预约演示
更新于:2025-05-07
DNA
更新于:2025-05-07
基本信息
别名 DNA |
简介- |
关联
427
项与 DNA 相关的药物靶点 |
作用机制 DNA调节剂 |
在研机构 |
原研机构 |
最高研发阶段批准上市 |
首次获批国家/地区 中国 |
首次获批日期2021-05-26 |
作用机制 CD19抑制剂 [+1] |
在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2021-04-23 |
靶点 |
作用机制 DNA抑制剂 [+1] |
在研机构 |
原研机构 |
在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2021-02-26 |
22,613
项与 DNA 相关的临床试验NCT04832438
Phase 2 Study of 9-ING-41, a Glycogen Synthase Kinase 3 Beta (GSK 3β) Inhibitor, Plus Carboplatin in Patients With Advanced, Metastatic Salivary Gland Carcinoma
9-ING-41 is a small molecule potent selective GSK-3β inhibitor with antitumor activity. This study investigates 9-ING-41 in combination with carboplatin chemotherapy in patients with incurable, recurrent or metastatic salivary gland carcinomas (SGC). Patients with advanced SGC (including all histologic subtypes and adenoid cystic carcinoma [ACC]) will receive 9-ING-41 intravenously (IV) along with carboplatin IV at standard dosing together on Day 1, and 9-ING-41 alone on Day 4 of a 21-day cycle. Participants will be enrolled to two histologic cohorts: Cohort 1 will be comprised of those with ACC, and Cohort 2 will include patients with non-ACC SGC (or all other salivary gland cancer histologies). Treatment will continue until progression of disease, death, or discontinuation of therapy for any reason.
开始日期2030-12-18 |
申办/合作机构 |
NCT05662904
Genetic Ablation of CD33 in Hematopoietic Stem Cells to Broaden the Therapeutic Index of CD33-directed Immunotherapy in Patients with Acute Myeloid Leukemia (AML)
The study "GALAXY33" is an open-label, prospective, nonrandomized, one arm phase I clinical trial in which patients with relapsed AML after allogeneic hematopoietic stem cell transplantation will be transplanted with CD33-deleted CD34+ HSC derived from the initially matched family donor.
开始日期2028-01-01 |
NCT04595981
Chemo-embolization for Head and Neck Cancer
The study will evaluate whether adjuvant chemo-embolization increases progression free and/or overall survival relative to standard of care radiation and chemo- and/or immunotherapy in cisplatin-ineligible head and neck cancer patients with an acceptable morbidity rate.
开始日期2027-05-01 |
100 项与 DNA 相关的临床结果
登录后查看更多信息
100 项与 DNA 相关的转化医学
登录后查看更多信息
0 项与 DNA 相关的专利(医药)
登录后查看更多信息
259
项与 DNA 相关的文献(医药)2025-06-01·Biochemical and Biophysical Research Communications
Structural and functional insights into the SARS-CoV-2 SUD domain and its interaction with RNA G-Quadruplexes
Article
作者: Zhang, Yu-Hang ; Hou, Xi-Miao ; Su, Ai-Min
2025-04-01·Bioorganic & Medicinal Chemistry Letters
Design and synthesis of novel structures with anti-tumor effects: Targeting telomere G-quadruplex and hTERT
Article
作者: Wang, Xutong ; Fang, Xiaodong ; Sun, Meng ; Gao, Zeyu ; Wang, Bing ; Liu, Yu ; Wang, Peiying ; Ma, Kejing ; Han, Weina
2025-03-01·Allergy
Short‐course subcutaneous treatment with birch pollen allergoids greatly improves symptom and medication scores in birch allergy
Article
作者: Rybachuk, Anna ; Xenofontos, Elena ; Neuhof, Christian ; Sahin, Hacer ; del Pozo Collado, Sandra ; Pfaar, Oliver ; Raskopf, Esther ; Zielen, Stefan ; Cuevas, Mandy ; Klimek, Ludger ; Casanovas, Miguel ; Acikel, Cengizhan ; Subiza, José Luis ; Mösges, Ralph ; Decker, Lea ; Allekotte, Silke
438
项与 DNA 相关的新闻(医药)2025-05-01
·抗体圈
