预约演示
更新于:2025-05-07
BRAF V600 mutation low-grade glioma
BRAF V600 突变的低级别胶质瘤
更新于:2025-05-07
基本信息
别名- |
简介- |
关联
2
项与 BRAF V600 突变的低级别胶质瘤 相关的药物作用机制 PDK1抑制剂 [+1] |
在研适应症 |
非在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2024-04-23 |
作用机制 MEK1抑制剂 [+1] |
原研机构 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2013-05-29 |
2
项与 BRAF V600 突变的低级别胶质瘤 相关的临床试验NCT05760586
Expanded Access to the Oral Pan-RAF Inhibitor DAY101 in Pediatric Patients With RAF-Altered, Relapsed or Refractory Low-Grade Glioma
The DAY101-EAP is a multicenter, open-label, expanded access treatment protocol designed to provide access to tovorafenib (DAY101) for eligible patients.
开始日期- |
NCT04775485
FIREFLY-1: A Phase 2, Open-Label, Multicenter Study to Evaluate the Safety and Efficacy of the Oral Pan-RAF Inhibitor DAY101 in Pediatric Patients With RAF-Altered, Recurrent or Progressive Low-Grade Glioma and Advanced Solid Tumors
This is a Phase 2, multi center, open-label study to evaluate the safety and efficacy of Type II RAF (tovorafenib) in pediatric participants with low-grade glioma or advanced solid tumors. Qualifying genomic alterations will be identified through molecular assays as routinely performed at Clinical Laboratory Improvement Amendments (CLIA) of 1988 or other similarly certified laboratories prior to enrollment into any of the arms. The study will consist of a screening period, a treatment period, a long-term extension phase, end of treatment (EOT) visit(s), a safety follow-up visit, and long-term follow-up assessments.
开始日期2021-04-22 |
申办/合作机构 |
100 项与 BRAF V600 突变的低级别胶质瘤 相关的临床结果
登录后查看更多信息
100 项与 BRAF V600 突变的低级别胶质瘤 相关的转化医学
登录后查看更多信息
0 项与 BRAF V600 突变的低级别胶质瘤 相关的专利(医药)
登录后查看更多信息
23
项与 BRAF V600 突变的低级别胶质瘤 相关的文献(医药)2025-03-01·Pediatric Blood & Cancer
Resolution of Ascites Following Targeted Therapy in a Pediatric Patient With BRAF V600E‐Mutant Low‐Grade Glioma
Letter
作者: Crentsil, Hannah E. ; Felker, James
2024-10-01·Child's Nervous System
Molecular-targeted therapy for childhood low-grade glial and glioneuronal tumors
Review
作者: Kilburn, Lindsay B ; Siegel, Benjamin I ; Packer, Roger J ; Duke, Elizabeth S
2024-05-15·Clinical Cancer Research
Response Rate and Molecular Correlates to Encorafenib and Binimetinib in BRAF-V600E Mutant High-Grade Glioma
Article
作者: Iacoboni, Michaella ; Tan, Sze K. ; Turakulov, Rust ; Schreck, Karisa C. ; Bettegowda, Chetan ; Rudek, Michelle A. ; Desideri, Serena ; Ye, Xiaobu ; Chang, Michael ; Nabors, Louis B. ; Ellingson, Benjamin M. ; Danda, Neeraja ; Aldape, Kenneth ; Surakus, Trisha ; Fisher, Joy ; Abdullaev, Zied ; Grossman, Stuart A. ; Strowd, Roy E.
19
项与 BRAF V600 突变的低级别胶质瘤 相关的新闻(医药)2024-08-09
On June 22, 2022, the Food and Drug Administration granted accelerated approval to dabrafenib (Tafinlar, Novartis) in combination with trametinib (Mekinist, Novartis) for the treatment of adult and pediatric patients ≥ 6 years of age with unresectable or metastatic solid tumors with BRAF V600E mutation who have progressed following prior treatment and have no satisfactory alternative treatment options. Dabrafenib in combination with trametinib is not indicated for patients with colorectal cancer because of known intrinsic resistance to BRAF inhibition. Dabrafenib is not indicated for patients with wild-type BRAF solid tumors.
