预约演示
更新于:2025-01-23
TLR9 x TLR7
更新于:2025-01-23
关联
10
项与 TLR9 x TLR7 相关的药物作用机制 TLR7拮抗剂 [+1] |
在研机构 |
原研机构 |
在研适应症 |
非在研适应症- |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
作用机制 TLR7拮抗剂 [+2] |
在研机构 |
原研机构 |
非在研适应症- |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
8
项与 TLR9 x TLR7 相关的临床试验EUCTR2015-003277-15-GB
A Phase 2, Randomized, Double-Blind, Placebo-Controlled Trial of IMO-8400 in Patients with Dermatomyositis
开始日期2016-09-02 |
申办/合作机构 |
NCT02612857
A Phase 2, Randomized, Double-Blind, Placebo-Controlled Trial of IMO-8400 in Patients With Dermatomyositis
The purpose of this study is to determine how safe and effective IMO-8400 is in adults with dermatomyositis.
开始日期2015-11-01 |
申办/合作机构 |
NCT02363439
An Extension Study to Evaluate the Long-Term Safety, Tolerability, and Clinical Activity of IMO-8400 in Patients With Relapsed or Refractory Waldenström's Macroglobulinemia Who Completed Study 8400-401
An extension study for patients who complete 24 weeks of IMO-8400 on study 8400-401.
开始日期2014-11-01 |
申办/合作机构 |
100 项与 TLR9 x TLR7 相关的临床结果
登录后查看更多信息
100 项与 TLR9 x TLR7 相关的转化医学
登录后查看更多信息
0 项与 TLR9 x TLR7 相关的专利(医药)
登录后查看更多信息
1,150
项与 TLR9 x TLR7 相关的文献(医药)2024-12-21·Biological and Pharmaceutical Bulletin
Amodiaquine Analogs Are Potent Inhibitors of Interleukin-6 Production Induced by Activation of Toll-Like Receptors Recognizing Pathogen Nucleic Acids
Article
作者: Kitabatake, Kazuki ; Morita, Tomoe ; Tanaka, Tomohiro ; Takenaka, Yohei ; Tsukimoto, Mitsutoshi ; Kanbe, Azusa ; Otaki, Shotaro ; Aoki, Shin ; Nakamura, Koki ; Yokoi, Kenta ; Tojo, Toshifumi ; Sekiguchi, Saki ; Uchiumi, Fumiaki ; Satoh, Hidetoshi
2024-12-01·eBioMedicine
NF-κB c-Rel is a critical regulator of TLR7-induced inflammation in psoriasis
Article
作者: Vadlakonda, Ananya ; Griffith, Alexis D ; Centore, Joshua T ; Cooper, Kevin D. ; Rohr, Bethany ; Cress, Jordan D ; de Jesus, Tristan J. ; Ramakrishnan, Parameswaran ; Centore, Joshua T. ; de Jesus, Tristan J ; Liu, Angela Rose ; Sarkar, Nandini ; Cooper, Kevin D ; McCormick, Thomas S. ; Griffith, Alexis D. ; McCormick, Thomas S ; Cress, Jordan D.
2024-12-01·Current Opinion in Immunology
The path ahead for understanding Toll-like receptor-driven systemic autoimmunity
Review
作者: Barton, Gregory M ; Hamerman, Jessica A
51
项与 TLR9 x TLR7 相关的新闻(医药)2025-01-15
·小药说药
关注小药说药,一起成长!
