预约演示
更新于:2025-01-23
USP1 x PARP
更新于:2025-01-23
关联
1
项与 USP1 x PARP 相关的药物作用机制 PARP抑制剂 [+1] |
在研机构 |
原研机构 |
在研适应症 |
非在研适应症- |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
100 项与 USP1 x PARP 相关的临床结果
登录后查看更多信息
100 项与 USP1 x PARP 相关的转化医学
登录后查看更多信息
0 项与 USP1 x PARP 相关的专利(医药)
登录后查看更多信息
7
项与 USP1 x PARP 相关的文献(医药)2024-11-15·Science Advances
USP1 deubiquitinates PARP1 to regulate its trapping and PARylation activity
Article
作者: Rampioni Vinciguerra, Gian Luca ; Giorda, Giorgio ; Gambelli, Alice ; Nespolo, Anna ; Karimbayli, Javad ; D’Andrea, Sara ; Puglisi, Fabio ; Schiappacassi, Monica ; Belletti, Barbara ; Nicoloso, Milena S. ; Segatto, Ilenia ; Canzonieri, Vincenzo ; Baldassarre, Gustavo ; Barozzi, Sara ; Pellarin, Ilenia ; Stefenatti, Linda ; Spizzo, Riccardo ; Orsenigo, Fabrizio ; Bartoletti, Michele ; Sonego, Maura ; Lucia, Emilio
2024-10-15·Cancer Research
Single-Stranded DNA Gap Accumulation Is a Functional Biomarker for USP1 Inhibitor Sensitivity
Article
作者: Lamarre, Benjamin P. ; Parmar, Kalindi ; Sadatrezaei, Golbahar ; Nguyen, Huy ; Ravindranathan, Ramya ; D’Andrea, Alan D. ; Moreau, Lisa ; Mukkavalli, Sirisha ; Iyer, Divya R. ; Somuncu, Ozge ; Liu, Joyce ; Shapiro, Geoffrey I. ; Martignetti, David B. ; Jiao, Yuqing ; Lazaro, Jean-Bernard ; da Costa, Alexandre A.
2024-10-15·Cancer Research
The USP1 Inhibitor KSQ-4279 Overcomes PARP Inhibitor Resistance in Homologous Recombination–Deficient Tumors
Article