摘要: 抗体-药物偶联物(ADC)包括抗体、接头和药物,它们通过抗体和表面抗原之间的特异性结合将其高效小分子药物引导至靶向肿瘤细胞。应正确设计或选择抗体、接头和药物,以达到所需的疗效,同时最大限度地减少脱靶毒性。由于结构独特而复杂,与产品相关的变化和制造过程会引入固有的异质性。本文主要介绍了ADC历史、临床开发状态、分子设计、制造工艺和质量控制方面的最新关键进展。制造过程,尤其是偶联过程,在其整个生命周期中应仔细开发、表征、验证和控制。质量控制是确保产品质量和患者安全的另一个关键要素。以患者为中心的策略已得到制药行业治疗性蛋白质的广泛认可和采用,并且也已成功应用于ADC,以确保ADC产品在保质期结束前保持其质量。对产品的深入了解和过程知识定义了属性测试策略(ATS)。质量源于设计(QbD)是用于流程和产品开发以及定义整体控制策略的强大方法。最后,我们总结了当前ADC开发面临的挑战,并提供了一些可能有助于给出相关方向并引发更多跨职能研究以克服这些挑战的观点。1.ADC药物概要 第一个ADC药物于2000年获得批准。然而,14种ADC药物中有9种在近 4 年内获得批准。ADC药物开发简史和关键事件 流通的ADC药物通过mAb部分与特定的肿瘤细胞表面抗原结合。结合的ADC药物被内化到内体的肿瘤细胞中。ADC药物从肿瘤细胞中回收或通过接头切割或mAb消化释放药物。释放的药物诱导细胞凋亡或杀死靶向微管或DNA的肿瘤细胞。释放的药物也可能通过渗透邻近细胞来诱导旁观者效应。ADC作用机制图截至2023年1月全球上市的ADC药物2.抗体-药物偶联物的关键要素2.1抗体-药物偶联物的抗原靶标 一个好的ADC抗原靶标具有相似的抗原特性,通常考虑以下方面: (1)抗原靶标的表达和丰度:抗原靶标在肿瘤组织中特异性高表达,在正常健康组织中低表达或无表达。理想的靶抗原应该是非分泌性的,不易从肿瘤细胞表面脱落,并且可溶性形式很少或没有可溶性。确定ADC靶标的抗基因选择性的关键不是肿瘤组织和健康组织中的绝对表达水平,而是表达比。ADC处理后,不应下调靶抗原的表达,以避免靶抗原表达下调介导的ADC耐药。(2)内化特性:抗原靶点内化的速度和水平是重要的参数,直接影响肿瘤细胞对ADC的吸收和释放。同时,非内部化ADC还可以通过有毒小分子的“旁观者效应”发挥治疗作用。(3)抗原共表达和协同效应:ADC可能面临诸如抗原靶点表达低的患者肿瘤,因此对治疗反应小,以及靶向非肿瘤毒性安全问题等问题。双特异性 ADC 结合了双特异性抗体和ADC的优点,为这个问题提供了新的方向。两种抗原靶标的共表达在肿瘤组织中相对常见,但在正常组织中非常罕见。双特异性抗体ADC可以更特异性地靶向肿瘤细胞,理论上可以减少靶向非肿瘤毒性并改善用药窗口 。其次,通过交联促进两种抗原的协同作用,增强内窥镜,通过减少细胞膜上受体蛋白的表达,可以进一步抑制肿瘤细胞生长信号,更有效地杀死肿瘤细胞,同时提高毒素进入肿瘤细胞的效率。2.2抗体-药物偶联物中的抗体 抗体是ADC的重要组成部分,通常考虑以下几个方面: (1)抗体的特异性和亲和力:理想的抗体需要高特异性,但也需要适当的亲和力以增加肿瘤部位的ADC通透性。优化抗原和抗体之间的合理亲和力是平衡ADC在靶细胞中的快速吸收和抗癌效用的关键点。(2)抗体的内化效率:大多数ADC在与靶抗原结合时被抗体介导的内吞有效内化。因此,在抗体选择中,首选具有高效和快速内吞活性的抗体。一些研究正在探索非内化ADC靶向肿瘤细胞微环境结构成分的可能性。这种方法可通过靶向肿瘤基质内高表达的抗原来克服实体瘤穿透障碍 。(3)分子类型和Fc功能:已上市和临床开发的ADC药物中使用最广泛的亚型是IgG1。ADC的Fc有效功能的丧失在某种程度上是有利的。ADC药物与效应细胞的结合可能会减少它们在肿瘤部位的积累,阻碍它们进入细胞,或导致对正常细胞的毒性增加。除了经典的全长单克隆抗体外,ADC的抗体部分还使用双特异性抗体和不同形式的小分子抗体。双特异性ADC提供更高的肿瘤靶向性,两种抗原的协同内吞作用提高了ADC的疗效,同时提高了安全性。为了获得更多的肿瘤渗透性ADC,新型ADC分子的设计策略已经转向不同的小分子抗体形式,包括Fab药物偶联物、scFv药物偶联物和单链抗体(VHH)偶联药物 。这些新型小分子抗体ADC比传统ADC具有更小的分子量和更好的肿瘤渗透性。然而,这些较小的形式可能与较短的半衰期和较快的清除率有关。(4)药物形成和药代动力学特性:抗体与毒性小分子偶联后,由于小分子毒物的强疏水性,ADC分子的特性可能会发生变化,因此抗体分子需要具有较好的药物凝固性。新一代ADC越来越多地使用免疫原性显著降低的全人源化抗体。ADC表现出与抗体相似的药理学特征。因此,理想的抗体选择还应考虑低免疫原性并保持最佳PK特性。2.3抗体-药物偶联物中的接头 ADC中的接头在将抗体连接到有效的小分子中发挥作用,是影响药物的关键元素稳定性、安全性和有效性 。接头需要在循环中保持稳定,并在进入肿瘤细胞后迅速释放负荷 。它主要分为可裂解型和非裂解型。此外,可切割类型进一步分为化学切割型和酶裂型。 不可切割的连接子具有良好的循环稳定性,降低了脱靶毒性,但它们需要进入肿瘤细胞的溶酶体,在那里被蛋白酶降解以释放负载复合物,释放率低。负载复合物通常由氨基酸 - 接头 - 有效载荷组成。它的渗透性很差,因此引发旁观者效应的能力有限。化学裂解接头主要包括酸裂解型和还原型裂解型。化学裂解接头从裂解中迅速释放,但它们在循环中的稳定性差,容易导致脱靶毒性 。酶裂解接头是使用最广泛和研究最广的接头。它们主要包括组织蛋白酶B、β-葡萄糖醛酸酶和硫酸酯酶裂解型。在这些类型中,组织蛋白酶B裂解型是最常用的。组织蛋白酶B是一种在各种癌细胞中过表达的溶酶体蛋白酶。此外,它可以优先识别PL、VC、VA和GGFG等特定氨基酸序列,并在这些序列的C端裂解肽键。这种接头既具有良好的循环稳定性,又具有在肿瘤细胞中快速裂解和释放毒素的能力。2.4抗体-药物偶联物中的细胞毒载量 细胞毒载量对ADC的作用至关重要。已知的毒性负荷通常包含微管蛋白抑制剂和DNA抑制剂。通常活性强,水溶性好,稳定性好。在有效杀伤肿瘤细胞的同时,还应降低对正常细胞的杀伤作用。除了传统的有毒负载外,还有许多创新负载正在开发和应用。微管破坏毒素 微管破坏毒素主要通过与微管蛋白结合来达到破坏细胞有丝分裂的目的,导致微管持续伸长或阻止微管聚合。它们主要包括奥司他汀、美登素类药物和艾日布林。特别是MMAE因其强大的活性和膜通透性而被广泛使用。DNA损伤毒素 DNA损伤毒素主要包括DNA片段化型、DNA烷基化型和拓扑异构酶 I/II抑制因子。DNA 损伤毒素不受细胞周期影响,半衰期短。它们不易积累,具有广泛的抗癌谱和指示。常用的毒素包括PBD、多利卡霉素、阿奇霉素、SN38和DXd,其中拓扑结构酶I抑制剂DXd是临床实践中最有效的毒素。从这种毒素开发的HER2 ADC已成为改变乳腺癌诊断和治疗的划时代产品。创新毒素 除了常规的毒性负荷外,还有各种创新的负荷产生应用,包括TLR7/8 激动剂、STING激动剂、Bcl-xL抑制剂和裂粒体抑制剂。2.5偶联 偶联技术对于ADC的均一性、稳定性和药代动力学至关重要。这些技术分为两大类:非定点偶联和定点偶联 。非定点耦合:赖氨酸偶联和半胱氨酸随机偶联 最早的非定点耦合技术是赖氨酸偶联技术,其偶联选择相对较差,均一性不足,对ADC的PK/PD有影响。该方法用于市售Kadcyla、Mylotarg 和 Besponsa的偶联。半胱氨酸随机偶联将偶联位点减少到8个,从而显着提高了偶联稳定性和均一性。这是目前使用最广泛的共轭结合方法。市售的Polivy、Padcev和Adcetris使用这种方法进行偶联 。一般来说,非定点偶联产物是稳定性差、易聚集、治疗窗相对较窄的混合物。定点偶联:半胱氨酸定点偶联、天然抗体定点偶联和抗体定点工程修饰 定点偶联可实现同质、更稳定的ADC并扩大治疗窗口。定点结合技术可分为以下几类:半胱氨酸的定点结合:(1)饱和结合:第一三共的 Enhertu在饱和结合方法中占据所有结合位点,以实现定点结合。(2) 硫瘤单抗:基因泰克将赫赛汀轻链第110位的缬氨酸突变为半胱氨酸,并特异性地与马来酰亚胺接头反应。Sevie、ImmunoGen、Abbagen、Pfizer和其他公司也有类似的技术。(3)桥接:Abzena/Poly therics开发了一种双苯磺酸接头,可以与两个游离硫醇连接,以获得药物抗体比(DAR)值为4的均质样品,并提高了ADC的稳定性。荣昌的triallytriazine连接剂也具有类似的作用。天然抗体的定点偶联:(1)赖氨酸的定点偶联:在特定的pH条件下,由于位置和微环境的差异,抗体上的赖氨酸与接头的反应速率不同。