预约演示
更新于:2025-05-07
Paraplegia
截瘫
更新于:2025-05-07
基本信息
别名 EXTREMITY PARALYSIS, LOWER、Erb syndrome、Erb-Charcot syndrome + [61] |
简介 Severe or complete loss of motor function in the lower extremities and lower portions of the trunk. This condition is most often associated with SPINAL CORD DISEASES, although BRAIN DISEASES; PERIPHERAL NERVOUS SYSTEM DISEASES; NEUROMUSCULAR DISEASES; and MUSCULAR DISEASES may also cause bilateral leg weakness. |
关联
6
项与 截瘫 相关的药物靶点 |
作用机制 SNAP25 抑制剂 |
最高研发阶段批准上市 |
首次获批国家/地区 奥地利 [+29] |
首次获批日期2007-10-24 |
靶点- |
作用机制- |
原研机构 |
非在研适应症- |
最高研发阶段临床3期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
靶点 |
作用机制 AP4M1刺激剂 |
原研机构 |
在研适应症 |
非在研适应症- |
最高研发阶段临床3期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
138
项与 截瘫 相关的临床试验NCT06708962
Shoulder Joint Range of Motion and Subacromial Space Changes After Mulligan's Mobilization for Paraplegic Patients with Subacromial Impingement: a Randomized Controlled Trial
The aim of the study is to determine the effect of mulligan's mobilization on shoulder range of motion and subacromial joint space in paraplegic patients with subacromial impingement so, thirty patients will be randomly assigned to two groups group (A) and Group (B)
开始日期2024-12-01 |
申办/合作机构 |
NCT06740006
Interest of Prefabricated Carbon Orthoses in the Therapeutic Arsenal of Children and Adolescents Affected by Central or Peripheral Neurological Pathologies: Retrospective Study on Gait Parameters and Prescription Criteria
The goal of this observational study is to assess the interest of prefabricated carbon fiber ankle foot orthoses (AFO) in children and teenagers with central or peripheral neurological pathologies. The main question it aims to answer is:
Does prefabricated carbon AFO improve gait and provide user satisfaction? In current practice, all patients with gait impairments were fitted with this orthosis and had a 3D gait analysis with and without AFO. Biomecanical and follow-up data collected in current practice are analysed in this study in order to answer the question.
Does prefabricated carbon AFO improve gait and provide user satisfaction? In current practice, all patients with gait impairments were fitted with this orthosis and had a 3D gait analysis with and without AFO. Biomecanical and follow-up data collected in current practice are analysed in this study in order to answer the question.
开始日期2024-09-03 |
申办/合作机构- |
NCT06680063
Correlation Between Clinical Assessment and Neurophysiological Assessment in Spinal Cord Injury
This study is a cross-sectional study that aims to explore the correlation between clinical assessment and neurophysiological assessment in SCI. The clinical outcome assessment are modified-Modified Ashworth scale (m-MAS) and lower extremity motor score. The neurophysiological outcome is H-reflex outcome.
开始日期2024-08-23 |
申办/合作机构 |
100 项与 截瘫 相关的临床结果
登录后查看更多信息
100 项与 截瘫 相关的转化医学
登录后查看更多信息
0 项与 截瘫 相关的专利(医药)
登录后查看更多信息
16,572
项与 截瘫 相关的文献(医药)2025-12-31·NMC Case Report Journal
Pitfalls in the Diagnosis and Treatment of Low Back Pain in an Elite Para-athlete Successfully Treated by Transforaminal Full-endoscopic Discectomy with Thermal Annuloplasty: A Case Reports
Article
作者: MIZUTANI, Kozaburo ; KANDA, Yutaro ; SUGIURA, Kosuke ; SOEDA, Saori ; TEZUKA, Fumitake ; YAMASHITA, Kazuta ; KUMON, Masashi ; SAIRYO, Koichi ; MORIMOTO, Masatoshi
2025-12-01·Current Pain and Headache Reports
The Potential of Spinal Cord Stimulation in Treating Spinal Cord Injury
Review
作者: Abd-Elsayed, Alaa ; Knezevic, Nebojsa Nick ; Rashid, Mohammed Yousif ; Nasr, Ned F ; Forouzan, Eli Justin
2025-12-01·Current Neurology and Neuroscience Reports
Insights on the Shared Genetic Landscape of Neurodevelopmental and Movement Disorders
Review
作者: Boesch, Sylvia ; Indelicato, Elisabetta ; Zech, Michael ; Eberl, Anna
73
项与 截瘫 相关的新闻(医药)2025-05-05
30-Day Data from Endospan's NEXUS TRIOMPHE IDE Trial Demonstrate 63% Reduction in Major Adverse Event (MAE) Rate compared with Reference Performance Goal
ATLANTA, May 5, 2025 /PRNewswire/ --
Artivion, Inc. (NYSE: AORT), a leading cardiac and vascular surgery company focused on aortic disease, today announced the presentation of new clinical data from Endospan's NEXUS TRIOMPHE trial at the 105th American Association for Thoracic Surgery (AATS) Annual Meeting in Seattle, Washington. The data presented analyzed clinical outcomes across a 54-patient chronic aortic dissection statistical cohort at 30 days following treatment with NEXUS. Patients enrolled were at high risk for open surgical repair.