作者: Schlabach, Michael ; Gannon, Hugh ; Cadzow, Louise ; Wylie, Andrew A. ; Chipumuro, Edmond ; Hixon, Jeff ; Dodson, Anne E. ; Mishina, Yuji ; Griffith, Jim ; Ganapathy, Suthakar ; Grasberger, Paula ; Wilt, Jeremy ; Krall, Elsa ; Matulonis, Ursula A. ; Shenker, Sol ; Gokhale, Prafulla C. ; Murray, Morgan ; Olaharski, Andrew ; Ali, Janid A. ; Sangurdekar, Dipen ; McGuire, Michael ; Sinkevicius, Kerstin ; Sullivan, Pamela ; Nayak, Sumeet ; Stegmeier, Frank ; Liu, Hanlan ; Liu, Joyce F. ; Brenneman, Jehrod ; Tobin, Erica
86
项与 USP1 x PARP 相关的新闻(医药)2025-01-17
·氨基观察
氨基观察-创新药组原创出品
作者 | 黄凯
今年的医保目录调整时间将提前。
1月17日,国家医保局召开医保部门“保障人民健康 赋能经济发展”主题新闻发布会,国家医保局有关负责人表示,今年的医保药品目录调整时间将提前。
此外,由于丙类目录需要与商业保险产品对接,需要留有一定时间,从今年开始国家基本医保药品目录的调整时间会适当提前,预计4月初开始申报,争取9月份完成。
为了尽量减少对行业的影响,医保局拟采取预申报的过渡措施,预计今年5月底前获批的新药可提前进行申报,在专家评审前正式获批就可以参加评审。
同时,关于行业关注的丙类目录,国家医疗保障局医药服务管理司司长黄心宇指出,丙类目录与每年的基本药品目录调整同步开展,计划在今年年内发布第一版,工作程序也参照基本医保药品目录的调整。
继艾伯维之后,又一家药企在精神分裂症栽了跟头。
1月16日,勃林格殷格翰公布iclepertin治疗成人精神分裂症认知障碍的3期临床项目CONNEX的主要结果,显示主要终点和关键次要终点均未达到。勃林格殷格翰决定立即终止长期拓展试验CONNEX-X。
在过去的一天里,国内外医药市场还有哪些热点值得关注?让氨基君带你一探究竟。
/ 01 /
市场速递
1)医保目录预计4月初开始申报,丙类目录年内推出
1月17日,国家医保局召开医保部门“保障人民健康 赋能经济发展”主题新闻发布会,国家医保局有关负责人表示,今年的医保药品目录调整时间将提前。
此外,由于丙类目录需要与商业保险产品对接,需要留有一定时间,从今年开始国家基本医保药品目录的调整时间会适当提前,预计4月初开始申报,争取9月份完成。
为了尽量减少对行业的影响,医保局拟采取预申报的过渡措施,预计今年5月底前获批的新药可提前进行申报,在专家评审前正式获批就可以参加评审。
关于行业关注的丙类目录,国家医保局医药服务管理司司长黄心宇指出,丙类目录与每年的基本药品目录调整同步开展,计划在今年年内发布第一版,工作程序也参照基本医保药品目录的调整。
在支持落地应用上,国家医保局将探索优化医保的支付管理政策,对于丙类目录可不计入参保人自费率的考核,和集采中选可替代药品的监测范围,符合条件的病例也可以不纳入按病种付费的范围,暂时实行按项目付费。具体的工作方案医保局将尽快进行完善、尽快向社会公开征求意见。
2)医保局2024年追回医保基金275亿元
医保局架起监管高压线。
1月17日,国家医保相关负责人在发布会上表示,2024年,全国共追回医保基金275亿元,查实欺诈骗保机构2008家,联合公安机关侦办医保案件3018起,抓获犯罪嫌疑人10741名。