The safety and efficacy were evaluated in 131 adult patients from open-label, multiple cohort trials BRF117019 (NCT02034110) and NCI-MATCH (NCT02465060), 36 pediatric patients from CTMT212X2101 (NCT02124772), and supported by results in COMBI-d, COMBI-v, and BRF113928 (studies in melanoma and lung cancer already described in product labeling). Study BRF117019 enrolled patients with BRAF V600E mutation positive specific solid tumors including high grade glioma (HGG), biliary tract cancer, low grade glioma (LGG), adenocarcinoma of small intestine, gastrointestinal stromal tumor, and anaplastic thyroid cancer (ATC). NCI-MATCH Subprotocol H enrolled adult patients with BRAF V600E mutation positive solid tumors except patients with melanoma, thyroid cancer, or CRC. Parts C and D of Study CTMT212X2101 enrolled 36 pediatric patients with BRAF V600 refractory or recurrent LGG or HGG. The major efficacy outcome measure of these studies was overall response rate (ORR) using standard response criteria. For the 131 adult patients, a total of 54 (41%, 95% CI: 33, 50) experienced an objective response. The studies enrolled patients with 24 tumor types, including different subtypes of LGG and HGG. Among the highest representative tumor types, ORR was 46% (95% CI: 31, 61) for biliary tract cancer, 33% (95% CI: 20, 48) for high grade glioma (combined) and 50% (95% CI: 23, 77) for low grade glioma (combined). For the 36 pediatric patients, the ORR was 25% (95% CI: 12, 42); DOR was ≥6 months for 78% of patients and ≥24 months for 44% of patients.
The most common (≥20%) adverse reactions in adult patients were pyrexia, fatigue, nausea, rash, chills, headache, hemorrhage, cough, vomiting, constipation, diarrhea, myalgia, arthralgia, and edema.
The most common (≥20%) adverse reactions in pediatric patients were pyrexia, rash, vomiting, fatigue, dry skin, cough, diarrhea, dermatitis acneiform, headache, abdominal pain, nausea, hemorrhage, constipation, and paronychia.
The recommended dabrafenib dose in adult patients is 150 mg (two 75 mg capsules) orally twice daily in combination with trametinib 2 mg orally once daily. The recommended doses for dabrafenib and trametinib in pediatric patients are based on body weight. A recommended dose has not been established in patients who weigh less than 26 kg.
View full prescribing information for Tafinlar and Mekinist
This review used the Assessment Aid, a voluntary submission from the applicant to facilitate the FDA’s assessment.
This application was granted priority review. A description of FDA expedited programs is in the Guidance for Industry: Expedited Programs for Serious Conditions-Drugs and Biologics.
Healthcare professionals should report all serious adverse events suspected to be associated with the use of any medicine and device to FDA’s MedWatch Reporting System or by calling 1-800-FDA-1088.
For assistance with single-patient INDs for investigational oncology products, healthcare professionals may contact OCE’s Project Facilitate at 240-402-0004 or email OncProjectFacilitate@fda.hhs.gov
For information on the COVID-19 pandemic, see the following resources:
FDA: Coronavirus Disease 2019 (COVID-19) NCI: Coronavirus: What People With Cancer Should Know CDC: Coronavirus (COVID-19)
Follow the Oncology Center of Excellence on Twitter @FDAOncologyExternal Link Disclaimer
Content current as of: 06/23/2022 Regulated Product(s) Drugs
06/23/2022
Drugs
Resources for Information | Approved Drugs Oncology (Cancer)/Hematologic Malignancies Approval Notifications Ongoing | Cancer Accelerated Approvals Verified Clinical Benefit | Cancer Accelerated Approvals Withdrawn | Cancer Accelerated Approvals Other | Cancer Accelerated Approvals Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book) Short Description
Oncology (Cancer)/Hematologic Malignancies Approval Notifications Ongoing | Cancer Accelerated Approvals Verified Clinical Benefit | Cancer Accelerated Approvals Withdrawn | Cancer Accelerated Approvals Other | Cancer Accelerated Approvals
Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book) Short Description
优先审批加速审批上市批准临床结果
2024-02-29
According to Lutris’ CEO, approximately 75% of cancer patients taking EGFR inhibitors develop some form of acneiform rash. Credit: sruilk via Getty Images.
Lutris Pharma has secured a US Food and Drug Administration (FDA) orphan drug designation for its BRAF inhibitor LUT014, an investigational treatment for epidermal growth factor receptor (EGFR) inhibitor-induced acneiform rash.