Dr. Bradley Leshnower, Professor of Surgery and Director of Thoracic Aortic Surgery, Emory University, presented the data from the NEXUS US IDE trial in an abstract titled, Endovascular Aortic Arch Repair Using a Novel Single-Branch Arch Stent Graft for Chronic Dissection: 30-Day Outcomes of the TRIOMPHE FDA Pivotal Study.
The data demonstrate statistically significant improvement in clinical outcomes and device technical performance compared with the performance goals set forth in the FDA-approved investigational device exemption (IDE).
*Major adverse events (MAEs) = mortality, disabling stroke, permanent paralysis/paraplegia, renal failure, aortic rupture, or development of new dissections in the thoracic aorta or brachiocephalic artery requiring intervention.
Dr. Leshnower said, "The 30-day results from the TRIOMPHE study demonstrate that aneurysms due to chronic aortic arch dissection involving the aortic arch can be successfully treated with this novel off-the-shelf stent graft system. The NEXUS device provides an excellent, minimally invasive alternative to open aortic arch replacement in select patients at high risk for open surgery. This novel technology will extend treatment to patients who previously would not be offered therapy due to prohibitive risk"
"We are pleased with the 30-day results and look forward to seeing the one-year outcomes," said Pat Mackin, Chairman, President and Chief Executive Officer of Artivion. "These results further validate this important therapeutic option for patients with aortic arch disease."
About the NEXUS TRIOMPHE Clinical Trial
The NEXUS TRIOMPHE trial is the US IDE study evaluating the NEXUS device in the endovascular treatment of chronic aortic dissection, either primary type B or residual dissection after prior type A repair, or aneurysm (not reported here). Inclusion criteria include the patient being high risk for open surgical repair. The clinical module of the PMA is anticipated to be filed after completion of one year of follow up with the 54 patients in the chronic aortic dissection statistical primary cohort.
About Artivion, Inc.
Headquartered in suburban Atlanta, Georgia, Artivion, Inc., is a medical device company focused on developing simple, elegant solutions that address cardiac and vascular surgeons' most difficult challenges in treating patients with aortic diseases. Artivion's four major groups of products include: aortic stent grafts, surgical sealants, On-X mechanical heart valves, and implantable cardiac and vascular human tissues. Artivion markets and sells products in more than 100 countries worldwide. For additional information about Artivion, visit our website, .
References
Leshnower B: Endovascular Aortic Arch Repair Using a Novel Single-Branch Arch Stent Graft for Chronic Dissection: 30-Day Outcomes of the TRIOMPHE FDA Pivotal Study. Presented May 3, 2025 at American Association of Thoracic Surgeons 105th annual meeting.
Contacts:
SOURCE Artivion, Inc.
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
临床研究临床结果
2025-04-28
CHESHIRE, UK I April 28, 2025 I
BlackfinBio Ltd, a clinical stage gene therapy company focused on the development of treatments for rare neurological diseases, announces today that the U.S. Food and Drug Administration (FDA) has cleared its Investigational New Drug (IND) application for a Phase 1/2 clinical trial of its novel adeno-associated virus (AAV) gene therapy, BFB-101, in children with Hereditary Spastic Paraplegia, Type 47 (SPG47).