前言
抗体偶联药物(ADC)是由靶向特异性抗原的单克隆抗体与小分子细胞毒性药物通过连接子链接而成,兼具传统小分子化疗的强大杀伤效应及抗体药物的肿瘤靶向性。ADCs于20世纪90年代中期首次进入临床试验以来,经过近30年的发展,已经成为一个非常成功的肿瘤学平台。
然而,要充分发挥ADC平台的潜力,就必须进行创新的分子设计,以及肿瘤特异性靶抗原和有效生物标志物的鉴定,以应对耐药性、肿瘤异质性和治疗相关不良反应等几个临床挑战。目前,多种新兴的ADC形式已经进入临床前和临床开发的早期阶段,包括双特异性ADC、条件激活ADC、免疫刺激ADC、蛋白质降解ADC和双载荷ADC,每种ADC都提供了应对这些各种挑战的独特能力,为癌症患者提供更多更好的治疗选择。
双特异性ADC
肿瘤异质性和耐药性通常限制针对单一靶点治疗的抗肿瘤活性。为了应对这一挑战,双特异性抗体是一种能够同时结合两种不同靶分子和/或细胞的方法。利用该技术的双特异性ADC作为增强抗肿瘤疗效的潜在途径成为最近的热点。迄今为止探索的设计可分为两种类型:针对同一抗原不同表位的双特异性ADC,以及靶向两种不同抗原的双特异性ADC。
MEDI4276是一种将曲妥珠单抗的scFv与另一种抗HER2 IgG1抗体39S的N末端融合的四价HER2靶向ADC。MEDI4276在治疗难治性HER2+癌症的小鼠异种移植物模型中显示出显著的活性,但在临床测试时并未表现出良好的疗效-安全性平衡。在乳腺癌的患者中,总体有效率(ORR)较低(9.4%),最大耐受剂量(MTD)确定为0.75 mg/kg每3周一次。
ZW49是另一种靶向HER2双靶点的ADC,其不对称结构能够实现二价HER2结合。在测试ZW49的I期剂量发现研究中,建议的II期剂量(RP2D)为2.5 mg/kg,每3周一次。在该给药方案下接受ZW49治疗的29名可评价疗效的患者中,多种HER2+晚期癌症类型的确认ORR为28%,疾病控制率为72%。只有9%的患者出现≥3级治疗相关不良事件(TRAE),另有三名患者出现临床严重事件。这些结果表明,ZW49在经过大量预处理的患者中具有可控的安全性和良好的抗肿瘤活性。MEDIO276和ZW49都被设计用于识别HER2并促进受体聚集、内化和溶酶体运输。
AZD9592是阿斯利康开发的EGFR/c-Met
ADC,通过可连接linker偶联新型拓扑异构酶1载荷,主要解决奥希替尼耐药。相较于EGFR,AZD9592 对 c-MET 具有更高的亲和力,目的是减少由EGFR驱动的正常组织毒性。在PDX及耐药模型中,单药或联用奥希替尼均展现出良好的抗肿瘤活性。
M1231是Sutro和默克子公司EMD
Serono合作开发的MUC1/EGFR双抗ADC,采用非天然氨基酸定点偶联技术,通过可裂解VC连接子偶联hemiasterlin衍生物(微管抑制剂),DAR为4。临床前研究显示,在ESCC和NSCLC患者衍生的异种移植模型中具有很强的抗肿瘤活性。
BL-B01D1 是中国首个进入I期临床的双抗ADC,靶向EGFR和HER3,接头采用其自有的Ac接头,相较于Mc接头具有更好的稳定性,亲水性更好,不易聚集;毒素为自有的喜树碱类似物ED04。其I期临床用药安全性较好,未出现药物相关的患者死亡情况。在安全性较好的10例可评估的NSCLC末线患者中,ORR为60%,DCR为90%。
条件激活ADC
靶向受体的传统ADC不仅在肿瘤细胞上表达,而且在某些非恶性组织上表达,通常与不可避免的靶向非肿瘤毒性有关,导致剂量减少或治疗中断。为了解决这个问题,已经开发了以条件活性抗体为特征的新型ADC设计。
这一设计概念的灵感来自小分子的前药设计,通过该设计赋予药物的药理学非活性形式,然后在循环或某些器官中代谢为其活性形式,从而提高体内稳定性和/或特异性。通过一种对多种蛋白酶敏感的可切割肽序列(LSGRSDNH),人们开发了几种条件激活的ADC,目前正在临床前和临床开发中,包括praluzatamab ravtansine(NCT03149549和NCT04596150)和CX-2029(NCT03543813)。
此外,人们还开发了具有pH响应性抗原结合位点的ADC。TME的酸性通常略高于大多数非恶性组织,由于抗原结合位点的可逆构象变化,这种pH差异可用于实现ADC的条件激活。迄今为止,已经开发了包括靶向EGFR、HER2、AXL和ROR2的各种pH依赖性ADC。在多种临床前小鼠异种移植物模型中显示出良好的抗肿瘤活性。
免疫激动ISAC
