Clinical Research of ROBO1 Specific BiCAR-NK Cells on Patients With Pancreatic Cancer
Immunotherapy has become the major breakthrough and the most promising treatment, with the host of development of tumor biology, molecular biology and immunology. ROBO1 is a potential target and spectacular paradigm in the treatment of solid tumors. This study is for evaluation of the safety and efficacy of ROBO1 CAR-NK cell immunotherapy for pancreatic cancer.
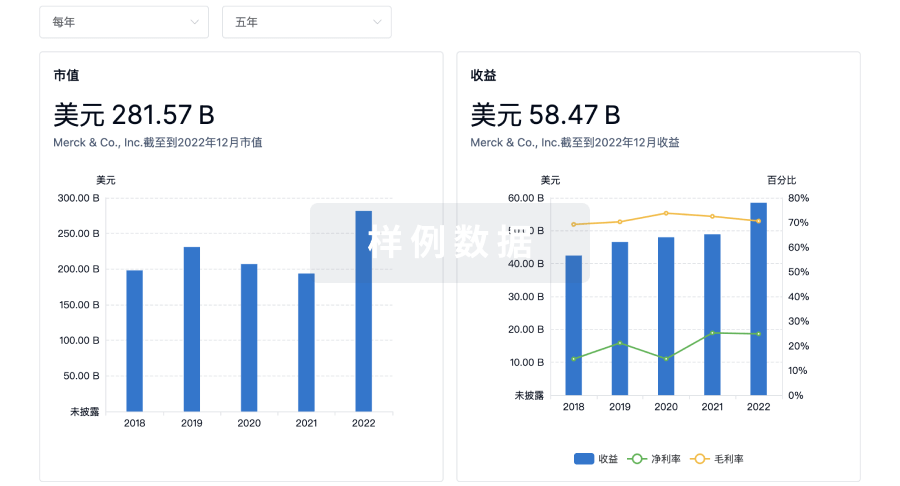
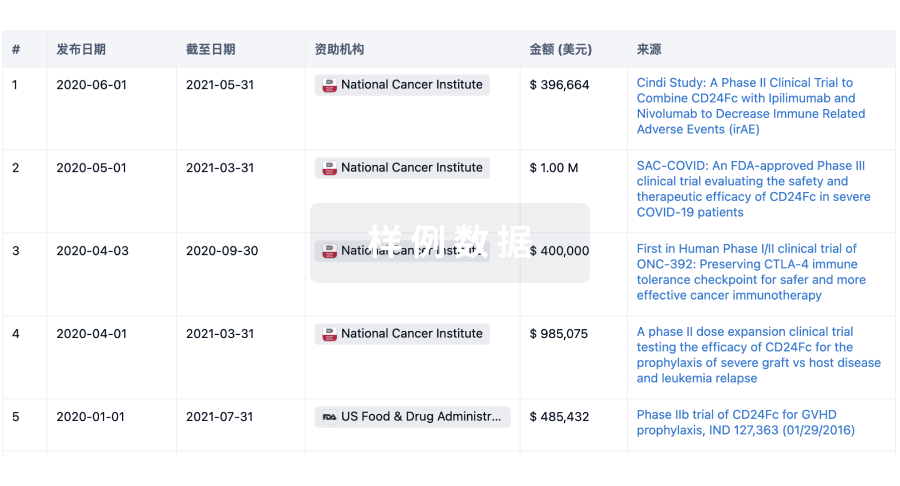
100 项与 Asclepius (Hangzhou) Medical Technology Co., Ltd. 相关的临床结果
0 项与 Asclepius (Hangzhou) Medical Technology Co., Ltd. 相关的专利(医药)