The trial will be conducted in the U.S. at Boston Children’s Hospital and is expected to commence recruiting by the end of 2025. The FDA has granted an orphan drug designation (ODD) and rare pediatric disease designation (RPDD) to BFB-101 for the treatment of SPG47.
SPG47 is a rare, autosomal-recessive, neurological disorder characterised by progressive lower-limb spasticity, developmental delays and intellectual disability in children. It is caused by deleterious changes in the AP4B1 gene. Currently, there is no cure or effective treatment available for this devastating condition. BFB-101 has been designed specifically to address the underlying genetic cause of SPG47 by delivering a functional copy of the AP4B1 gene, with the goal of halting or reversing disease progression. Preclinical results with BFB-101 have demonstrated promising activity and safety (1).
The Phase 1/2 clinical trial is a single-center open-label study with a primary objective to assess the safety and efficacy of a single injection into the cerebrospinal fluid at the base of the brainstem near the spinal cord, termed an ‘ICM’ administration. This type of delivery generally allows for rapid and efficient delivery of agents to the central nervous system. BFB-101 will be administered by ICM in up to five children with AP4B1-associated SPG47. The secondary objectives will aim to evaluate the impact of BFB-101 on motor function, development and health-related quality of life.
“The FDA’s clearance of the BFB-101 IND is an important milestone for our rare neurological disease program and the company. We look forward to initiating enrollment in the U.S. later this year and are working closely with the investigator team at Boston Children’s Hospital to evaluate the therapeutic utility of this gene therapy in children with SPG47,” said Peter Nolan, Founder and Chief Executive Officer of BlackfinBio Ltd.
“We are proud to be leading this important clinical trial at Boston Children’s Hospital, bringing forward a precision medicine approach for children affected by SPG47. As clinicians, we see firsthand the impact of this disorder and the urgency for effective treatments. BFB-101 represents a promising gene therapy candidate, and this trial is a critical step toward delivering meaningful change for patients and families affected by AP-4-associated hereditary spastic paraplegia and related conditions,” said Dr. Darius Ebrahimi-Fakhari, M.D., Ph.D., Boston Children’s Hospital (Principal Investigator).
“Having overseen this therapeutic innovation from discovery stage, it is a huge source of excitement to reach this important IND milestone. Given the incredibly high unmet need for children with this devastating condition, we are on a mission at BlackfinBio to transform treatment options for these young lives and make a real impact to the lives of their families,” added Professor Mimoun Azzouz, Founder and Chief Scientific Officer of BlackfinBio Ltd.
Reference:
(1):
Pre-clinical development of AP4B1 gene replacement therapy for hereditary spastic paraplegia type 47
Disclosure Statement:
Dr. Darius Ebrahimi-Fakhari has consulted for BlackfinBio.
About BFB-101
BFB-101 aims to address the underlying genetic cause of Hereditary Spastic Paraplegia, Type 47 (SPG47) by delivering a functional copy of the AP4B1 gene, with the goal of halting or reversing disease progression. It is administered via the brain as a single lifetime dose and has shown potential in restoring AP-4 function
in vitro
and improving motor function in AP4B1 mutant mice. BFB-101 was originally developed by Professor Mimoun Azzouz at the University of Sheffield with the support of Cure AP-4 (
https://cureap4.org
), LifeArc (
www.lifearc.org
). This project has also received partial funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No 945473 to support AAV9 capsid synthesis for immune response study. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA. The FDA has granted an orphan drug designation (ODD) and rare pediatric disease designation (RPDD) to BFB-101 for the treatment of SPG47.
About BlackfinBio Ltd
BlackfinBio is a clinical stage gene therapy company focused on the development of treatments for rare neurological diseases. The pipeline comprises BFB-101, a clinical stage AP4B1 replacement AAV gene therapy for spastic paraplegia 47 – an ultra-rare genetic neurological disease for which no treatment currently exists. BFB-201 is a preclinical stage gene therapy to treat several rare dopamine deficiency disorders. For more information, please visit
https://blackfin.bio
and follow us on
LinkedIn
.