科伦生物的A166是基于该方法开发的HER2定点偶联ADC。(2)糖基修饰的定点偶联:天然抗体的N297位点有不同类型的糖基修饰。Synaffix的GlycoConnect技术使用甘油苷酶和糖基转移酶修饰,将叠氮化物基团连接到 N-乙酰氨基葡萄糖上。这可以触发与环辛炔的点击化学反应,形成DAR值为2的位点导向偶联ADC。此外,中国科学院上海药物研究所发现,内切糖苷酶Endo-S2可在水解抗体的N297位点的同时,将N-乙酰乳糖胺转移到去糖基化位点。这实现了一步法糖定点偶联 。抗体的定点工程修饰:(1)非天然氨基酸偶联:Ambrx的EuCODE技术允许将对乙酰苯丙氨酸 (pAcF)精确插入抗体中定义的位置,通过肟键进行定点偶联。Sutro Biopharma的Xpress CF技术将对叠氮甲基苯基羊毛氨酸(pAMF)引入抗体中,并通过点击化学反应进行定点偶联。(2)肽酶偶联:为了达到定点偶联的目的,酶识别抗体的特定位置。以下技术具有代表性:NBE Therapeutics的Sortase技术、Redwood Bioscience的SMARTag技术、LegoChem的ConjuAll技术和辉瑞的BTG 技术。3.抗体偶联物的生产 为了建立可靠且可重复的ADC制造工艺,我们根据上市前风险评估的概念,使用实验设计(DoE)进行实验设计。确定对药物质量属性有影响的工艺参数,控制工艺开发和生产过程中的关键工艺参数,确定合理的设计空间,不仅可以确保生产按预期进行,还可以获得质量可控的ADC产品。整个ADC生产过程一般包括单克隆抗体制备(细胞扩增和蛋白表达阶段、蛋白质纯化阶段)、接头和小分子药物制备、偶联和最终产物制备(图3)。基于抗体药物生物大分子的特殊属性,除了抗体的质量属性、工艺特性和过程控制外,还需要关注ADC生产过程中的工厂、设备、设施和污染控制。【ADC工艺开发/生产/风险等内容请参考原文】图3:ADC生产过程的流程图mAb部分是通过典型的单克隆抗体生物制造工艺生产的,包括细胞培养和纯化步骤。抗体中间体(AI)与通过化学合成或生物制造工艺生产的药物偶联。偶联过程是mAb与接头药物(例如半胱氨酸碱基)之间的化学反应或mAb和接头之间的后续反应,然后是与药物的反应(例如赖氨酸定向)。所得 ADC 原料药经过纯化,并在适用的情况下通过无菌灌装和冻干工艺进一步生产成药品。4.抗体-药物偶联物的质量控制 控制策略包括一组基于当前产品和过程理解的计划控制措施,以确保过程性能和产品质量。该规范是整体控制策略的一部分,其定义为测试列表、分析程序的参考和适当的验收标准。选择基本特性是为了确认原料药和药品的全部质量,而不是建立完整的表征。因此,应采用系统性方法来设定适当的规范。为生物治疗产品和杂质设定商业规范的方法已经建立并得到业界的广泛认可。这种方法强调基于产品知识的基于风险的方法,以确保患者的安全性和有效性。已经发布了一种系统方法来识别和评估关键质量属性(CQA)这些CQA包括产品变体、与工艺相关的杂质、强制性CQA、原材料和可浸出化合物。使用代表性材料在抗体、小分子或DS水平表征产品,以获得对产品物理化学和生物特性的深入了解至关重要。色谱和电泳技术、光谱技术和质谱法广泛用于物理化学表征,而结合测定、ELISA和基于细胞的生物分析则广泛用于生物表征。ADC产品的典型CQA如图4所示。CQA的可接受考虑范围基于临床经验,以及来自特定产品临床研究、先验知识、文献和法规的信息,其中考虑了工艺能力和方法可变性。属性测试策略基于过程能力和产品稳定性。这些是确定是否应在过程控制(IPC)、发布或稳定性期间测试CQA以及哪些CQA最适合测试的有益步骤。图4:抗体、小分子、原料药和药品的典型CQA。小分子包括接头、药物或接头-药物复合物(如适用)ADC的CQA4.1药物抗体比(DAR) DAR对产品安全性、有效性和PK有直接影响。它在DS工艺 中使用适当的分析方法进行控制和测试。应进一步评估DS稳定性、药品(DP)过程和稳定性影响,以确定是否应在这些阶段测试DAR。验收标准在很大程度上依赖于特定于产品的临床经验。目前,包括紫外-可见分光光度法、疏水相互作用色谱法(HIC)、反相色谱法(RPLC)和尺寸排阻色谱-质谱法(SEC-MS)在内的方法通常用于表征制造过程和质量控制中的DAR。紫外-可见分光光度法可以利用Drug-Linker和mAb的不同最大吸收波长来确定DAR,但无法获得DAR分布信息,并且容易受到游离药物的干扰。HIC 可以根据疏水差异分离不同的DAR分子,然后获得分析DAR和DAR 分布。作为一种天然分析方法,HIC常用于Cys偶联的ADC。然而,该方法受到高DAR样品的挑战,并且对DAR6、DAR7和DAR8的分离度很差。RPLC可以在变性下分离不同的DAR分子或相关亚基条件,并且其分辨率通常优于HIC。然而,在RPLC 条件下,Cys偶联的ADC会解离成LC和 HC,因此只能获得LC和HC上的平均DAR和药物分布。当使用非变性分离条件、软电离和高分辨率质谱(HRMS)时,SEC-MS对不同的DAR表现出优异的分辨率,并与大多数ADC兼容。因此,SEC-MS越来越多地用于早期开发和ADC表征。4.2偶联位点分布和非偶联mAb部分 ADC产品具有异质性,并且包含具有不同偶联药物数量和不同偶联位点的分子,这可能会影响安全性和有效性。异质性的程度在很大程度上取决于ADC平台。根据目前的知识,赖氨酸导向、链间导向的半胱氨酸和位点特异性偶联平台按降序排列。偶联位点分布和非偶联的mAb部分连接可与DAR相关,用于产物特征化和通过DAR测试进行受控表征。4.3游离药物 游离药物,也是一种非偶联药物,具有潜在的全身毒性。尽管游离药物可以有多种形式,例如药物或连接药物,但它们通常被集体处理。游离药物的验收标准是根据其毒性设定的,同时考虑到工艺的清除能力。在DS释放期间需要控制游离药物。此外,根据ADC产品在DS存储、DP工艺和 DP存储期间的稳定性,也需要在这些阶段进行测试。4.4残留溶剂 有机溶剂通常用于DS生产中,以溶解疏水性细胞毒素。应根据溶剂的毒性和生产过程的去除能力确定残留溶剂的可接受标准。残留溶剂通常在DS放行时进行测试,其他阶段的测试要求应根据残留溶剂对DS稳定性、DP工艺和 DP稳定性的影响来确定。4.5大小变体 大小变体通常包括片段和聚集体。聚集体通常以可解离和不可解离的形式存在。DP在保质期结束时的验收标准应确保疗效和患者安全,这主要基于产品特定的临床经验。根据工艺和稳定性影响进行逆向计算,为抗体即刻(AI)、DS和DP阶段设置适当的验收标准。低分子量型(LMW) 可以通过肽键或链间切割形成。可以使用相同的策略为聚集体设置适当的 LMW标准,但要特别注意ADC片段可能携带偶联药物并引起脱靶毒性。因此,LWM形式应在安全水平以下进行表征和控制。4.6电荷异构体 电荷异构体常见于抗体和ADC中。修饰,包括脱酰胺、天蕨酸异构化、糖基化和 C端赖氨酸,可导致电荷转移和电荷形式变体。应根据产品特定的临床经验,单独评估和设置适当的验收标准。对于某些ADC,在共轭后测试电荷变化可能具有挑战性,因为共轭可能会导致电荷偏移。在这种情况下,产品知识和过程理解有助于使用适当的分析方法制定适当的控制策略。如果产品在代表性的DS和DP工艺和储存条件下保持稳定,则在AI级别进行测试可能足以控制电荷变体。4.7其他形式的变体 根据产品特性和ADC平台,需要考虑一些变体。这些包括但不限于由氧化引起的变体、序列变体、游离二硫键和硫醚。这些属性可能会影响产品的安全性或有效性,或者可能会影响ADC产品质量。例如,三硫键可能不是mAb的CQA,但由于它对DAR的影响,它对链间半胱氨酸ADC平台至关重要。应根据具体情况评估这些不同的蚂蚁。4.8数量或强度 蛋白质含量通常包含在 DS 和 DP 放行中,以确保产品量正确。4.9效价 效价方法应通过反射动作来定义。对于ADC产品,它们主要用于杀死癌症患者的肿瘤细胞。基于细胞的生物测定法有望用于效价测试。这些 生物检定表示也应该具有稳定性,如方法验证所证明的那样。我们必须考虑方法的可变性,以便设置适当的验收标准。4.10糖基化修饰 众所周知,糖基化模式可能会影响效率、安全性或PK。对于ADC产品,mAb的主要作用是将药物递送至靶细胞,因此糖基化可能不会影响疗效。