EGFR inhibitors are used as cancer treatments, however dermatological side effects such as acneiform lesions, a skin condition that appears as small red bumps or pustules on the skin, seen with the treatment affect a patient’s quality of life.
Approximately 75% of cancer patients taking EGFR inhibitors develop some form of an acneiform rash, according to Lutris’ CEO Noa Shelach.
The candidate is currently being investigated in a Phase II clinical trial (NCT04759664), where 117 colorectal cancer patients with acneiform lesions receive either one of two strengths of LUT014 gel once a day for four weeks, or a placebo.
EGFR, found on cell surfaces including skin, regulates crucial cell functions such as growth and differentiation. It is overexpressed in cancer cells, so EGFR inhibitors prevent cancer cells from receiving the signals needed for growth and division, inhibiting tumour growth and progression.
See Also:
Market cap upturn for top 20 biopharma companies with obesity drug successes in 2023
LUT014 is a BRAF inhibitor that is applied topically on the skin. BRAF, a protein translated from the BRA gene, is part of the EGFR signalling pathway. LUT014 helps to shrink tumours with mutated BRAF proteins while promoting cell growth in normal cells, helpful for conditions such as an acneiform rash.
In the announcement accompanying the orphan drug designation, Shelach said: “Receipt of orphan drug designation for LUT014 is strategically important for Lutris, as it reflects the significant unmet need for patients treated with EGFR inhibitors.
“The [orphan drug designation] also qualifies us for incentives including tax credits for qualified clinical trials, exemption from user fees and potentially seven years of marketing exclusivity for LUT014, should it gain approval for the treatment of EGFRI inhibitor acneiform rash.”
In March 2023, the
FDA approved Novartis’
Tafinlar and Mekinist BRAF/MEK inhibitor combination therapy to treat BRAF V600E low-grade glioma in paediatric patients starting at one year of age. Findings from the Phase II/III TADPOLE trial listed acneiform rash as one of the most common adverse reactions observed.

临床2期孤儿药上市批准临床结果
2023-08-03
Announced Positive Results from Phase 2 Portion of the RISE UP Pivotal Study of Mitapivat in Sickle Cell Disease; On Track to Enroll First Patient in Phase 3 Portion of the Study in Q4 2023
Completed Enrollment in Phase 3 ENERGIZE and ENERGIZE-T Studies of Mitapivat in Thalassemia and Phase 2a Study of AG-946 in Lower-Risk MDS
Announced Exclusive Worldwide License Agreement with Alnylam Pharmaceuticals to License Alnylam’s Novel siRNA for the Potential Treatment of Polycythemia Vera
U.S. PYRUKYND® (mitapivat) Net Revenue of $6.7 Million in Q2; $947 Million of Cash, Cash Equivalents and Marketable Securities as of June 30, 2023
CAMBRIDGE, Mass., Aug. 03, 2023 (GLOBE NEWSWIRE) -- Agios Pharmaceuticals, Inc. (Nasdaq: AGIO), a leader in the field of cellular metabolism pioneering therapies for rare diseases, today reported business highlights and financial results for the second quarter ended June 30, 2023.
“Since our last quarterly update, Agios has made tremendous progress executing across our industry-leading pipeline of PK activators, and today we are further expanding our portfolio beyond PK activation through focused business development,” said Brian Goff, chief executive officer at Agios. “We announced positive data from the Phase 2 portion of the RISE UP study of mitapivat in sickle cell disease, completed enrollment in three clinical studies, licensed a compelling preclinical program from Alnylam, and continued to strengthen our commercial capabilities to support future anticipated launches. We look forward to the readout of the Phase 2a study of AG-946 in lower-risk MDS by the end of this year and the readouts of the Phase 3 studies of mitapivat in thalassemia next year.”
Second Quarter 2023 & Recent Highlights
PYRUKYND® U.S. Launch: Generated $6.7 million in U.S. net revenue for the second quarter of 2023, a 20 percent increase over the first quarter of 2023. A total of 147 unique patients have completed prescription enrollment forms, representing an increase of 16 percent over the first quarter of 2023. A total of 99 patients are on PYRUKYND® therapy, representing an 11 percent increase over the first quarter of 2023.