一批欺诈骗保机构被解除或暂停协议,并移送公安、纪检等部门立案调查,真正架起监管的高压线。
国家医保局研究开发一系列管用有效的大数据监管模型,为飞行检查、打击欺诈骗保、精准监管提供有力支撑。
3)先声药业与费米子科技就SSTR4 靶点镇痛药物达成合作
1月17日,先声药业和广州费米子科技共同宣布,双方已就一款靶向SSTR4的临床阶段镇痛候选创新药 FZ002-037 达成合作。根据协议,先声药业将获得大中华地区(中国大陆、香港、澳门和台湾)开发和商业化 FZ002-037的独家权利。费米子科技将获得首付款及里程碑付款,以及基于未来销售额的分层特许权使用费。
4)流感高峰正在过去
1月17日,国家卫生健康委举行新闻发布会,介绍时令节气与健康有关情况。中国疾病预防控制中心病毒病预防控制所研究员王大燕介绍,近期的流感高发属于正常的季节性流行,每年冬春季都会出现。
这次流感的流行强度低于上个冬季流行季。最新一周监测数据显示,北方大部分省份流感病毒检测阳性率开始下降,南方个别省份也开始出现下降。
/ 02 /
资本信息
1)艾威药业完成A++轮融资
1月17日,艾威药业宣布完成A++轮融资,本轮融资由重庆长生胜合投资基金领投,杭州玖润投资、杭州易路医疗跟投, 原有投资者Proxima Ventures Fund I L.P 、比邻星创投继续加持投资。
根据新闻稿,本轮融资募集资金将用于推进艾威药业研发管线项目的临床试验及注册,主要支持该公司研发的创新靶点治疗干眼症的1类创新化学药IVW-1001的临床研究,该项目已在美国10个临床中心完成治疗干眼症临床1/2期研究的所有患者治疗,预计2025年Q1将出顶线数据。
/ 03 /
医药动态
1)金赛药业自免PD-1获批临床
近日,金赛药业收到NMPA准签发的《药物临床试验批准通知书》,同意公司自主研发的GenSci120注射液开展临床试验,本次获批适应症为成人系统性红斑狼疮(SLE)和成人原发性干燥综合征。GenSci120是一款人源化抗PD-1单抗,具有潜在治疗自身免疫病的特性。
2)奥赛康/信达第三代EGFR-TKI获批治疗非小细胞肺癌
1月16日,NMPA官网公示,奥赛康递交的利厄替尼市申请已获得批准,适用于既往经表皮生长因子受体(EGFR)酪氨酸激酶抑制剂(TKI)治疗时或治疗后出现疾病进展,并且经检测确认存在 EGFR T790M 突变阳性的局部晚期或转移性非小细胞肺癌(NSCLC)成人患者的治疗。
3)英派药业 PARP抑制剂塞纳帕利获批上市
1月16日,NMPA官网公示,英派药业 PARP抑制剂「塞纳帕利」获批上市,适用于晚期上皮性卵巢癌、输卵管癌或原发性腹膜癌成人患者在一线含铂化疗达到完全缓解或部分缓解后的维持治疗。
4)亚虹医药“合成致死”USP1抑制剂获NMPA批准开展临床试验
1月16日,亚虹医药宣布,其自主研发的APL-2302(ASN-3186胶囊)用于治疗晚期实体瘤的新药临床研究(IND)获NMPA批准。
/ 04 /
器械跟踪
1)全国医疗器械监督管理工作会议:加快高端创新医疗器械上市步伐
1月17日,2025年全国医疗器械监督管理工作会议在河北省石家庄市召开。会议对2025年医疗器械监管工作部署五项重点任务:
以推进医疗器械管理法制定为牵引,加快完善法律标准体系;以深化审评审批制度改革为核心,加快高端创新医疗器械上市步伐;以守牢安全底线为目标,加快提升医疗器械产业合规水平;以突出实战实训为抓手,加快建设适应产业发展和安全需要的体系和能力;以提升监管国际化水平为导向,加快对外开放合作步伐
/ 05 /
海外药闻
1)勃林格殷格翰精神分裂症新药3期临床失败