如果存在可能受糖基化模式影响的继发性作用机制(MOA),例如抗体依赖性细胞介导的细胞毒性(ADCC)、抗体依赖性细胞介导的吞噬作用(ADCP)或补体依赖性细胞毒性(CDC),则应充分表征ADC产物。在这种情况下,糖基化可以作为CQA进行评估,并包含在 AI IPC或放行测试中,具体取决于工艺能力。4.11接头、药物或接头-药物 小分子药物中间体(包括接头、药物和接头药物)的质量控制与小分子药物活性药物成分(API)的质量控制相似,特别考虑了其对ADC产品的影响。4.12纯度 纯度是典型的CQA。更广泛的验收标准取决于其对ADC产品质量和 DS工艺的影响。如果接头或接头药物被过度添加而对DAR没有影响,并且可以在整个过程中有效地去除,则可以证明验收标准是合理的。4.13可偶联杂质 可偶联杂质具有反应性官能团,因此它们可以与AI反应生成副产物。这种副产品很难去除,因为这些蛋白质材料很难与ADC产品分离。因此,在小分子生产阶段对可偶联杂质进行控制和测试。4.14不可偶联杂质 不可偶联杂质没有官能团,不能与mAb连接。这些杂质被控制在低水平,以最大限度地降低对患者的潜在风险。此类杂质可以在小分子阶段进行控制和测试。4.15其他与工艺相关的杂质 其他工艺杂质可以从各种来源进入最终产品,例如DS工艺中残留的重金属。应单独评估每种杂质以识别CQA。应根据临床经验、先验知识或法规要求将这些杂质控制在安全水平以下。过程能力 是设置测试策略的关键考虑因素。如果该工艺已被证明可以有效去除杂质,则质量控制测试将增加最低价值。4.16表面活性剂含量 通常将表面活性剂添加到配方中以保护产品并提高稳定性。表面活性剂含量的验收标准是根据配方开发数据设定的。测试策略取决于制造工艺和储存条件下表面活性剂的稳定性。4.17其他质量属性 还有其他质量属性,如药典要求,包括颜色、透明度、内毒素水平、纯度、可见颗粒、亚可见颗粒、pH、渗透压、可提取体积、固体剂量的填充重量、再溶解时间和冻干的水分。可接受性可以根据药典要求设置,也可以根据配方开发进行设置。现代过程控制可能比最终产品测试更强大。一个示例是在线填充重量,它将提供比最终产品填充重量或可萃取体积测试更强大的过程控制。可浸出物可能来自不同的来源,例如制造过程 或容器密封系统,它们包括有机杂质和元素杂质。这些杂质通常在过程代表性条件或储存条件下进行表征,以证明杂质处于安全水平。ICH指南,如ICH Q3系列,提供了有关安全水平的详细信息。浸出物通常或不一定包含在AI、DS或DP测试中,因为水平通常得到很好的控制。5.结论 ADC生产包括抗体生产、接头和药物化学或生物合成、偶联和药品的最终制剂。与传统的单克隆抗体药物相比,偶联过程存在特殊挑战。偶联过程取决于所选的连接子和药物,以及要偶联的氨基酸。本文说明了已上市ADC药物的常用工艺,但并非全部包括制造工艺。来自开发活动、工艺验证、临床生产、文献和先验知识的理解知识有助于确定关键工艺参数和控制措施。ADC药物的质量控制策略是根据对工艺的理解和产品知识制定的。以患者为中心的方法被广泛用于根据临床经验和法规要求定义产品规格。考虑到工艺能力和产品稳定性,我们可以定义质量控制策略,包括要测试的质量属性、在哪个阶段以及适当的验收标准。因此,定义了一个总体控制策略,以确保制造过程始终如一地提供具有适当安全性和有效性的 ADC产品。 在过去的20年里,ADC的发展是由对替代和有效肿瘤治疗的高度未满足的临床需求推动的。第三代ADC的成功为ADC研究领域提供了更令人鼓舞的前景。全球ADC药物市场具有巨大的增长潜力。目前,全球共有15种ADC获批上市(13项FDA批准、1项中国批准和1项日本批准),还有更多ADC正在临床开发中。ADC的治疗潜力正在通过临床适应症扩大,从血液肿瘤到实体瘤,从单一疗法扩展到联合疗法(如常规化疗、免疫检查点抑制剂、靶向不同抗原的单克隆抗体以及小分子抑制剂)。近年来,寻找新型靶点、选择新型抗体形式、选择合适的有效载荷以及设计具有良好释放机制的连接子,成功开启了ADC药物在肿瘤以外的疾病中的应用,如自身免疫性疾病和感染性疾病 。在其他领域,这些应用也将进一步扩大 ADC 的临床应用。 识别微信二维码,添加抗体圈小编,符合条件者即可加入抗体圈微信群!请注明:姓名+研究方向!本公众号所有转载文章系出于传递更多信息之目的,且明确注明来源和作者,不希望被转载的媒体或个人可与我们联系(cbplib@163.com),我们将立即进行删除处理。所有文章仅代表作者观点,不代表本站立场。
抗体药物偶联物
2025-04-30
·肿瘤界
点击蓝字 关注我们引用本文:李明珠,赵超,魏丽惠.人乳头瘤病毒疫苗应用与宫颈癌筛查分流进展[J/CD].肿瘤综合治疗电子杂志,2025,11(2):152-157.据统计,2022年全球宫颈癌新发病例约66.2万例,死亡病例约34.9万例,占全球女性癌症发病和死亡总数的6.8%和8.1%,宫颈癌是女性发病率和死亡率第4大常见癌症[1]。我国同样面临着严峻的宫颈癌疾病负担,2022年我国宫颈癌新发病例约15.1万,死亡病例约5.6万,占全球发病和死亡总数的22.8%和16.0%,且近20年来我国发病率和死亡率呈现上升趋势,到2022年世标发病率和世标死亡率分别为13.83/10万和4.54/10万[2]。同时,发病年龄呈年轻化趋势,2000—2014年宫颈癌发病率的峰值年龄段由>70岁逐渐降为40~49岁,农村地区标化平均发病年龄下降5.18岁,降幅明显大于城市地区[3]。以上数据表明,我国仍需付出巨大努力来消除宫颈癌。目前,我国在宫颈癌防治上的进展主要集中在人乳头瘤病毒(human papilloma virus,HPV)疫苗应用和宫颈癌筛查分流2个方面。1预防性人乳头瘤病毒疫苗接种进展现状1.1推动适龄女性接种预防性HPV疫苗我国高度重视宫颈癌防治工作,《“健康中国2030”规划纲要》《中国妇女发展纲要(2021―2030年)》等重大规划纲要均将宫颈癌防治工作作为重要内容,明确目标为进一步完善宫颈癌防治服务体系,提高综合防治能力,构建社会支持环境,努力遏制宫颈癌发病率和死亡率上升趋势,减轻宫颈癌社会疾病负担。2023年1月,国家卫生健康委员会等10部门联合发布的《加速消除宫颈癌行动计划(2023—2030年)》[4],提出了我国2025、2030年消除宫颈癌主要目标:到2025年,试点推广适龄女生HPV疫苗接种服务,适龄女性宫颈癌筛查率达到50%,宫颈癌及癌前病变患者治疗率达到90%;到2030年,持续推进适龄女性HPV疫苗接种试点工作,适龄女性宫颈癌筛查率达到70%,宫颈癌及癌前病变患者治疗率达到90%。世界卫生组织(World Health Organization,WHO)提出《加快消除宫颈癌全球战略》,其中明确提出到2030年,全球90%的9~14岁女孩应完成HPV疫苗接种。截至2025年2月4日,全球已有146个国家和地区(约占75%)将预防性 HPV疫苗纳入国家免疫接种计划[5]。我国HPV疫苗的覆盖率也相对较低,虽然全国首剂HPV疫苗覆盖率已从2017年的0提升至2022年的10.15%,但3剂次全程接种率仅为6.21%。9~14岁女孩群体的首剂覆盖率仅为4%,3剂次覆盖率更低,仅0.31%。15~19岁女性的首剂覆盖率为4.66%,3剂次覆盖率同样仅0.31%[6]。相比全球的HPV疫苗覆盖率(根据WHO发布的数据,全球9~14岁女孩接种第1剂HPV疫苗的覆盖率从2022年的20%上升至2023年的27%)[7],我国的接种水平仍存在较大差距。近年来,我国在推动健康城市建设的过程中,大力推进HPV疫苗的接种工作。自2017年中国正式启动适龄女性HPV疫苗接种以来,每年的接种剂量和覆盖率均呈持续增长趋势。自2021年以来,广东省、海南省、福建省、江苏省、江西省、重庆市、西藏自治区、浙江省、山东省、湖北省和广西壮族自治区已为本地区13~14岁的适龄女孩提供免费的HPV疫苗接种服务。截至2024年10月,HPV疫苗免费接种这一政策已经覆盖了全国约40%的适龄女孩,已有500万适龄女孩接种了HPV疫苗[8]。1.2扩大HPV疫苗接种年龄和男性接种HPV疫苗1.2.1 在9~14岁女孩接种2剂次HPV疫苗2024年,国家药品监督管理局依据国内临床研究数据,批准了HPV四价疫苗在9~14岁女孩接种2剂次[9]。