SOURCE:
BlackfinBio
基因疗法临床研究孤儿药临床申请
2025-03-17
·美通社
——
Longuette&Ankura Pro裙边分支重建系统,开启烟囱技术"防漏+久畅"双核时代
深圳
2025年3月17日
/美通社/ -- 近日,中国医学科学院阜外医院舒畅教授领衔与先健科技(股份代号:1302.HK)联合研发的主动脉分支重建覆膜支架系统(烟囱型)成功获得国家药品监督管理局(NMPA)
批准上市
!
该产品为全球首款针对烟囱技术重建弓部分支的主动脉-分支动脉覆膜支架系统,由Longuette裙边支架(长)和Ankura Pro主动脉主体覆膜支架系统组成,不仅为术者提供了一款应对复杂弓部病变的创新良器,为弓部病变患者带来了更优的治疗新选择,更是中国血管介入领域迈向国际领先水平的重要里程碑!
以技术革新重新定义烟囱技术新标准
主动脉弓部病变(如夹层、动脉瘤)因涉及弓上三大分支动脉,即无名动脉、左颈总动脉和左锁骨下动脉,加之各种复杂的解剖学形态,一直以来是腔内治疗的难点。烟囱支架技术由于操作简便、安全性高、手术成功率高,在弓部分支扭曲、解剖形态复杂、急诊等情况下更具优势
[1]
。
然而,由于烟囱技术在累及主动脉弓部病变的临床应用上长期存在Ⅰa型内漏率高(约10.7%~19%)
[
2-5
]
和远期分支闭塞(约10.7%)
[6]
两大痛点,使用受到限制。
为了攻克烟囱技术固有的短板和瓶颈,国家心血管病中心、中国医学科学院阜外医院舒畅教授团队与先健联合研发了针对烟囱技术的主动防漏裙边支架—Longuette裙边支架(长)。
该创新解决方案于2021年进入国家创新医疗器械特别审查程序"绿色通道",并通过由舒畅教授牵头开展的"前瞻性、多中心、单组目标值法"上市前临床研究,充分证明了该产品在治疗近左锁骨下动脉的Stanford B型夹层病变的安全和有效性(共纳入患者150例),成为全球首款获批上市的烟囱技术分支重建专用覆膜支架系统。
该创新产品为广大临床医生提供了前所未有的标准化的烟囱技术专用器械,通过结构性创新攻克了烟囱技术内漏与闭塞的双重难题,同时极大程度地保留了烟囱技术的临床优势,实现了效率与精准并行,防漏与通畅共存,使烟囱技术能够在主动脉弓部疾病治疗中发挥更加重要的作用,从而为患者带来更大的临床获益。
医工结合汇集交融的中国智慧,重新定义烟囱技术新标准,让我国在血管介入治疗领域的自主创新能力达到又一新高度,并将为全球行业的发展和进步注入强劲的中国创新力量!
五重维度赋予烟囱技术新能量
主动脉分支重建覆膜支架(烟囱型)通过五个维度的创新,开启烟囱技术"防漏+久畅"双核时代。其中Ankura Pro主体支架采用ePTFE覆膜,拥有多种长度和锥度设计,可满足不同的临床需求。主动防漏Longuette裙边支架(长)采用带外层裙边的双层结构设计,重建分支血管可预防内漏发生,降低并发症风险。
无隙可击
外层超薄ePTFE覆膜"自适应裙边"设计,主动防漏
即刻Ia型内漏发生率较传统烟囱技术直降64.20%
[2-4
]
,术后3年随访内漏率仅2.80%
通畅标杆
内层镍钛合金骨架,近端刚性支撑,增强抗挤压能力,抗挤压强度对比传统烟囱技术提升85.71%
远端柔性段顺应分支血管形态,内刚外柔,无惧扭曲,避免远期闭塞风险
术后1年分支通畅率高达98.58%,较传统烟囱技术大幅提升
急诊利器
操作简便,拿来即用,分支重建效率与精准并行,急诊抢救争分夺秒
即刻技术成功率达99.33%
高效安全
术后1年患者存活率98.00%,无卒中、截瘫事件发生
全域适配
6-20mm直径全系规格覆盖,灵活适配各种复杂解剖
先健主动脉分支重建覆膜支架系统(烟囱型)这一中国医工结合创新的典范之作,既保留了烟囱技术的"快"和"简",又创新性的攻克了内漏和远期闭塞的"痛",拓宽了高危弓部病变的腔内治疗适应症,使更多临床医生能够高效快速地完成复杂主动脉弓部分支重建,不仅是为烟囱技术重新定义新标准、赋予新能量,更是在不断拓展腔内治疗的边界!