过去十年癌症免疫疗法的变革性进展,激发了人们对该领域的新兴趣。在由肿瘤细胞释放损伤相关分子模式(DAMP)触发的先天免疫激活的背景下,与模式-认知受体(PRRs)相互作用的免疫佐剂分子已成为癌症药物开发的焦点。在将PRR激动剂特异性递送至肿瘤的各种尝试中,与特异性PRR激动药的偶联抗体已成为一种有前途的先天免疫局部激活方法。
临床前研究已经证明,与携带细胞毒性有效载荷的传统ADC相比,ISAC具有独特的潜在优势:首先,ISAC介导的抗肿瘤反应可以靶向多种肿瘤相关的DAMP;其次,ISAC介导的免疫刺激最终不仅激活抗原呈递细胞(APC),而且可能激活其他肿瘤浸润免疫细胞,如T细胞;第三,ISACs在整个细胞免疫反应中引发免疫记忆效应,提供持久的抗肿瘤作用并降低复发风险。
在迄今为止表征的所有TLR中,TLR7、TLR8和TLR9是迄今为止开发的大多数ISAC的主要靶标。抗HER2 TLR8 ISAC SBT6050即pertuzumab
zuvotolimod,其由通过可裂解的连接子与pertuzumab偶联的TLR8激动剂组成。在I期研究(NCT04460456)中,其作为单一疗法与抗PD-1抗体pembrolizumab或cemiplimab组合进行测试;在I/II期研究(NCT05091528)中与其他HER2靶向疗法组合进行测试。但由于细胞因子相关的不良事件以及缺乏足够的单药活性,最终导致这些研究终止。免疫激动型ISAC的药效和安全性仍有待临床的进一步检验。
基于PROTAC的DAC
DAC携带蛋白水解靶向嵌合体(PROTAC)而不是传统的细胞毒性有效载荷,是一类新的靶向疗法。DAC形式有可能进一步提高这种新模式的临床实用性,通过利用基于抗体的药物递送的能力提供高水平的肿瘤特异性和持久活性。
DAC的单克隆抗体部分识别肿瘤相关抗原,触发DAC-抗原复合物的内化。连接子在蛋白水解、酸性和/或还原条件下降解,从而将结合的PROTAC分子释放到细胞质中。通过E3连接酶的参与导致感兴趣的蛋白质(POI)泛素化,从而导致POI的降解。一些DAC正处于早期临床开发阶段,如靶向含溴结构域蛋白4(BRD4)的DAC正在为CLL1+急性髓细胞白血病(AML)患者中进行测试;以及靶向G1至S相变1蛋白(GSPT1)的ORM-5029,该DAC目前正在HER2+乳腺癌症患者中测试(NCT05511844)。
双载荷ADC
大多数实体瘤由异质性癌细胞亚群组成,具有不同的基因表达谱和对药物的敏感性水平,具有不同作用机制。因此,临床实践中通常采用涉及具有不同作用模式的多种药物的联合方案。双有效载荷ADC有可能作为单一药物引发相加或协同效应,并在维持简单给药范围的同时克服治疗难治性肿瘤患者的耐药性。
2017年报道了一种使用含有两个正交掩蔽的半胱氨酸残基的支链化学连接子生产双载荷ADC的方法。通过顺序偶联有效载荷能够以16的DAR将MMAE和MMAF均匀偶联到抗CD30抗体上。这种双载荷ADC在表达CD30+MDR-的间变性大细胞淋巴瘤(ALCL)的小鼠异种移植物模型中显示出强大的活性。
除了具有MMAE和MMAF有效载荷的ADC之外,随着结合两种不同有效载荷类别的ADC的开发,双载荷ADC的临床潜力得到了进一步探索。如asterlin加上与抗FolRα抗体偶联的TLR激动剂的组合,其在小鼠模型中显示出协同抗肿瘤活性和免疫记忆。目前,双载荷ADC潜力的研究仍处于探索的早期阶段。
小结
ADC的治疗潜力巨大,但要实现这一潜力,需要克服几个关键挑战,如耐药性、肿瘤内和瘤瘤间异质性以及TRAE的风险。新兴的ADC模式,包括双特异性和双载荷ADC,显示出解决耐药性和肿瘤异质性的潜力,而条件激活ADC可能增加肿瘤特异性并降低不良事件的发生率。将ADC平台与其他干预策略相结合,如免疫调节和降解传统上不可治愈的靶点,为实施多模式癌症治疗以及化疗、放疗、免疫疗法和其他靶向治疗提供了机会。
ADC的发展正处于变革性增长的边缘,有望大幅改变癌症治疗格局。随着我们更好地了解肿瘤生物学并改进ADC设计,我们将更接近真正有效、安全和个性化的癌症治疗目标,这将最终为顽固性癌症患者带来新的希望。
参考文献:
1.Exploring
the next generation of antibody-drug conjugates. Nat Rev Clin Oncol.2024
Jan 8.2. Antibody–Drug Conjugates: The Last Decade. Pharmaceuticals 2020, 13,
245
公众号内回复“ADC”或扫描下方图片中的二维码免费下载《抗体偶联药物:从基础到临床》的PDF格式电子书!