Sickle Cell Disease: Announced positive results from the Phase 2 portion of the RISE UP pivotal study of mitapivat in sickle cell disease.
Thalassemia: Completed enrollment of the Phase 3 ENERGIZE and ENERGIZE-T studies of mitapivat in not regularly transfused and regularly transfused adults with thalassemia, respectively.
Lower-Risk Myelodysplastic Syndromes (LR-MDS): Completed enrollment of the Phase 2a study of AG-946 in LR-MDS.
Business Development: Announced an exclusive worldwide license agreement with Alnylam Pharmaceuticals for a novel siRNA for the potential treatment of polycythemia vera.
Leadership:Appointed Catherine Owens to the board of directors. Kaye Foster assumed the role of lead independent director.
Other: Data from Servier’s Phase 3 trial of vorasidenib in patients with residual or recurrent IDH mutant low-grade glioma were presented during the plenary session at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting and published in the New England Journal of Medicine. As part of the divestiture of Agios’ oncology business to Servier, Agios retains rights to a potential $200 million milestone upon FDA approval of vorasidenib and 15% royalties on potential U.S. net sales.
Key Upcoming Milestones & Priorities
Agios expects to execute on the following additional key milestones and priorities by the end of 2023:
Pediatric PK Deficiency: Enroll more than half of patients in the Phase 3 ACTIVATE-kids and ACTIVATE-kidsT studies of mitapivat.
Sickle Cell Disease: Enroll first patient in Phase 3 portion of RISE UP study of mitapivat, with the 100 mg dose selected from the successful Phase 2 portion.
Lower-risk Myelodysplastic Syndromes (LR-MDS): Announce data from the Phase 2a study of novel PK activator AG-946.
Pipeline: File investigational new drug (IND) application for phenylalanine hydroxylase (PAH) stabilizer for the treatment of phenylketonuria (PKU).
Second Quarter 2023 Financial Results
Revenue: Net U.S. product revenue from sales of PYRUKYND® for the second quarter of 2023 was $6.7 million, compared to $3.1 million for the second quarter of 2022. PYRUKYND® received FDA approval on February 17, 2022.
Cost of Sales: Cost of sales for the second quarter of 2023 was $1.1 million.
Research and Development (R&D) Expenses: R&D expenses were $68.9 million for the second quarter of 2023 compared to $74.5 million for the second quarter of 2022. The year-over-year decrease was primarily driven by a decrease in workforce related expenses as a result of reduced headcount related to the evolution of our research organization.
Selling, General and Administrative (SG&A) Expenses: SG&A expenses were $30.4 million for the second quarter of 2023 compared to $28.3 million for the second quarter of 2022. The year-over-year increase was primarily attributable to an increase in stock-based compensation expense.
Net Loss: Net loss was $83.8 million for the second quarter of 2023 compared to $91.8 million for the second quarter of 2022.
Cash Position and Guidance: Cash, cash equivalents and marketable securities as of June 30, 2023, were $946.9 million compared to $1.1 billion as of December 31, 2022. Agios expects that its cash, cash equivalents and marketable securities together with anticipated product revenue, interest income and vorasidenib milestone will enable the company to fund its operating expenses and capital expenditures at least into 2026. This does not include potential royalties from vorasidenib, commercializing mitapivat outside of the U.S. through one or more partnerships, or other potential strategic business or financial agreements.
Conference Call Information
Agios will host a conference call and live webcast with slides today at 8:00 a.m. ET to discuss second quarter 2023 financial results and recent business activities. The live webcast can be accessed under “Events & Presentations” in the Investors section of the company’s website at The archived webcast will be available on the company's website beginning approximately two hours after the event.
About Agios
Agios is the pioneering leader in PK activation and is dedicated to developing and delivering transformative therapies for patients living with rare diseases. In the U.S., Agios markets a first-in-class pyruvate kinase (PK) activator for adults with PK deficiency, the first disease-modifying therapy for this rare, lifelong, debilitating hemolytic anemia. Building on the company's leadership in the field of cellular metabolism and rare hematologic diseases, Agios is advancing a robust clinical pipeline of investigational medicines with programs in alpha- and beta-thalassemia, sickle cell disease, pediatric PK deficiency and MDS-associated anemia. In addition to its clinical pipeline, Agios has TMPRSS6 siRNA as a potential treatment for polycythemia vera, a PAH stabilizer in preclinical development as a potential treatment for phenylketonuria (PKU), and deep scientific expertise in classical hematology. For more information, please visit the company’s website at .