1月16日,勃林格殷格翰公布iclepertin治疗成人精神分裂症认知障碍的3期临床项目CONNEX的主要结果,显示主要终点和关键次要终点均未达到。勃林格殷格翰已决定立即终止长期拓展试验CONNEX-X。
iclepertin(BI 425809)是一种口服甘氨酸转运蛋白1(GlyT1)抑制剂,由于疗效突出,iclepertin此前还曾被美国FDA授予突破性疗法认定,并被中国CDE纳入突破性治疗品种。
遗憾的是,iclepertin仍未能穿越CNS研发死亡谷。这也是近两个月,继艾伯维巨资收购Cerevel得到的“王牌项目”emraclidine遭遇滑铁卢,未能在两项针对精神分裂症的II期研究中达到预期终点之后,药企在精神分裂症领域的又一败绩。
2)FDA拒绝批准Atara现货型T细胞疗法
1月16日,Atara Biotherapeutics 宣布已收到FDA针对Ebvallo生物制品许可申请 (BLA) 的完整回复函 (CRL)。该申请适应症为 Ebvallo作为单一疗法用于2岁及以上患有 Epstein-Barr病毒阳性移植后淋巴细胞增殖性疾病 的成人和儿童患者,这些患者之前至少接受过一种疗法,包括含抗CD20的方案。
根据Atara新闻稿,本次未获得FDA批准仅与第三方生产商的检查结果有关。CRL中没有提及BLA中与临床疗效或安全性数据相关的缺陷,FDA也未要求进行任何新的临床研究来支持Ebvallo的获批。
PS:欢迎扫描下方二维码,添加氨基君微信号交流。
申请上市临床3期
2025-01-12
·精准药物
在癌症治疗的漫长征程中,科学家们一直在不懈探索新的靶点和疗法,其中DNA损伤反应(DDR)抑制剂成为了近年来的研究热点。今天,就让我们深入了解一下DDR抑制剂在癌症治疗领域的发展情况。
DDR抑制剂的发展
早期探索:最初针对MGMT的研究试图阻断其对烷化剂DNA损伤的修复,但因 MGMT抑制剂在正常细胞和肿瘤细胞中均降低MGMT水平,导致毒性增加,临床效果未达预期;
PARP 抑制剂的突破:PARP抑制剂的研发起初旨在增强化疗或放疗效果,但因毒性问题受限。随后发现HRR缺陷细胞对PARP抑制剂单药治疗极为敏感,基于合成致死原理,PARP抑制剂在HRD癌症治疗中取得显著成果,已有六种PAR 抑制剂在全球获批。
PARP 抑制剂的临床经验教训
化疗组合:PARPi与化疗联合研究众多,但均未获批,主要毒性问题为骨髓抑制。研究表明,化疗药物的选择、PARPi的特性、剂量、给药方案和顺序等因素均会影响联合治疗效果。例如,PARPi与DNA甲基化剂等联合具有较高的超加和性,不同PARPi在与化疗药物联合时效果存在差异,且给药剂量和顺序也需根据具体情况优化。
与其他 DDR 抑制剂联合:PARPi与ATR抑制剂、WEE1抑制剂等联合具有理论依据,部分临床前数据支持,但临床研究中常因重叠毒性(如骨髓抑制)限制发展。不过,通过调整给药顺序(如在Ib STAR研究中)可提高耐受性,且PARPi与非细胞毒性靶向疗法(如抗血管生成和免疫检查点抑制剂)联合在临床研究中展现出良好的耐受性和应用前景,已有多种联合方案进入临床试验后期阶段并取得一定成果。
放疗联合:PARPi与放疗联合的机制基于DNA复制 - 细胞周期依赖模型,虽有理论优势且部分临床试验显示出一定疗效,但目前尚未成为标准治疗方案。仍需探索放疗剂量、PARPi与放疗的间隔时间、正常组织毒性的规避与缓解以及联合治疗的顺序等问题。
单药治疗:PARPi作为单药在HRD癌症治疗中取得成功,如在卵巢癌、乳腺癌、前列腺癌和胰腺癌等。但早期研究在剂量确定方面存在不足,后期研究提示可利用生物标志物优化患者选择,且PARPi单药治疗的潜在靶点可能不限于HRD癌症,如核糖核酸酶H2和ARID1A突变相关癌症。