公众号已建立“小药说药专业交流群”微信行业交流群以及读者交流群,扫描下方小编二维码加入,入行业群请主动告知姓名、工作单位和职务。
抗体药物偶联物临床1期临床结果
2024-12-20
欢迎关注凯莱英药闻
(收集周期:12.16-12.20,收集部分为首次申请临床、首次申请上市、首次批准上市的创新药)
国内创新药IND汇总
1、安达生物:注射用PCNAT-01
作用机制:——
适应症:肿瘤
12月18日,安达生物的注射用PCNAT-01的临床试验申请(IND)获CDE受理。PCNAT-01一款根据患者自身肿瘤的新抗原,定制生产的个性化治疗性多肽疫苗;该产品经皮下注射后,能够诱导患者自身免疫细胞对肿瘤杀伤以清除肿瘤,解决了自身肿瘤免疫原暴露、递呈不足的问题,避免传统肿瘤药物非特异杀伤带来的副作用。此外,该疫苗还借助免疫记忆机制形成长效抑瘤作用,恢复机体免疫稳态,降低复发。PCNAT-01采用个性化疫苗多靶点设计,能够解决单一靶点药物覆盖不足的问题,最大程度避免因靶点丢失引起的肿瘤复发与耐药。
由研究者发起的临床研究显示,采用PCNAT-01治疗的胰腺癌患者最长已达术后6年多无复发,受试者的OS和RFS明显高于同期常规辅助治疗(即历史对照)的患者;5年无复发生存率为43.8%(历史对照28%),5年总生存率66%(历史对照35.1%)。
2、博锐生物:注射用BR111
作用机制:靶向ROR1 ADC
适应症:血液瘤和实体瘤
12月19日,博锐生物的注射用BR111的IND获CDE受理,用于治疗ROR1阳性血液瘤和实体瘤。BR111一款靶向ROR1双表位的抗体偶联药物(ADC),采用新一代CysXTM不可逆定点偶联技术平台,将靶向ROR1双表位的抗体与小分子毒素艾日布林偶联。该药物可识别肿瘤细胞表面ROR1的两个非重叠表位,双表位识别带来更强的亲和力和内吞活性;被内吞进入ROR1阳性肿瘤细胞后,在溶酶体内释放小分子毒素,有效杀伤肿瘤细胞。BR111具有较高的均一性和循环稳定性,显著减少毒素的脱落,具有更好的安全性及更优的治疗窗。
临床前研究显示,在多种动物模型中,BR111展示了比临床同类分子更优异的体内抗肿瘤药效,且具有更好的安全性。此外,BR111还能诱导旁观者效应,并在肿瘤微环境中发挥免疫激活作用,具有与包括靶向药物和免疫疗法在内的多种药物联用的潜力。
3、第一三共:DS-3939a注射液
作用机制:靶向TA-MUC1 ADC
适应症:肿瘤
12月19日,第一三共的DS-3939a注射液的IND获CDE受理。DS-3939a是一款靶向MUC1的抗体偶联药物(ADC),MUC1(CD227)是粘蛋白家族(Mucins)的一员,在健康组织中,MUC1在分泌性上皮细胞的顶端表面表达;此外,当上皮细胞受到外来信号的刺激时,细胞极性可逆性丢失,MUC1在整个细胞表面表达且与位于细胞基地外侧的各种因子作用并参与其下游信号通路、促进细胞的修复与存活。肿瘤细胞中的MUC1高度唾液酸化,引发糖链延伸的提前终止从而形成截短的糖链分支,导致肿瘤相关MUC1(TA-MUC1)糖基化水平降低、肿瘤细胞粘附力下降,从而为肿瘤转移提供了条件。MUC1的过表达通常与结肠癌,乳腺癌,卵巢癌,肺癌和胰腺癌有关。
4、信达生物:IBI3007
作用机制:靶向TROP2 ADC;TLR7/TLR8激动剂
适应症:肿瘤