构建弓部分支重建完整解决方案引领行业未来
先健主动脉分支重建覆膜支架系统(烟囱型)——Longuette 裙边支架(长)& Ankura Pro主动脉主体覆膜支架系统的获批上市,进一步丰富和完善了先健的创新版图。该项创新产品的问世,不仅重铸了主动脉弓部病变患者的生命之盾,更成为推动中国医疗行业迈向创新发展高地的有力引擎。
随着CSkirt 裙边支架(短)和CS三分支覆膜支架重建系统上市注册进程的持续推进,未来先健主动脉弓部分支重建整体解决方案将让患者获得更精准和个性化的治疗。这不仅展现了先健在技术创新上的雄厚实力,亦在全球医疗领域树立了中国智造的新标杆,为行业整体技术突破和在国际医疗创新影响力的提升上注入源源不断的动力。
创新智愈
无限未来
未来,先健将继续携手广大临床专家,深度聚焦血管健康领域的前沿探索与创新实践,全力攻克行业技术"制高点",让更多中国医疗创新智慧跨越山海,守护亿万生命的澎湃律动!
参考文献
[1]
中华医学会外科学分会血管外科学组 海峡两岸医药卫生交流协会血管外科分会 中国医师协会血管外科医师分会胸主动脉学组. 主动脉腔内修复术中弓上分支重建技术应用专家共识(2024版)[J]. 中华外科杂志, 2024, 62(12): 1069-1081.DOI: 10.3760/cma.j.cn112139-20241003-00449.
[2]
HuangW, DingH, JiangM, et al. Outcomes of chimney technique for aortic arch diseases: a single-center experience with 226 cases[J]. Clin Interv Aging, 2019, 14:1829-1840.DOI: 10.2147/CIA.S222948.
[3]
ShuC, FanB, LuoM, et al. Endovascular treatment for aortic arch pathologies: chimney, on-the-table fenestration, and in-situ fenestration techniques[J]. J Thorac Dis, 2020, 12(4):1437-1448. DOI: 10.21037/jtd.2020.03.10.
[4]
ZhangL, WuMT, ZhuGL, et al. Off-the-shelf devices for treatment of thoracic aortic diseases: midterm follow-up of TEVAR with chimneys or physician-made fenestrations[J]. J Endovasc Ther, 2020, 27(1):132-142. DOI:10.1177/1526602819890107.
[5]
Huanyu Ding
a,c,Yuan Liu a,c,Nianjin Xie, et al. Outcomes of Chimney Technique for Preservation of the Left Subclavian Artery in Type B Aortic Dissection. Eur J Vasc Endovasc Surg (2019) 57, 374–381.
[6]
Mangialardi N. ,Serrao E.,Kasemi H, Alberti V.Fazzini S. Ronchey S Chimney technique for aortic arch pathologies: an 11-year single-center experience. J Endovasc Ther 2014 Apr,21(2):312-23.