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Such forward-looking statements include those regarding the potential benefits of PYRUKYND® (mitapivat), AG-946, TMPRSS6 siRNA and its PAH stabilizer; Agios’ plans, strategies and expectations for its preclinical, clinical and commercial advancement of its drug development, including PYRUKYND®, AG-946 and its PAH stabilizer; Agios’ strategic vision and goals, including its key milestones for 2023; and the potential benefits of Agios’ strategic plans and focus. The words “anticipate,” “expect,” “goal,” “hope,” “milestone,” “plan,” “potential,” “possible,” “strategy,” “will,” “vision,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Such statements are subject to numerous important factors, risks and uncertainties that may cause actual events or results to differ materially from Agios’ current expectations and beliefs. For example, there can be no guarantee that any product candidate Agios is developing will successfully commence or complete necessary preclinical and clinical development phases, or that development of any of Agios’ product candidates will successfully continue. There can be no guarantee that any positive developments in Agios’ business will result in stock price appreciation. Management's expectations and, therefore, any forward-looking statements in this press release could also be affected by risks and uncertainties relating to a number of other important factors, including, without limitation: risks and uncertainties related to the impact of the COVID-19 pandemic or other public health emergencies to Agios’ business, operations, strategy, goals and anticipated milestones, including its ongoing and planned research activities, ability to conduct ongoing and planned clinical trials, clinical supply of current or future drug candidates, commercial supply of current or future approved products, and launching, marketing and selling current or future approved products; Agios’ results of clinical trials and preclinical studies, including subsequent analysis of existing data and new data received from ongoing and future studies; the content and timing of decisions made by the U.S. FDA, the EMA or other regulatory authorities, investigational review boards at clinical trial sites and publication review bodies; Agios’ ability to obtain and maintain requisite regulatory approvals and to enroll patients in its planned clinical trials; unplanned cash requirements and expenditures; competitive factors; Agios' ability to