12月19日,信达生物的IBI3007的IND获CDE受理。IBI3007是一款免疫激动剂抗体偶联药物(ISAC),由Trop2抗体偶联TLR7/8激动剂而成。临床前数据显示,IBI-3007在体内药效显著,在小鼠移植瘤模型中,单次给药5mg/kg就能达到抑制肿瘤目的;且将IBI-3007低剂量1mg/kg与ADC联用,也具有很好的疗效。此外,IBI-3007的安全性良好,比silverback TLR7 ISAC安全性高很多,第二次给药后如果有过敏反应会引起体温下降。在猴子毒理实验中,HNSTD为15
mg/kg,4 doses,IV注射且TK数据良好。
5、荣瑞医药:OVV-01注射液
作用机制:靶向NY-ESO-1溶瘤病毒
适应症:实体瘤
12月19日,荣瑞医药的OVV-01注射液的IND获CDE受理。OVV-01是一种经过基因工程改造的VSV溶瘤病毒,旨在选择性扩增肿瘤细胞并表达肿瘤相关抗原NY-ESO-1。在一项I期、首次人体试验、开放标签、多中心研究中,显示在11例可进行疗效评估的晚期实体瘤患者中,16 周时的ORR(ORR16w)为 27.3%,3×107PFU、3×108PFU、3×109PFU和6×1010PFU剂量组的ORR分别为0%、33.3%、25.0%和100%。16周时的DCR(DCR16w)为63.6%。剂量高于3.0×109 PFU 时,ORR16w 为 40%(2/5),DCR16w 为 80%(4/5)。值得注意的是,在四例可评估的晚期STS患者中,ORR16w 为 75%,DCR16w 为 75%。两例STS患者在剂量超过3.0×109PFU时达到CR。总体PFS24w率为43.1%。
在安全性上,94.4%(17/18)的患者出现了治疗相关不良事件(TRAEs)(≥G3 33.3%)。最常见的不良反应(≥20%)包括发热(66.7%)、淋巴细胞减少(27.8%)、贫血(22.2%)和呕吐(22.2%)。未观察到与治疗相关的严重不良事件。通过qPCR,未在患者唾液、尿液或粪便中检测到病毒脱落。
6、和誉生物:ABSK131胶囊
作用机制:PRMT5抑制剂
适应症:肿瘤
12月20日,和誉生物的ABSK131胶囊的IND获CDE受理。ABSK131是一款新型小分子PRMT5-MTA协同抑制剂,具有强效的抗肿瘤活性。MTAP参与编码多胺和嘌呤代谢中的关键限速酶,在嘌呤和甲硫氨酸合成补救途径中起重要作用,PRMT5在MTAP缺失肿瘤中表现出“合成致死”效应。在临床前研究中,ABSK131对MTAP缺失的肿瘤细胞显示出极好的选择性,口服给药具有良好的药物代谢和药代动力学特性。
7、海昶生物:HC016脂质复合物注射液
作用机制:TLR9激动剂
适应症:肿瘤
12月20日,海昶生物的HC016脂质复合物注射液的IND获CDE受理。HC016以公司自主研发的ToII样受体9(TLR9)激动剂为活性成分,搭配拥有自主知识产权的QTsomeTM递送系统,拟用于黑色素瘤、头颈癌等多种实体瘤的临床治疗。该药物是全球首款采用脂质纳米颗粒递送的瘤内注射产品,临床前数据显示出优秀的实体瘤治疗潜力及更高的安全性。
国内创新药NDA汇总
1、云顶新耀:伊曲莫德片
作用机制:S1P受体调节剂
适应症:溃疡性结肠炎