自此,我国现有的5种HPV疫苗,在9~14岁女孩均可以接种2剂次。对于小年龄组采用2剂次的免疫接种计划,从促进适龄女性疫苗接种的普及,到卫生经济学的效益考量,均将极大地推动我国HPV疫苗接种工作的进程,为更多女性的健康提供坚实保障。1.2.2 男性HPV感染现状及HPV疫苗接种相关问题1.2.2.1 男性HPV感染情况男性HPV感染趋势与女性不同。在女性中,HPV感染率在第1次性行为后会达到顶峰,并随着年龄的增长而不断下降。一项纳入全球65项研究,涵盖35个国家共44769名≥15岁男性的Meta分析显示,全球男性任意型别HPV的总体感染率为31%,高危HPV的感染率为21%。HPV感染率在25~29岁年轻成年男性中达到峰值,任意型别HPV、高危型HPV和HPV 16的感染率分别为35%、24%和5%,且在整个成年期均居高不下,甚至延续至50岁。此外,15~19岁的青少年中HPV感染率同样较高,任意型别HPV和高危型HPV的感染率分别为28%和20%,且男性整个生命周期内感染HPV的风险均较高[10]。我国男性普通人群HPV感染率为8.0%~16.9%,其中高危型HPV感染率为5.5%~9.4%[11]。1.2.2.2 男-男性行为者(men who have sex with men,MSM)HPV感染情况一项Meta分析发现,HPV感染在MSM人群中非常普遍,肛门、阴茎、尿道和口腔的HPV感染率分别为78.4%、36.2%、15.4%和17.3%;人类免疫缺陷病毒(human immunodeficiency virus,HIV)阳性MSM人群中各部位HPV感染率均高于HIV阴性MSM人群[12]。一项纳入64项研究共29900名男性肛门HPV感染率的Meta分析显示,HIV阴性和HIV阳性MSM人群中高危型HPV感染率分别为41.2%和74.3%,其中HPV 16感染率分别为13.7%和28.5%[12]。MSM人群不同部位感染由高至低依次为肛门、阴茎和尿道;各部位常见HPV高危型别也不同,肛门最常见高危型分别为HPV 16、51、18、52、58、39型[13]。我国MSM人群中肛门HPV感染率为85.1%(HIV阳性)、53.6%(HIV阴性)和59.2%(未知HIV状态)。最常见感染型别为HPV 6、11、16、18、52、58。HIV阳性MSM人群多重感染率高于HIV阴性MSM(52.1%比21.9%);高危型HPV在我国北方更常见(41.2%),而低危型HPV在我国南方更常见(35.7%)。在HIV阴性MSM人群中,HPV的流行随着年龄的增长而增加[13]。1.2.2.3 男性HPV疫苗接种根据我国临床研究数据,国家药品监督管理局于2025年1月8日正式批准 9~26岁男性接种四价HPV疫苗。2025年4月14日我国九价疫苗也被批准用于16~26岁男性[14]。1.3关于HPV疫苗1剂次接种进展和我国策略当前的HPV疫苗接种方案建议适龄女性完成3剂次接种,而9~14岁女性可选择2剂次接种。然而,随着研究的深入,单剂接种方案的有效性逐步得到认可。2022年4月,WHO免疫战略咨询专家组(Strategic Advisory Group of Experts,SAGE)召开相关评估会议后指出,单剂HPV疫苗可提供可靠保护,与2剂或3剂接种方案效果相当,并建议将其作为9~20岁年轻女性的可选接种方案之一[15]。2022年12月,WHO基于SAGE建议正式更新立场文件,指出单剂HPV疫苗可以提供与2剂方案相当的持久保护。目前,单剂HPV疫苗在9~14岁女孩、成年女性及男性中的长期免疫原性、保护效力及有效性仍是研究重点,需进一步积累证据。研究表明,单剂HPV疫苗可有效诱导免疫反应,提供对初始及持续性HPV感染的保护,单剂接种者的血清抗体阳转率并不低于2剂或多剂接种者,9~14岁女孩接种单剂HPV二价疫苗后12个月和24个月,HPV 16和HPV 18抗体血清阳转率高达98.6%~99.32%[16],且在接种后11年内抗体水平保持稳定,无明显下降趋势[17]。单剂四价HPV疫苗接种后第10年,97.8%的受试者仍可检测到HPV 16中和抗体,且抗体水平保持稳定,无明显衰减[18]。单剂九价疫苗接种12个月和24个月后,HPV 16和HPV 18抗体血清阳转率为96.3%~100%,9~14岁女孩接种单剂九价HPV疫苗可产生长达2年的强烈免疫应答[15]。在我国,由于尚缺乏单剂次接种的研究数据,目前仍按照国家药品监督管理局批准的说明书推荐HPV疫苗3剂次接种方案,9~14岁女孩可采用2剂次接种方案。2治疗性人乳头瘤病毒疫苗研究进展治疗性HPV疫苗通过抗原呈递细胞激活特异性细胞免疫[19]。其机制是打破慢性感染者体内的免疫耐受,重建或增强患者的免疫应答机制,消除病原体和癌变细胞,阻断癌细胞的扩散与转移。与预防性HPV疫苗主要依赖体液免疫(中和抗体)不同,治疗性HPV疫苗旨在诱导特异性细胞免疫。根据抗原递送形式,治疗性HPV疫苗分为多肽疫苗、蛋白疫苗、细菌载体疫苗、病毒载体疫苗、细胞疫苗和核酸疫苗(包括DNA和RNA疫苗)。2024年WHO制定了《治疗性HPV疫苗的优选产品特性》(PPCs)[20],旨在加快治疗性HPV疫苗的研发,为疫苗产品特征提供指导,该文件明确提出了用于清除HPV感染和用于逆转癌前病变2类优选产品特性。目前治疗性HPV疫苗仍处于临床研究阶段,部分候选疫苗(如VGX-3100)已取得一定进展。WHO提出,治疗性HPV疫苗可作为HPV感染及癌前病变的潜在治疗手段,并建议其适应证扩大至更广泛的人群,为全球消除宫颈癌提供创新策略。针对HPV感染和宫颈上皮内瘤变(cervical intraepithelial neoplasia,CIN)1的治疗性候选疫苗,旨在清除特异性高危型HPV亚型感染,并预防其向CIN 2/3甚至宫颈癌的进展。其主要临床终点包括以下内容。①HPV清除率或CIN 1消退率 :≥2次HPV检测阴性,或者CIN 1逆转。②疾病进展率:检测HPV感染是否进展至CIN 2/3。③次要临床终点:包括再感染率、CIN 1复发率、非疫苗HPV亚型的清除率及非宫颈部位(口咽、肛门、外阴、阴道等)的清除率。根据美国临床试验数据库(https://www.clinicaltrials.gov)统计,截至2024年2月21日(目前暂无更新),全球已完成或正在进行的治疗性HPV疫苗临床试验超过90项,涉及30余种候选疫苗。目前,全球尚无治疗性HPV疫苗被批准上市。目前全球已有7种候选治疗性HPV DNA疫苗进入临床试验,其中以HPV 16/18 E6/E7抗原为主要靶点进行研究[21]。我国也已开始治疗性HPV疫苗临床试验。未来,治疗性HPV疫苗的研发与推广将成为预防性HPV疫苗的重要补充,有望填补HPV感染治疗的空白,助力消除宫颈癌。3完善宫颈癌筛查及筛查异常分流的管理策略2024年11月,在已发布的《中国子宫颈癌筛查指南(一)》[22]的基础上,结合国内外最新研究证据和临床实践经验,由中国优生科学协会阴道镜和宫颈病理学分会、中华医学会妇科肿瘤学分会等7个学(协)会的专家共同制定了《中国子宫颈癌筛查指南(二)》[23]。该指南通过规范子宫颈癌筛查异常的分流策略,实现精细化管理,旨在避免过度诊断和治疗,同时降低对子宫颈高级别病变及早期子宫颈癌的漏诊。该指南介绍了当前几种新的筛查分流方法,以优化筛查流程并提高检测的精准性。3.1p16/Ki-67细胞学双染检测分流方法根据美国IMPACT前瞻性多中心临床研究结果,p16/Ki-67细胞学双染检测(以下简称双染)在HPV阳性个体的分流管理中优于传统细胞学分流。2024年3月,美国阴道镜与宫颈病理学会(American Society for Colposcopy and Cervical Pathology,ASCCP)推荐双染检测用于高危型HPV初筛或联合筛查中HPV阳性人群的分流管理[24]。结合国际研究数据和我国临床实践,该指南提出以下分流策略。①HPV不分型初筛:对高危型HPV阳性女性采用双重染色检测进行分流。②HPV分型初筛:HPV16/18阳性者直接转诊阴道镜,其他12种高危型HPV阳性(HPV 31/33/35/39/45/51/52/56/58/59/66/68)者进行双重染色分流。