资格认定,用于治疗心包炎;3.百济神州Sonrotoclax获NMPA拟纳入优先审评,用于治疗既往接受过治疗的慢性淋巴细胞白血病(CLL)/小淋巴细胞淋巴瘤(SLL)成人,以及治疗既往接受过抗CD20治疗和BTKi治疗的套细胞淋巴瘤(MCL)成人患者;4.华毅乐健GS1191-0445获NMPA拟纳入突破性治疗,用于治疗先天性凝血因子VII缺乏引起的血友病A;临床进展1.荣昌生物启动Disitamab Vedotin的III期临床研究,用于治疗HER2低表达的晚期胃/胃食管结合部腺癌;2.信达生物启动Mazdutide的III期临床,用于治疗中重度阻塞性睡眠呼吸暂停且肥胖;3.复宏汉霖完成HLX-22的II期临床首例患者给药,用于治疗HER2低表达HR阳性的局部晚期或转移性乳腺癌;4.恒瑞医药启动HRS-5817的I期临床,用于治疗肥胖;
上市批准优先审批临床3期突破性疗法孤儿药
分析
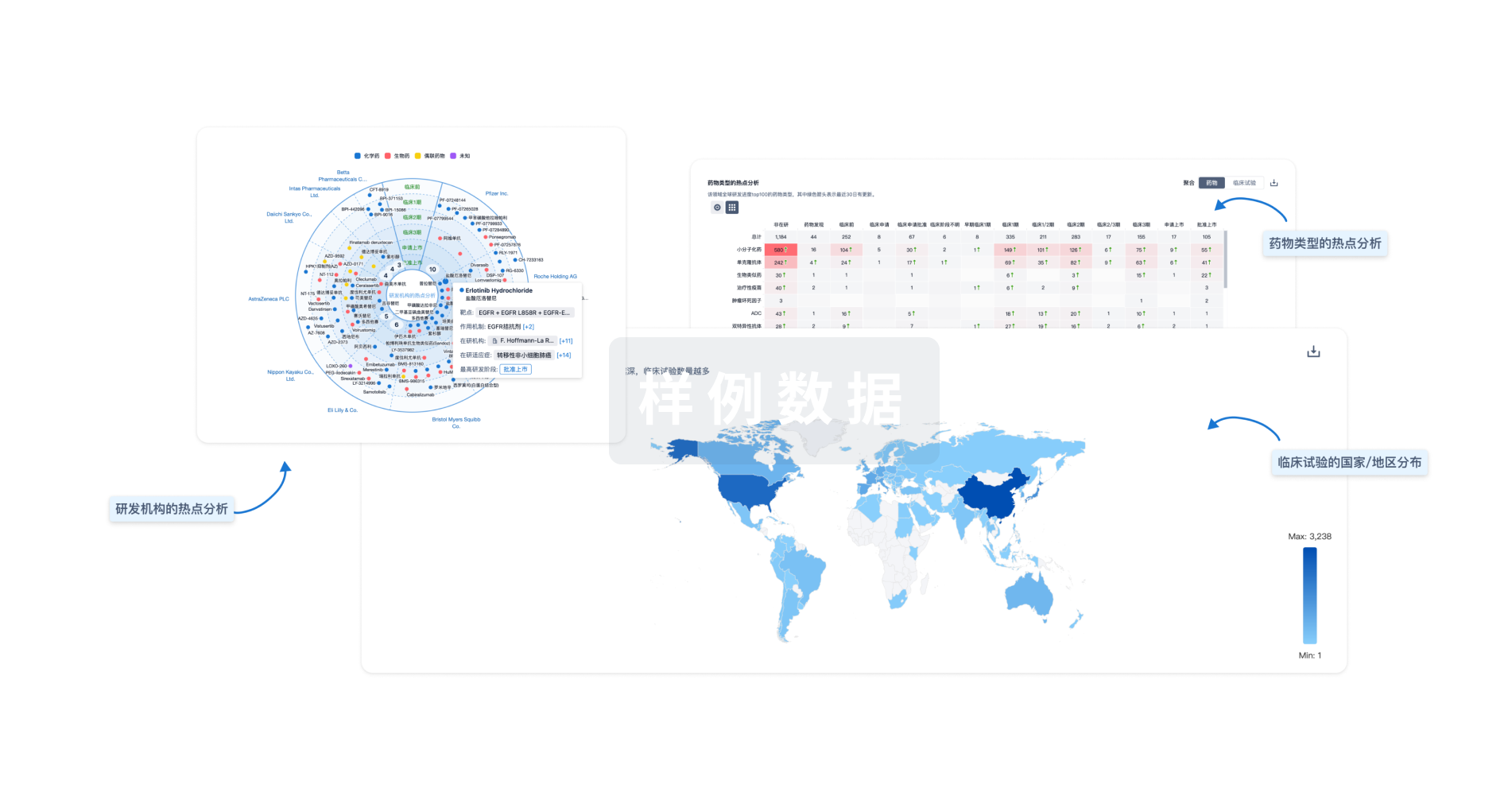
对领域进行一次全面的分析。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用