12月17日,云顶新耀的伊曲莫德片的新药上市申请(NDA)获CDE受理,拟用于治疗中重度活动性溃疡性结肠炎(UC)。伊曲莫德是新一代S1P受体调节剂,通过每日一次口服的治疗方案,可快速起效,并达到无激素缓解、黏膜愈合。伊曲莫德最初由Arena Pharmaceuticals公司开发,辉瑞于2022年以总价67亿美元完成了对Arena的收购,而云顶新耀早在2017年已从Arena获得了在大中华区和韩国开发、生产和商业化伊曲莫德的独家权利。该药物已于去年10月和今年2月先后在美国和欧盟获得新药上市批准。
一项在亚洲开展的最大规模中重度活动性UC患者III期临床研究公布的数据显示,12 周的诱导治疗后,伊曲莫德组达到主要研究终点,临床缓解率为 25%(vs 安慰剂组 5.4%)。在关键次要终点部分,内径改善和临床应答率分别为 37.3% 和 58.3%(vs 安慰剂组 9.8% 和 27.7%)。在安全性方面,伊曲莫德的安全性和耐受性良好,与之前的研究一致。
国内新药上市获批
1、礼来:多奈单抗注射液
作用机制:靶向Aβ单抗
适应症:阿尔茨海默病
12月18日,礼来的多奈单抗注射液(记能达®)获NMPA批准上市,用于治疗成人因阿尔茨海默病引起的轻度认知功能障碍和阿尔茨海默病轻度痴呆。多奈单抗(donanemab)一款靶向pGlu3-Aβ的抗体,每四周一次静脉输注,是目前唯一一个有证据支持在清除淀粉样蛋白斑块后可停药的靶向淀粉样蛋白的疗法。
TRAILBLAZER-ALZ 2三期研究显示,使用donanemab治疗可以显著减缓两组人群的临床衰退。在疾病进展较早的人群中,接受donanemab治疗的患者在综合阿尔茨海默病评分量表(iADRS),包含记忆、思维和日常功能的衰退速度与安慰剂相比显著减缓了35%。在两组受试者中,接受donanemab治疗的受试者,相比接受安慰剂的受试者展现了高达39%的降低疾病进展至下一临床阶段的风险。在整体受试者人群中,与研究开始时相比,使用donanemab
6个月时平均减少了61%的淀粉样蛋白斑块,12个月时为80%,18个月时为84%。
2、信达生物/葆元医药:己二酸他雷替尼胶囊
作用机制:ROS1/NTRK抑制剂
适应症:非小细胞肺癌
12月20日,国家药监局网站显示,信达生物和葆元医药合作开发的新一代ROS1/NTRK抑制剂他雷替尼获批上市,用于治疗既往接受过ROS1-TKI治疗的ROS1融合阳性非小细胞肺癌(NSCLC)患者。
他雷替尼(研发代号:DS-6051)原本由第一三共开发,其开发、生产和商业化权益于2018年12月被授予葆元医药。2021年6月,信达生物与葆元生物达成协议,以在大中华区合作开发和商业化他雷替尼。葆元医药负责泰莱替尼在中国大陆地区的临床开发和注册报批以及大中华地区的临床生产和商业化生产,信达生物则有权负责他雷替尼在中国香港、澳门和台湾地区的开发和注册报批。
他雷替尼治疗经治ROS1阳性NSCLC患者的II期TRUST-I研究结果显示,接受过克唑替尼治疗的患者在接受他雷替尼治疗后,中位缓解持续时间(DOR)为10.6个月,中位无进展生存期(PFS)为7.6个月。
他雷替尼治疗经治ROS1阳性NSCLC患者的II期TRUST-II研究结果显示,接受过克唑替尼或恩曲替尼治疗的患者在接受他雷替尼治疗后,经确认的客观缓解率(cORR,标准为肿瘤体积缩小)为55%,DOR达到12个月的患者比例为75%,中位PFS为11.9个月。
感谢关注、转发,转载授权、加行业交流群,请加管理员微信号“hxsjjf1618”。
“在看”点一下
临床申请抗体药物偶联物免疫疗法疫苗
2024-12-11
·医药速览