③联合筛查:无论HPV不分型阳性或12种高危型阳性,若细胞学结果正常或低度病变,可采用双重染色进行分流。需使用经国内外权威机构认可和临床试验验证可用于高危型HPV阳性人群分流的p16/Ki-67双染试剂。3.2DNA甲基化检测DNA甲基化是一种表观遗传学改变,涉及关键基因启动子区域CpG岛的甲基化,从而导致抑癌基因转录失活或基因组去甲基化,引发肿瘤[25]。已有研究证实,多种基因的高甲基化水平与子宫颈高级别CIN 2/3和子宫颈癌密切相关[26-27]。目前我国推荐在高危型HPV初筛后,采用DNA甲基化检测对其他12种高危型HPV阳性个体进行分流,以降低不必要的阴道镜的转诊率,但需选择经国内外权威机构认可和经临床试验证实可用于高危型HPV分流管理的甲基化检测试剂。3.3HPV基因整合检测HPV基因整合是指HPV感染宿主后,其致病基因E6/E7片段嵌入到子宫颈上皮细胞基因组DNA的过程,是HPV持续感染导致子宫颈癌的关键分子事件[28-29]。华中科技大学同济医学院附属同济医院马丁院士团队开发的HPV基因整合检测技术,通过液相捕获技术富集HPV基因及其整合位点,并结合高通量测序技术精确识别整合状态。在一项纳入1.2万例人群的前瞻性队列研究中,完成5年随访的4611例数据显示,与传统细胞学分流相比,HPV基因整合检测对CIN 3检出的敏感度和阴性预测值相似,但特异度更高,且可降低阴道镜转诊率[30]。另一项包含1393例HPV感染的医院筛查队列研究中,HPV基因整合检测相比传统细胞学检查,CIN 3检出的敏感度相当,但特异度更高[31]。对于HPV基因整合阳性者,尤其对于HPV 16/18阳性个体,应视为高风险人群,必要时行诊断性宫颈锥切术。整合阴性者建议1年后随访,且需选择经国内外权威机构认可和临床试验验证可用于高危型HPV阳性人群分流的基因整合试剂。3.4HPV拓展分型检测HPV拓展分型检测是在HPV 16和HPV 18分型检测外,对其他高危型HPV型别进行单一型别或相关型别分组检测。国际癌症研究机构根据HPV型别在子宫颈癌中的占比及CIN 3+的9年累积风险(Onclarity HPV临床试验数据),将高危型HPV分为最高风险(HPV 16)、高风险(HPV 18/45)、中风险(HPV 33/31/52/58/35)和较低风险(HPV 39/51/59/56/68)四组[32-33]。2025年1月ASCCP发布了关于HPV基因分型的永久指南,提出以下分流策略:①高风险(HPV 16/18),推荐直接转诊阴道镜。②中风险(HPV 45/33/58/31/52/35/39/68/51),需进一步分流,决定是否转诊阴道镜。③低风险(HPV 56/59/66),建议1年后随访。拓展分型检测的意义在于精准识别不同风险水平的HPV感染者,避免“一刀切”式的阴道镜转诊,从而减少过度诊断与治疗。4总结与展望WHO已将加速消除宫颈癌作为全球战略,中国作为人口大国,在推进HPV疫苗接种和研发的同时,需不断完善宫颈癌筛查体系,探索适合国情的防控策略。2024年,我国在优化宫颈癌筛查分流体系的基础上,进一步推动了HPV疫苗接种覆盖率的提升和筛查技术的规范化应用,双管齐下,提高了宫颈癌的早期预防和精准筛查能力,为尽早实现消除宫颈癌的目标奠定了坚实基础。参考文献[1] 王裕新, 潘凯枫, 李文庆.2022全球癌症统计报告解读[J/CD].肿瘤综合治疗电子杂志,2024,10(3):1-16.[2] HAN B, ZHENG R, ZENG H, et al. Cancer incidence and mortality in China, 2022[J]. J Natl Cancer Cent, 2024, 4(1):47-53.[3] LI X, ZHENG R, LI X, et al. Trends of incidence rate and age at diagnosis for cervical cancer in China, from 2000 to 2014[J]. Chin J Cancer Res, 2017, 29(6):477-486.[4] 国家卫生健康委,教育部,民政部,等.关于印发加速消除宫颈癌行动计划(2023—2030年)的通知[EB/OL].(2023-01-05)[20243-12-12]. https://www.gov.cn/zhengce/zhengceku/2023-01/21/content_5738364.htm.[5] World Health Organization. Immunization, vaccines and biologicals[EB/OL]. (2024-11-26)[2024-12-12]. https://www-who-int.libproxy1.nus.edu.sg/health-topics/vaccines-and-immunization/#tab.[6] World Health Organization. Human papillomavirus (HPV) vaccination coverage(2025-01-21)[2025-02-11]. https://immunizationdata-who-int.libproxy1.nus.edu.sg/global/wiise-detail-page/human-papillomavirus-(hpv)-vaccination-coverage?CODE=Global&ANTIGEN=HPV_FEM&YEAR=.[7] CHEN J, ZHANG Z, PAN W, et al. Estimated human papillomavirus vaccine coverage among females 9-45 years of age-China, 2017-2022[J]. China CDC Wkly, 2024, 6(19):413-417.[8] 央视网.国家卫健委:HPV疫苗免费接种政策已覆盖全国约40%适龄女孩[EB/OL].(2024-10-18)[2024-12-12].https://m.toutiao.com/article/7427028790305440295/?upstream_biz=doubao&show_loading=0&webview_progress_bar=1.[9] 丁香园用药助手.四价人乳头瘤病毒疫苗(酿酒酵母)说明书[EB/OL].[2024-12-12].https://drugs.dxy.cn/business/drug/ 153304.htm.[10] BRUNI L, ALBERO G, ROWLEY J, et al. Global and regional estimates of genital human papillomavirus prevalence among men: a systematic review and meta-analysis[J]. Lancet Glob Health, 2023, 11(9):e1345-e1362.[11] 靳大川,武淑芳,郭师,等.我国男性HPV流行病学特征的描述性评价[J].医学研究杂志,2018,47(6):104-108.[12] WEI F, GAISA M M, D'SOUZA G, et al. Epidemiology of anal human papillomavirus infection and high-grade squamous intraepithelial lesions in 29 900 men according to HIV status, sexuality, and age: a collaborative pooled analysis of 64 studies[J]. Lancet HIV, 2021, 8(9):e531-e543.