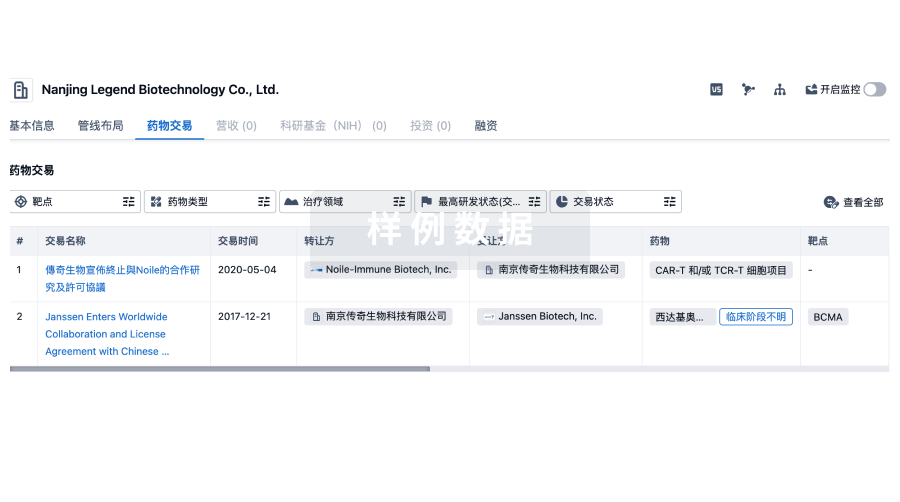
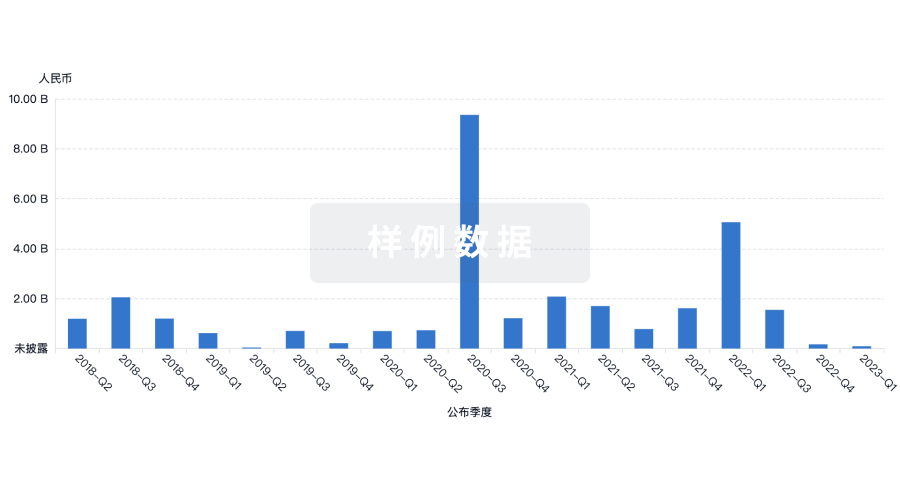
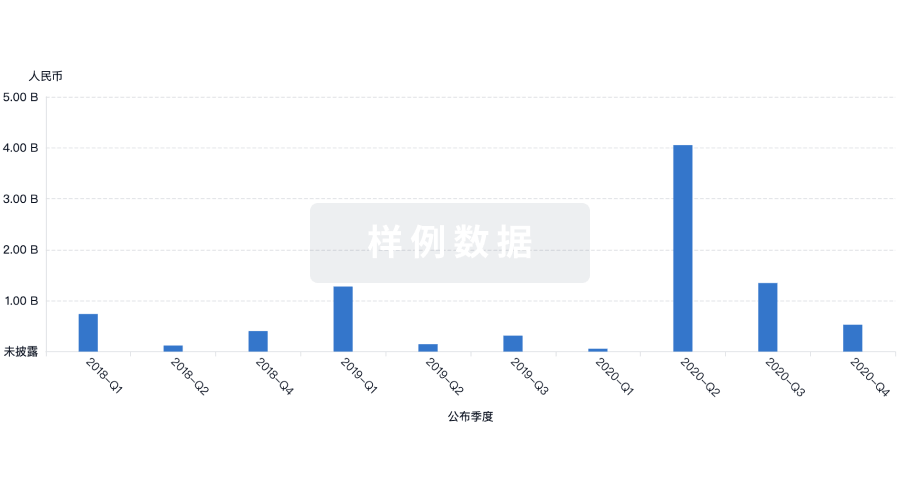
100 项与 Asclepius (Hangzhou) Medical Technology Co., Ltd. 相关的药物交易
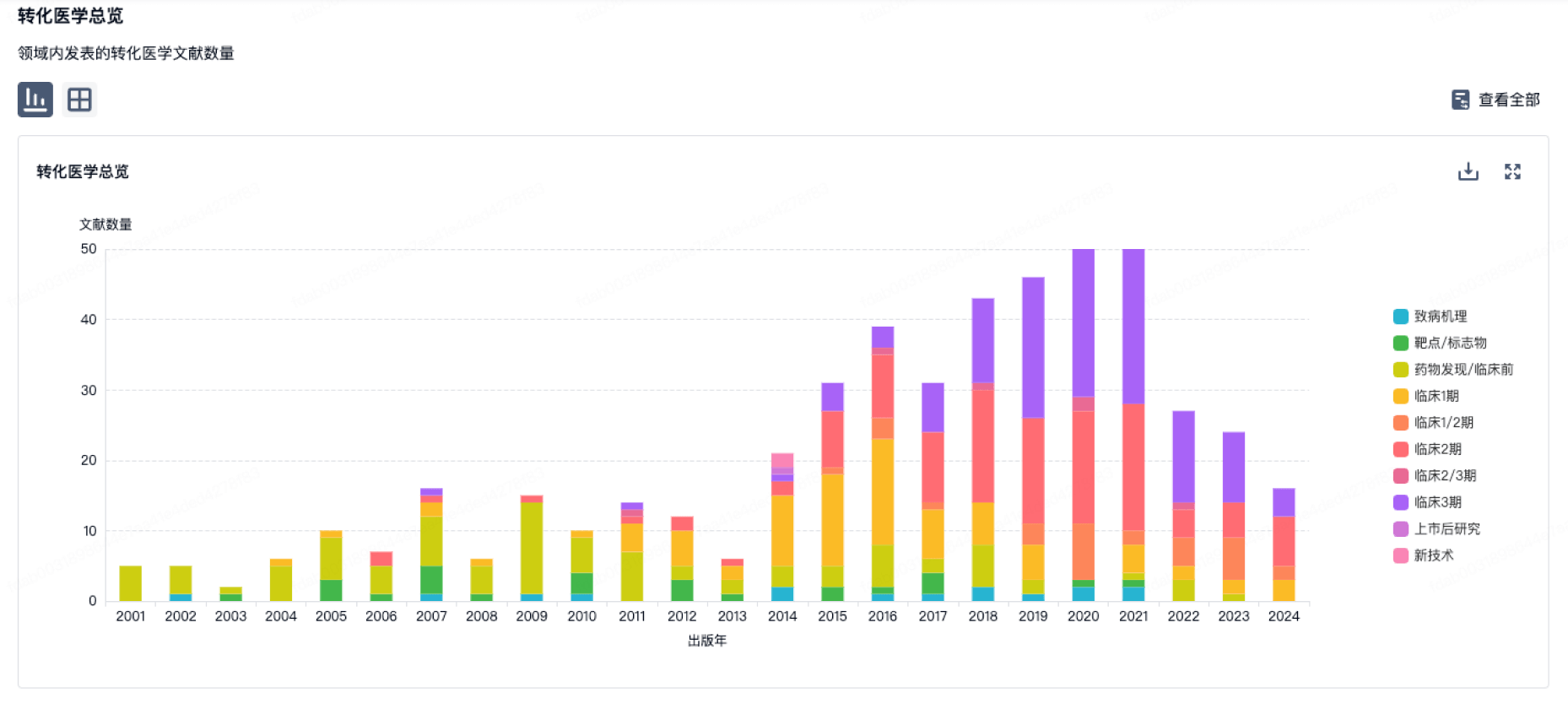
100 项与 Asclepius (Hangzhou) Medical Technology Co., Ltd. 相关的转化医学