本文作者对SLE发病机制结合近些年的研究进展做了一些更新,本公众号不做详细解读,仅用来抛砖引玉,建议读者朋友感兴趣的可以阅读原文,对临床助益不多,但对拓展科研思维或可帮助一二。
图1.SLE发病易感因素概览
图2.狼疮发病病理机制近期进展
SLE中核抗原的主要来源之一是凋亡细胞(A),
导致胞内物质的释放,包括核抗原(eg. DNA,RNA)和核蛋白(B)。
细胞释放到细胞外空间的微粒和细胞外囊泡可以刺激免疫系统并促进SLE的发病机制(C)。
凋亡衍生的膜囊泡也驱动cGAS-STING途径并增加SLE中I型IFN的产生(D)。
异常NETosis在SLE的发病机制中有所贡献(E)。
反转录转座子的异常表达有助于免疫复合物的形成(F)。
血小板通过刺激血小板FcγRIIA释放线粒体DNA、分泌血小板相关凝集素、半乳糖苷结合、可溶性3结合蛋白和与Tregs (G)的相互作用(mtDNA)参与SLE的发病机制(G)。
来自红细胞线粒体的mtDNA触发涉及cGAS和STING的先天DNA感应,导致干扰素的诱导(H)。
TLR7和TLR9分别介导对RNA和DNA的耐受性丧失(I)。
I型IFN,特别是IFN - α的产生失调,有助于自身免疫的启动和进展(J)。
DCs识别和内化自身抗原,如核酸、免疫复合物、凋亡细胞碎片(K)。
自体抗原呈递给自身反应性T细胞,导致其活化和增殖(L)。
人浆细胞样树突状细胞(pDCs)在调节免疫应答中起着至关重要的作用,并且在SLE的发病机制中起着至关重要的作用(M)。
巨噬细胞和中性粒细胞通过各种机制参与SLE的发病机制(N)。
补体成分或调节蛋白的缺陷导致清除受损,从而导致核抗原的持久性和免疫激活的增强(O)。
B细胞功能异常,包括B细胞受体信号的改变、B细胞耐受机制的缺陷、B细胞凋亡受损和B细胞亚群的失调,都是SLE的发病机制(P)。
生发中心的异常形成和失调会导致自身反应性B细胞和致病抗体的产生(Q)。
CD4+ T细胞与自身反应性B细胞相互作用,激活并分化为产生抗体的细胞(R)。
Tregs的功能和数量改变有助于免疫耐受的破坏,表现为抑制能力降低、生存受损和对细胞因子的反应性改变(S)。
SLE的一个标志是免疫复合物(T)的形成和沉积,导致SLE的组织特异性表现和并发症。
图3.角化细胞和I型干扰素在皮肤红斑狼疮发病机制中的作用的最新进展
紫外线照射和核酸增强角质形成细胞产生I型干扰素,包括IFN-κ、IFN-α、IFN-β和IFN-λ。
I型IFNs诱导干扰素刺激基因(ISGs)的转录,从而导致促炎细胞因子和趋化因子的产生(A)。
促炎性因子和趋化因子诱导树突状细胞(cDCs和pDCs)、巨噬细胞、CD4+ T细胞和细胞毒性细胞(NK细胞和CD8+ T细胞)的募集和激活(B)。
狼疮皮损处的炎症浸润也产生I型IFN。最近的研究表明,浆细胞样树突状细胞(pDCs)并不是IFN-α和其他I型IFN的主要产生细胞,它们主要由常规树突状细胞(cDCs)和CD14+/CD16+巨噬细胞(C)产生。
紫外线和I型IFN通过增强几种诱导细胞死亡的膜蛋白(FAS、CD40、TRAIL)的表达和减少核酸清除来诱导角化细胞凋亡。pDCs表达颗粒酶B,其与细胞毒性细胞(NK细胞和细胞毒性CD8+ T细胞)在上皮损伤部位共定位于CLE病变,表明其在角化细胞凋亡中起作用(D)。
角化细胞产生高水平的B淋巴细胞刺激因子(BLyS),其在B细胞活化和自身抗体产生中起作用(E)。
Ref.