[13] FARAHMAND M, MONAVARI S H, TAVAKOLI A. Prevalence and genotype distribution of human papillomavirus infection in different anatomical sites among men who have sex with men: a systematic review and meta-analysis[J]. Rev Med Virol, 2021, 31(6):e2219.[14] 默沙东(中国)投资有限公司官网.默沙东九价人乳头瘤病毒疫苗(酿酒酵母)在华获批上市男性适应证佳达修®9成为中国境内首个且目前唯一、可供适龄男性女性接种的九价HPV疫苗[EB/OL].(2025-04-14)[2025-04-15]. https://www.msdchina.com.cn/2025/04/14/company_news_04-14/.[15] World Health Organization. Strategic Advisory Group of Experts on Immunization (SAGE)-April 2022. (2022-04-07)[2024-12-12]. https://www-who-int.libproxy1.nus.edu.sg/news-room/events/detail/2022/04/04/default-calendar/sage_meeting_april_2022.[16] WATSON-JONES D, CHANGALUCHA J, WHITWORTH H, et al. Immunogenicity and safety of one-dose human papillomavirus vaccine compared with two or three doses in Tanzanian girls (DoRIS): an open-label, randomised, non-inferiority trial[J]. Lancet Glob Health, 2022, 10(10): e1473-e1484.[17] KREIMER A R, SAMPSON J N, PORRAS C, et al. Evaluation of durability of a single dose of the bivalent HPV vaccine: the CVT trial[J]. J Natl Cancer Inst, 2020, 112(10):1038-1046.[18] JOSHI S, ANANTHARAMAN D, MUWONGE R, et al. Evaluation of immune response to single dose of quadrivalent HPV vaccine at 10-year post-vaccination[J]. Vaccine, 2023, 41(1):236-245.[19] MORSE M A, GWIN W R, MITCHELL D A. Vaccine therapies for cancer: then and now[J]. Target Oncol, 2021, 16(2):121-152.[20] World Health Organization. WHO preferred product characteristics for therapeutic HPV vaccines[EB/OL]. (2024-07-04) [2024-12-12]. https://cdn-who-int.libproxy1.nus.edu.sg/media/docs/default-source/reproductive-health/cervical-cancer/who-ppc-therapeutic-hpvvaccines-public-comment.pdf?sfvrsn=dccee58_3.[21] 张秀军,赵超,魏丽惠.治疗性HPV疫苗的研发和临床应用展望[J].中国医药导刊,2024,26(8):743-747.[22] 中国优生科学协会阴道镜和子宫颈病理学分会,中华医学会妇科肿瘤学分会,中国抗癌协会妇科肿瘤专业委员会,等.中国子宫颈癌筛查指南(一)[J].肿瘤综合治疗电子杂志,2023,9(3):41-48.[23] 中国优生科学协会阴道镜和宫颈病理学分会,中华医学会妇科肿瘤学分会,中国抗癌协会宫颈癌专业委员会,等.中国子宫颈癌筛查指南(二)[J].现代妇产科进展,2025,34(1):1-9.[24] CLARKE M A, WENTZENSEN N, PERKINS R B, et al. Recomm endations for use of p16/Ki67 dual stain for management of individuals testing positive for human papillomavirus[J]. J Low Genit Tract Dis, 2024, 28(2):124-130.[25] SCHLESINGER Y, STRAUSSMAN R, KESHET I, et al. Polycomb-mediated methylation on Lys27 of histone H3 premarks genes for de novo methylation in cancer[J]. Nat Genet,2007, 39(2):232-236.[26] SU P H, LAI H C, HUANG R L, et al. Paired box-1 (PAX1) activates multiple phosphatases and inhibits kinase cascades in cervical cancer[J]. Sci Rep, 2019, 9(1):9195.[27] WENTZENSEN N, SHERMAN M E, SCHIFFMAN M, et al. Utility of methylation markers in cervical cancer early detection: appraisal of the state-of-the-science[J]. Gynecol Oncol, 2009, 112(2):293-299.[28] ZHOU L, QIU Q, ZHOU Q, et al. Long-read sequencing unveils high-resolution HPV integration and its oncogenic progression in cervical cancer[J]. Nat Commun, 2022, 13(1): 2563.[29] CAO C, XU Q, ZHU Z, et al. Three-dimensional chromatin analysis reveals Sp1 as a mediator to program and reprogram HPV-host epigenetic architecture in cervical cancer[J]. Cancer Lett, 2024, 588:216809.[30] TIAN X, WENG D, CHEN Y, et al. Risk assessment and triage strategy of cervical cancer primary screening on HPV integration status: 5-year follow-up of a prospective cohort study[J]. J Natl Cancer Cent, 2024, 4(4):311-317.[31] HU T, LI K, HE L, et al. Testing for viral DNA integration among HPV-positive women to detect cervical precancer: an observational cohort study[J]. BJOG, 2024, 131(3):309-318.[32] IARC. Cervical cancer screening[EB/OL]. (2022-08-26)[2024-12-12]. https://publications.iarc.fr/Book-And-Report-Series/Iarc-HandbooksOf-Cancer-Prevention/Cervical-Cancer-Screening-2022.[33] PERKINS R B, WENTZENSEN N, GUIDO R S, et al. Cervical cancer screening: a review[J]. JAMA, 2023, 330(6):547-558.推荐阅读● 指南 | 预防性人乳头瘤病毒疫苗中国临床应用指南(2025 版)● 国际HPV知晓日 | 解码HPV疫苗的科学力量声明:本文由“肿瘤界”整理与汇编,欢迎分享转载,如需使用本文内容,请务必注明出处。编辑:lagertha审核:南星