当前DDR抑制剂研究概况
众多学术和企业机构积极探索DDR靶点,蛋白激酶是最常见的药物靶点类别,目前多数药物处于临床前阶段,部分已进入临床研究,但也有不少药物研发中断。
ATR抑制剂:通过破坏复制应激反应导致细胞死亡,在实体瘤治疗中进行了广泛研究,与多种疗法联合应用,部分临床试验显示出良好效果,且对其敏感性与基因背景相关,但临床转化仍面临挑战。
WEE1抑制剂:可调控细胞周期G2/M转换,与化疗、放疗联合有一定疗效,但因毒性问题(如骨髓抑制)阻碍了临床发展,相关生物标志物可用于指导剂量调整。
DNA-PK抑制剂:是NHEJ的核心成分,单独抑制对癌细胞活力影响有限,但与放疗或化疗联合可增强细胞毒性,部分抑制剂已进入临床试验阶段。
其他酶类抑制剂:如Pol Θ、USP1、WRN等的抑制剂也在研发中,部分已进入早期临床阶段,其作用机制和潜在治疗价值各有特点。
回顾DDR抑制剂的研发历程,我们可以得出以下重要启示:在联合治疗的探索中,我们需要更加灵活地评估剂量和给药方案,不妨考虑低剂量化疗与DDR抑制联合的创新策略。同时,要高度重视靶点结合情况的证明,并在临床前充分研究药物的作用机制和相关动力学。预测性生物标志物对于患者的精准选择至关重要,除了关注基因突变外,还应深入开展功能检测,全面了解肿瘤对DDR抑制剂的耐药机制。
声明:发表/转载本文仅仅是出于传播信息的需要,并不意味着代表本公众号观点或证实其内容的真实性。据此内容作出的任何判断,后果自负。若有侵权,告知必删!
长按关注本公众号
粉丝群/投稿/授权/广告等
请联系公众号助手
觉得本文好看,请点这里↓
临床结果免疫疗法临床2期
2024-12-12
·医药观澜
▎药明康德内容团队报道
根据中国国家药监局药品审评中心(CDE)官网以及公开资料,本周(11月18日~11月24日),有9款1类创新药首次在中国获得临床试验默示许可(IND)。通过梳理,这些产品包括了“合成致死”靶点新药USP1抑制剂、PRMT5抑制剂,以及长效代谢酶生物药物、GIP/GLP-1/FGF21三靶点融合蛋白等,拟开发的适应症涵盖不同类型的癌症,以及2型糖尿病、超重或者肥胖等代谢性疾病。本文将根据公开资料介绍其中部分产品的基本信息。
图片来源:123RF
普利药业:注射用PL002
作用机制:荧光/磁共振双模态造影剂
适应症:用于脑胶质瘤患者的术前诊断及术中导航
普利药业申报的1类新药注射用PL002获批临床,拟用于脑胶质瘤患者的术前诊断及术中导航。根据普利药业新闻稿介绍,这是一款荧光/磁共振双模态造影剂。相较于单独使用,术中荧光技术与核磁共振成像的结合能够更准确地识别肿瘤组织与功能区的位置关系,以及患者的肿瘤浸润边缘。这种联合使用能在保留患者功能的前提下实现功能区脑胶质瘤的最大限度安全切除,且能有效降低术中神经损伤的风险。该产品此前已经于2023年在美国获批IND,用于原发性肝癌患者的术前诊断及术中导航。
康缘药业:KYS202003A注射液
作用机制:生物制品新药
适应症:晚期实体瘤
康缘药业1类新药KYS202003A注射液获批临床,拟开发治疗晚期实体瘤患者。目前尚未从公开渠道查询到该药的具体作用机制,从受理号可知为一款生物制品新药。
派金生物:PJ016注射液
作用机制:长效代谢酶生物药物
适应症:晚期实体瘤
派金生物1类新药PJ016注射液获批临床,拟开发治疗晚期实体瘤。根据派金生物新闻稿介绍,PJ016注射液是一种长效聚乙二醇化精氨酸耗竭生物新药,基于天然来源精氨酸脱亚胺酶,经派金生物自主创新持续迭代的MAS-PEG(多位点饱和定向改造PEG)技术平台研发而成的共价二聚体大分子酶。经体内外药理药效的研究证明,PJ016通过耗竭外周血中的精氨酸抑制肿瘤细胞的生长,并诱导癌细胞的凋亡和焦亡,将“冷肿瘤”转变为“热肿瘤”;调动和促进免疫细胞浸润于瘤体内杀伤癌细胞,同时协同免疫调节剂抗体药物的抑瘤作用。