Arnaud L, et al. Autoimmun Rev. 2024 Oct;23(10):103648
文章来源:猫司令的学习笔记微信公众号
推文用于传递知识,如因版权等有疑问,请于本文刊发30日内联系医药速览。
原创内容未经授权,禁止转载至其他平台。
有问题可发邮件至yong_wang@pku.edu.cn获取更多信息。
©2021 医药速览 保留所有权利
往期链接
“小小疫苗”养成记 | 医药公司管线盘点
人人学懂免疫学 | 人人学懂免疫学(语音版)
综述文章解读 | 文献略读 | 医学科普 | 医药前沿笔记
PROTAC技术 | 抗体药物 | 抗体药物偶联-ADC
核酸疫苗 | CAR技术 | 化学生物学
温馨提示
医药速览公众号目前已经有近12个交流群(好学,有趣且奔波于医药圈人才聚集于此)。进群加作者微信(yiyaoxueshu666)或者扫描公众号二维码添加作者,备注“姓名/昵称-企业/高校-具体研究领域/专业”,此群仅为科研交流群,非诚勿扰。
简单操作即可星标⭐️医药速览,第一时间收到我们的推送
①点击标题下方“医药速览”
②至右上角“...” ③点击“设为星标
细胞疗法
分析
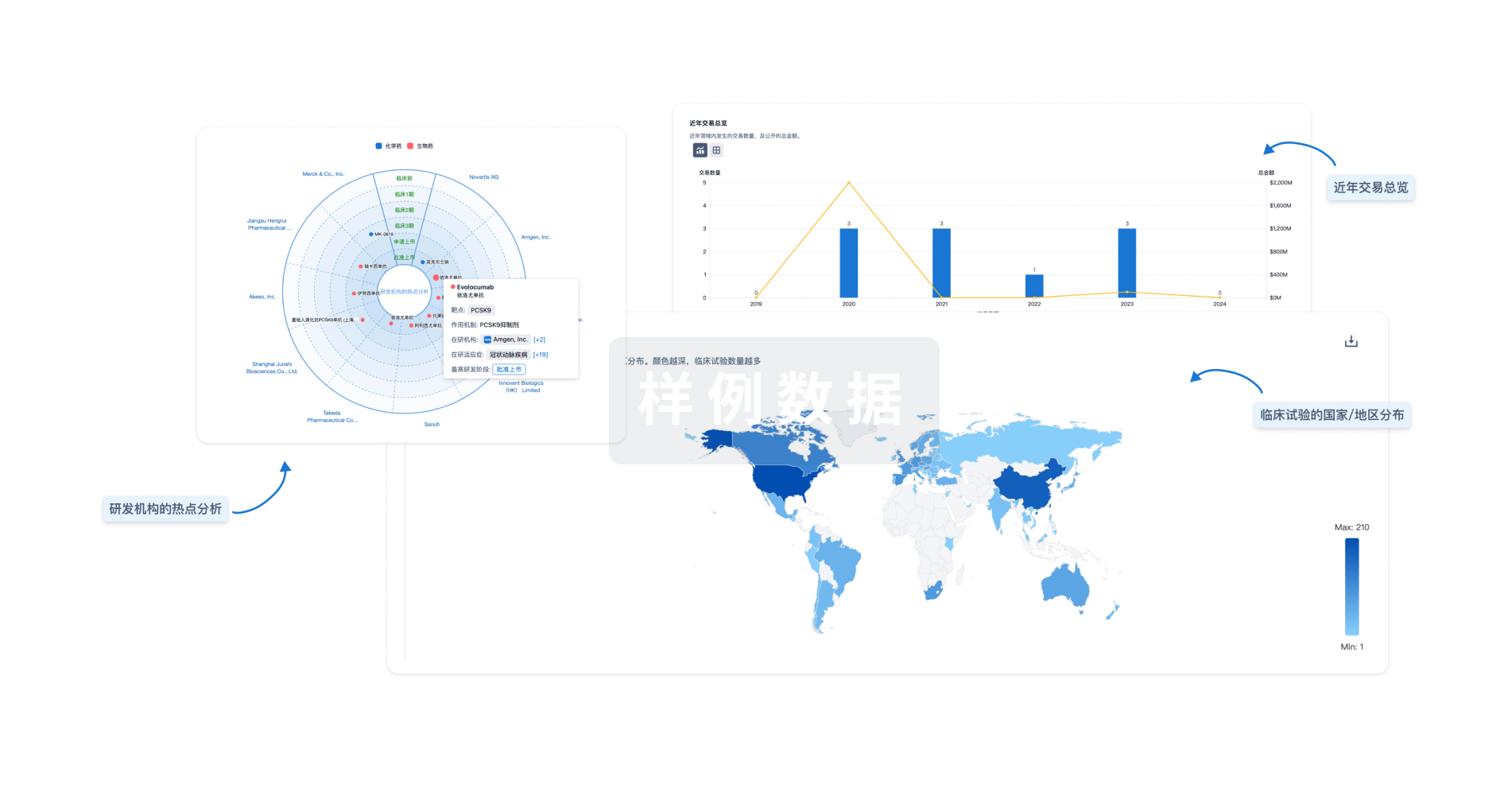
对领域进行一次全面的分析。
登录
或
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用