疫苗临床研究
2025-04-26
·抗体圈
摘要:抗体 - 药物偶联物(ADCs)作为一类靶向癌症治疗药物,凭借单克隆抗体的特异性将细胞毒性有效载荷精准输送到肿瘤细胞,减少对正常组织的损伤。本文回顾了 ADCs 的发展历程,详细介绍其组成部分、设计原理、作用机制以及在临床试验中的表现。同时探讨了面临的挑战,如脱靶毒性、耐药性和蛋白质聚集等问题,并提出相应的解决策略。ADCs 在癌症治疗领域优势显著,未来通过技术创新和联合治疗有望取得更大突破,为患者带来更多希望 。一、ADCs:癌症治疗的 “魔法子弹”癌症是全球健康的重大威胁,传统化疗药物虽能治疗癌症,但存在治疗窗口窄、副作用大的问题 。抗体 - 药物偶联物(ADCs)应运而生,它由单克隆抗体、细胞毒性有效载荷和连接子组成,能像 “魔法子弹” 一样,精准地将细胞毒性药物输送到肿瘤细胞,减少对正常细胞的伤害 。ADCs 的发展经历了多个阶段,早期产品存在诸多问题,不过随着技术进步,如今已取得显著进展,在癌症治疗中展现出巨大潜力 。二、ADCs 的关键组成与设计单克隆抗体:精准导航仪:单克隆抗体在 ADCs 中起着关键的靶向作用 。目前常用的抗体骨架是免疫球蛋白 G(IgG),尤其是 IgG1。选择合适的抗体时,需要考虑其对肿瘤相关抗原的特异性、亲和力、免疫原性等因素 。高特异性的单克隆抗体能确保 ADCs 准确地找到肿瘤细胞,提高治疗效果,降低副作用。靶抗原:治疗的关键靶点:靶抗原的选择对 ADCs 的疗效至关重要 。理想的靶抗原应在癌细胞上高表达,而在正常细胞上低表达或不表达,同时还应具备良好的内化特性,便于 ADCs 进入细胞发挥作用 。例如 HER2 受体在癌细胞上的表达量远高于正常组织,是一个常用的靶抗原 。生物信息学和计算机辅助药物筛选技术的发展,有助于更精准地选择靶抗原。连接子:稳定与释放的平衡者:连接子是连接细胞毒性有效载荷和单克隆抗体的重要部分,它的稳定性和裂解特性会影响 ADCs 的疗效和安全性 。连接子分为非裂解型和裂解型,非裂解型连接子能确保药物在血液循环中稳定存在,直到被肿瘤细胞内化后才释放;裂解型连接子则能在特定环境下,如肿瘤微环境的酸性条件或特定酶的作用下,释放药物 。不同类型的裂解型连接子,如酸敏感型、酶敏感型和可还原型连接子,各自有着独特的作用机制(原文 Fig. 2,展示各种裂解型连接子的结构)。细胞毒性有效载荷:癌细胞的 “杀手”:细胞毒性有效载荷是 ADCs 发挥抗癌作用的核心部分 。常见的有效载荷包括微管靶向剂、DNA 损伤剂等,它们能在进入肿瘤细胞后,通过不同机制抑制癌细胞生长或诱导其死亡 。例如微管靶向剂通过破坏微管的正常功能,阻止癌细胞分裂;DNA 损伤剂则直接作用于癌细胞的 DNA,引发细胞死亡(原文 Fig. 3,展示不同类型细胞毒性有效载荷的分类)。三、ADCs 的作用机制与临床应用作用机制:精准打击癌细胞:ADCs 通过静脉注射进入血液循环,到达肿瘤部位后,其单克隆抗体部分会与肿瘤细胞表面的靶抗原结合,然后通过受体介导的内吞作用进入细胞 。在细胞内,根据连接子的类型,药物会以不同方式释放出来,发挥细胞毒性作用,诱导癌细胞凋亡或死亡 。此外,ADCs 还可能产生旁观者效应,对周围未结合 ADCs 的癌细胞也产生杀伤作用(原文 Fig. 6,展示 ADCs 的作用机制示意图)。临床应用:多种癌症的新希望:目前,已有多种 ADCs 获得 FDA 批准用于临床治疗不同类型的癌症 。例如,Mylotarg 用于治疗复发性 CD33 + 急性髓细胞白血病,Adcetris 用于治疗 CD30 阳性的霍奇金淋巴瘤和间变性大细胞淋巴瘤,T-DM1 用于治疗 HER2 阳性的转移性乳腺癌等(原文 Table 2,展示 FDA 批准的 ADCs 相关信息,包括商品名、抗原、有效载荷、连接子类型、疾病适应症、开发者和批准年份等)。这些药物在临床试验中展现出良好的疗效,为癌症患者带来了新的治疗选择 。此外,还有许多 ADCs 正在进行临床试验,有望为更多癌症患者提供有效的治疗方案 。四、ADCs 面临的挑战与应对策略面临的挑战:尽管 ADCs 在癌症治疗中取得了一定成果,但仍面临不少挑战 。脱靶毒性是其中一个重要问题,由于细胞毒性分子可能在血液中提前释放,会对正常组织产生不良影响,不同的药物 - 连接子组合可能导致不同的毒性反应 。肿瘤细胞对 ADCs 产生耐药性也是一大难题,其机制包括降低靶抗原表达、激活药物外排泵等 。此外,ADCs 在开发过程中还可能出现蛋白质聚集的问题,影响药物的质量和疗效 。应对策略:为了克服这些挑战,研究人员采取了多种策略 。针对耐药性问题,可以通过改变细胞毒性药物、调整连接子的亲水性来降低肿瘤细胞的耐药性,或者采用双靶点或双有效载荷的设计,增强对肿瘤细胞的杀伤作用 。在减少脱靶毒性方面,优化连接子的设计,使其更稳定,减少药物提前释放;同时,精准选择靶抗原,避免正常细胞摄取 ADCs 。对于蛋白质聚集问题,在生产和储存过程中优化条件,采用合适的表征方法监测聚集情况 。五、ADCs 的未来展望技术创新:未来,ADCs 的发展将聚焦于技术创新 。通过识别新的肿瘤相关抗原和开发下一代抗体,如双特异性和多特异性抗体,可以提高 ADCs 的靶向特异性,同时作用于多个靶点,克服肿瘤的异质性和耐药性 。在有效载荷和连接子技术方面,引入新型细胞毒性剂,如免疫调节剂、蛋白质降解剂和 RNA 药物等,能够更有效地杀死肿瘤细胞,减少脱靶效应;而改进连接子技术,可以更精确地控制药物释放,提高治疗效果 。联合治疗:联合治疗是 ADCs 未来发展的重要方向 。将 ADCs 与免疫疗法,如免疫检查点抑制剂、癌症疫苗或表观遗传调节剂联合使用,能够增强免疫系统对肿瘤细胞的攻击,提高治疗效果,降低耐药性的发生 。通过生物标志物指导个性化治疗,根据患者的基因、蛋白质等特征选择最适合的 ADCs,能够实现精准医疗,提高患者的治疗反应和生存率 。拓展应用领域:ADCs 的应用领域有望进一步拓展 。除了癌症治疗,ADCs 在自身免疫性疾病和感染性疾病的治疗中也展现出潜力 。通过将细胞毒性药物或免疫调节药物精准输送到病变细胞,ADCs 可以为这些疾病的治疗提供新的思路和方法 。随着生物制造技术的进步,ADCs 的生产将更加高效、低成本,监管框架也会不断完善,以适应新型 ADCs 的发展 。六、结论抗体 - 药物偶联物(ADCs)作为一种创新的癌症治疗方法,已经在临床实践中取得了显著的成效,为癌症患者带来了新的希望 。尽管目前还面临一些挑战,但随着技术的不断进步和研究的深入,ADCs 在未来有望通过技术创新、联合治疗和拓展应用领域等方式,进一步提高治疗效果,成为个性化和精准医学的重要组成部分,为更多患者带来更好的治疗体验和生存机会 。识别微信二维码,添加抗体圈小编,符合条件者即可加入抗体圈微信群!请注明:姓名+研究方向!本公众号所有转载文章系出于传递更多信息之目的,且明确注明来源和作者,不希望被转载的媒体或个人可与我们联系(cbplib@163.com),我们将立即进行删除处理。所有文章仅代表作者观点,不代表本站立场。
抗体药物偶联物
分析
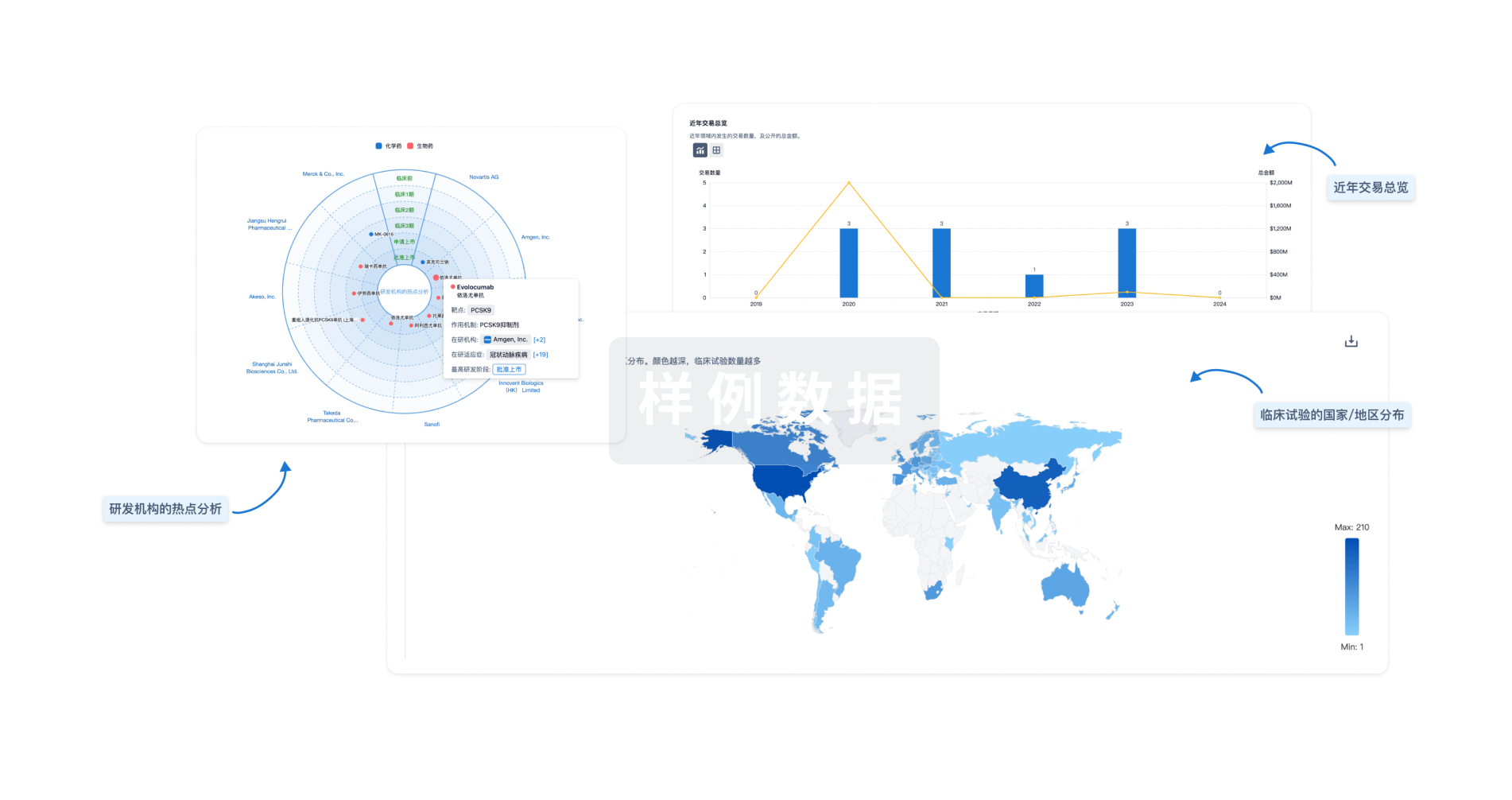
对领域进行一次全面的分析。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用