关于先健科技:
先健科技公司(股份代号:1302.HK)是业内领先的心脑血管和外周血管介入医疗器械企业,于1999年成立于中国深圳,为国家级高新技术企业,国家工信部第三批专精特新"小巨人"企业。公司在研、在售产品涵盖结构性心脏病、外周血管病、起搏电生理
、神经介入
等领域,并拥有全球首创的铁基生物可吸收材料平台,在多个细分领域实现自主创新技术突破。截至2024年6月30日,公司已实现高质量专利布局2,198项,目前累计15款产品获NMPA批准进入"创新医疗器械特别审查程序"。 秉承"创新"和"国际化"发展战略,公司主要在售产品的市场份额长期处于国内领先地位,并拥有7个境外子公司,销售网络覆盖全球近120个国家和地区,是国内少有的业务具高度国际化的三类介入医疗器械企业。
上市批准
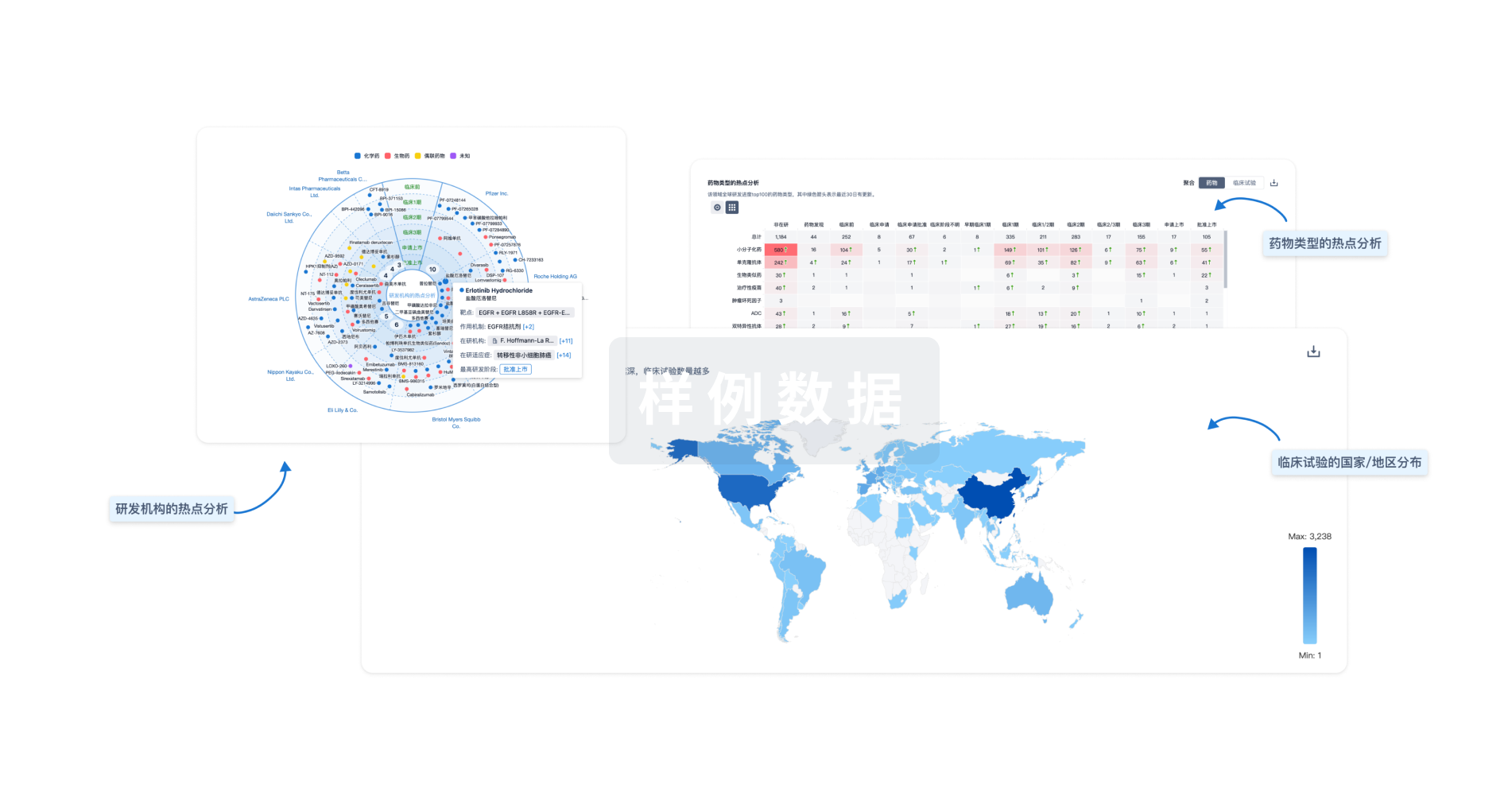
分析
对领域进行一次全面的分析。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用