轩竹生物:XZP-6924-TS1片
作用机制:USP1抑制剂
适应症:实体瘤
轩竹生物申报的XZP-6924-TS1片获批临床,拟用于实体瘤的治疗。公开资料显示,XZP-6924是一种有效的高选择性USP1抑制剂,具有提高疗效和克服PARP抑制剂耐药性的潜力。该产品的临床前研究结果还入选了2024年美国癌症研究协会(AACR)年会Late-Breaking Research(突破性研究)。USP1是“合成致死”领域的新兴靶点,因其在癌症治疗中的潜力在近年来倍受新药研发企业青睐。
恒瑞医药:注射用SHR-4394
作用机制:生物制品新药
适应症:前列腺癌
恒瑞医药1类新药注射用SHR-4394获批临床,拟开发治疗前列腺癌。目前尚未从公开渠道查询到该药的具体作用机制,从受理号可知为一款生物制品新药。
百济神州:BGB-58067片
作用机制:PRMT5抑制剂
适应症:晚期实体瘤
百济神州1类新药BGB-58067片获批临床,拟开发治疗晚期实体瘤患者。根据百济神州公开资料,BGB-58067为该公司未来的核心项目之一。这是一款第二代MTA协同PRMT5抑制剂,可选择性杀伤MTAP缺失肿瘤细胞,同时避免影响正常血液细胞。研究表明,在MTAP缺失细胞中极具活性和选择性,具有良好的血脑屏障穿透力和颅内药效,拥有理想的半衰期,可以实现每日给药。
原研药港生命科学:PE-003片
作用机制:化药新药
适应症:成年男性早泄
原研药港生命科学申报的1类新药PE-003片获批临床,拟开发用于治疗成年男性早泄。目前尚未从公开渠道查询到该药的具体作用机制,从受理号可知为一款化药新药。
昆药集团:KPC000154片
作用机制:化药新药
适应症:非酒精性脂肪性肝炎
昆药集团申报的1类新药KPC000154片获批临床,拟开发用于治疗非酒精性脂肪性肝炎。目前尚未从公开资料查询到该药的具体作用机制。根据昆药集团公告介绍,这是一款化药新药,昆药集团对KPC000154片累计研发投入约5514万元人民币。
民为生物:MWN105注射液
作用机制:GIP/GLP-1/FGF21三靶点融合蛋白
适应症:2型糖尿病、超重或者肥胖
民为生物1类新药MWN105注射液获批临床,拟开发治疗2型糖尿病、超重或者肥胖。根据民为生物官网管线资料可知,MWN105注射液为一款GIP/GLP-1/FGF21三靶点融合蛋白。
参考资料:
[1]中国国家药监局药品审评中心(CDE)官网. Retrieved Nov 24,2024, From https://www.cde.org.cn/main/xxgk/listpage/4b5255eb0a84820cef4ca3e8b6bbe20c
[2]各公司官网及公开资料
本文来自药明康德内容团队,欢迎个人转发至朋友圈,谢绝媒体或机构未经授权以任何形式转载至其他平台。转载授权或其他合作需求,请联系wuxi_media@wuxiapptec.com。
免责声明:药明康德内容团队专注介绍全球生物医药健康研究进展。本文仅作信息交流之目的,文中观点不代表药明康德立场,亦不代表药明康德支持或反对文中观点。本文也不是治疗方案推荐。如需获得治疗方案指导,请前往正规医院就诊。
临床获批
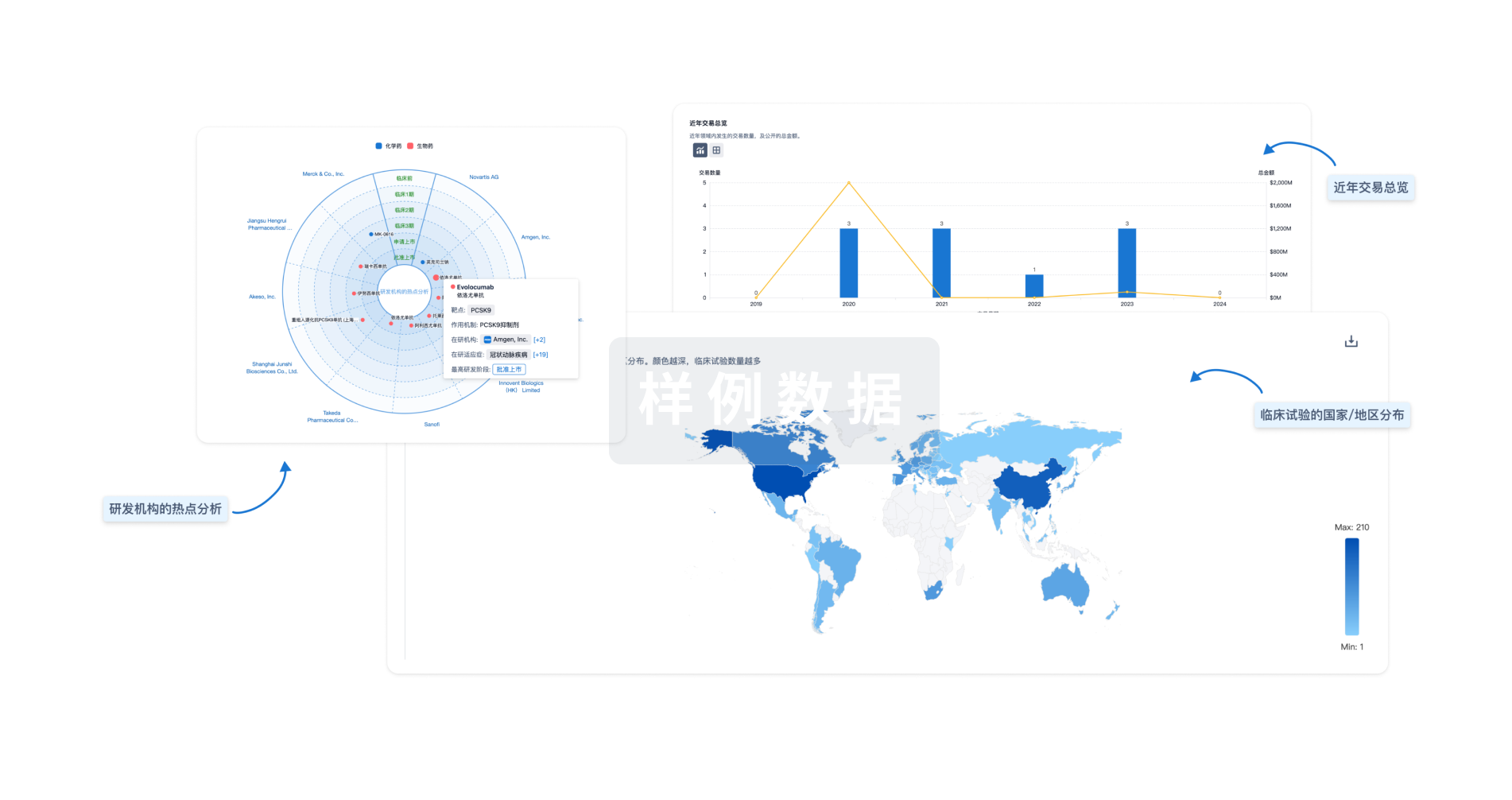
分析
对领域进行一次全面的分析。
登录
或
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