obtain, maintain and enforce patent and other intellectual property protection for any product candidates it is developing; Agios’ ability to maintain key collaborations; uncertainty regarding any milestone or royalty payments related to the sale of its oncology business or its in-licensing of TMPRSS6 siRNA, and the uncertainty of the timing of any such payments; uncertainty of the results and effectiveness of the use of proceeds from the transaction with Servier; and general economic and market conditions. These and other risks are described in greater detail under the caption "Risk Factors" included in Agios’ public filings with the Securities and Exchange Commission. Any forward-looking statements contained in this press release speak only as of the date hereof, and Agios expressly disclaims any obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
Consolidated Balance Sheet Data
(in thousands)
(Unaudited)
June 30, 2023
December 31, 2022
Cash, cash equivalents, and marketable securities
$
946,923
$
1,096,993
Accounts receivable, net
2,251
2,206
Inventory
15,671
8,492
Total assets
1,085,153
1,238,718
Stockholders' equity
964,236
1,100,814
Consolidated Statements of Operations Data
(in thousands, except share and per share data)
(Unaudited)
Three Months Ended June 30,
Six Months Ended June 30,
2023
2022
2023
2022
Revenues:
Product revenue, net
$
6,712
$
3,082
$
12,321
$
3,914
Milestone revenue
—
2,500
—
2,500
Total revenue
6,712
5,582
12,321
6,414
Operating expenses:
Cost of sales
$
1,108
$
435
$
1,662
$
774
Research and development
68,895
74,523
136,196
144,646
Selling, general and administrative
30,409
28,264
58,776
59,779
Total operating expenses
100,412
103,222
196,634
205,199
Loss from operations
(93,700
)
(97,640
)
(184,313
)
(198,785
)
Royalty income from gain on sale of oncology business
—
2,704
—
5,408
Interest income, net
8,254
1,793
16,345
2,487
Other income, net
1,640
1,337
3,144
4,310
Net loss
$
(83,806
)
$
(91,806
)
$
(164,824
)
$
(186,580
)
Net loss per share - basic and diluted
$
(1.51
)
$
(1.68
)
$
(2.97
)
$
(3.41
)
Weighted-average number of common shares used in computing net loss per share – basic and diluted
55,604,330
54,799,680
55,435,796
54,678,249
Contacts:
Investor Contact
Chris Taylor, VP Investor Relations and Corporate Communications
Agios Pharmaceuticals
IR@agios.com
Media Contact
Dan Budwick
1AB Media
dan@1abmedia.com
临床2期临床3期财报引进/卖出高管变更
分析
对领域进行一次全面的分析。
登录
或
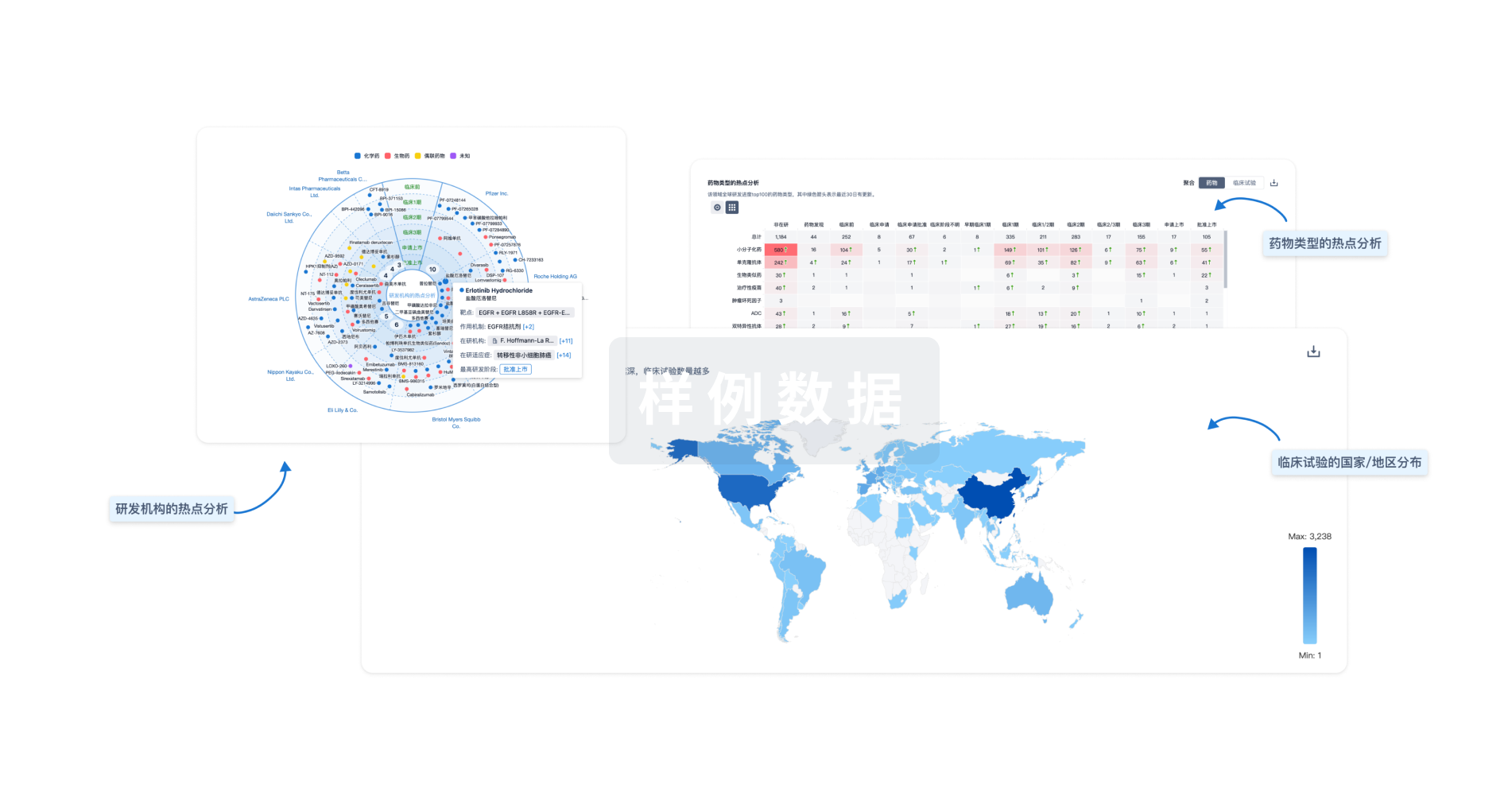
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


