更新于:2024-11-01

Physicians Committee for Responsible Medicine, Inc.
更新于:2024-11-01
概览
关联
30
项与 Physicians Committee for Responsible Medicine, Inc. 相关的临床试验Physicians Committee for Responsible Medicine, a Randomized, Crossover Trial of the Effect of a Dietary Intervention on Intracellular Lipid, Insulin Sensitivity, and Glycemic Control in Type 2 Diabetes
The purpose of this study is to compare the effects of a low-fat, plant-based dietary intervention and a portion-controlled dietary intervention (compliant with current American Diabetes Association (ADA) guidelines) on changes in intramyocellular and hepatocellular lipid content in adults with type 2 diabetes. Changes in insulin sensitivity and glycemic control will also be assessed in this study. The study duration is 44 weeks.
开始日期2025-07-01 |
The Effect of a Low-fat Vegan Dietary Intervention on Intracellular Lipid, Insulin Sensitivity, and Glycemic Control in Type 2 Diabetes
The goal of this 16-week clinical trial is to assess the health benefits of a low-fat vegan diet on insulin sensitivity and glycemic control in type 2 diabetes. Participants will receive at no cost, study-related weekly nutrition education classes and one-on-one consultation with a registered dietitian.
开始日期2024-02-13 |
The Feasibility, Implementation and Efficacy of a Diabetes Reversal Online Program and Study (DROP Study) in an Office Setting
This is an interventional study that will evaluate the feasibility, implementation, and efficacy of an online, 12-week, plant-based, nutrition education program in a community setting. Data will also be analyzed to see if participation in the program results in reductions glycated hemoglobin levels, body weight, and cholesterol levels.
开始日期2023-09-21 |
100 项与 Physicians Committee for Responsible Medicine, Inc. 相关的临床结果
登录后查看更多信息
0 项与 Physicians Committee for Responsible Medicine, Inc. 相关的专利(医药)
登录后查看更多信息
218
项与 Physicians Committee for Responsible Medicine, Inc. 相关的文献(医药)2024-08-01·Regulatory Toxicology and Pharmacology
The need for guidance in antidepressant drug development: Revisiting the role of the forced swim test and tail suspension test
Review
作者: Stibbe, Tina ; Trunnell, Emily R ; Baines, Julia ; Smith, Rachel ; Farghali, Stephen ; Jackson, Tara ; Jayne, Kimberley
2024-07-01·Obesity Reviews
Weight management using meal replacements and cardiometabolic risk reduction in individuals with pre‐diabetes and features of metabolic syndrome: A systematic review and meta‐analysis of randomized controlled trials
Review
作者: Salas-Salvadó, Jordi ; Leiter, Lawrence A ; Kahleová, Hana ; Blanco Mejia, Sonia ; Rahelić, Dario ; Khan, Tauseef A ; Lean, Michael E J ; Noronha, Jarvis C ; Sievenpiper, John L ; Nishi, Stephanie K ; Kendall, Cyril W C
2024-06-01·Clinical Diabetes
Effect of a Dietary Intervention on Insulin Requirements and Glycemic Control in Type 1 Diabetes: A 12-Week Randomized Clinical Trial
Article
作者: Barbaro, Robby ; Barnard, Neal D. ; Holtz, Danielle N. ; Holubkov, Richard ; Sklar, Mark ; Kahleova, Hana ; Pineda, Desiree ; Smith, Karen ; Sutton, Macy ; Znayenko-Miller, Tatiana ; Khambatta, Cyrus
1
项与 Physicians Committee for Responsible Medicine, Inc. 相关的新闻(医药)2022-12-15
WASHINGTON--(
BUSINESS WIRE
)--A physicians group is
warning
the Food and Drug Administration that its
proposed rule
to allow meat and dairy products high in saturated fat to be labeled as “healthy,” a highly-coveted food industry marketing term, puts Americans at risk for heart disease, cancer, and other significant health risks. The Physicians Committee is a nonprofit of more than 17,000 doctors.
In a
complaint
submitted today to FDA Commissioner Robert M. Califf, MD, the group says, “The proposed rule should not be making special accommodations for the sale and marketing of harmful foods such as
eggs
, dairy, and meat as 'healthy.' Besides being the predominate sources of saturated fat in American’s diets, the consumption of these foods poses other significant
health risks
.”
In the proposed rule, titled “Food Labeling: Nutrient Content Claims; Definition of Term ‘Healthy,’” the FDA is suggesting special accommodations to the allowable percentage of daily value (DV) per reference amount customarily consumed (RACC) of saturated fat for eggs,
dairy
, and
meat
products at levels higher than other foods.
Acknowledging the need to limit saturated fat in Americans’ diet, the proposed rule recommends a baseline limit of 5% of the DV per RACC for most foods. However, the proposed rule suggests
doubling
the allowable level of saturated fat to allow eggs, game meat, and seafood to be labeled has “healthy” compared to plant-based proteins sources like beans, peas, and soy products.
Additionally, the proposed rule, in a blatant and apparent concession to allow
eggs
to be marketed as “healthy,” completely ignores the
dangers
of cholesterol. Dietary cholesterol, along with saturated fat, causes blood cholesterol levels to steadily increase leading to adverse health.
“Limiting saturated fat and cholesterol should be paramount for the 'healthy' label,” says Anna Herby, DHSc, RD, CDE, nutrition education program manager for the Physicians Committee. “Truly healthy foods, like fruits, vegetables, grains, and beans, are low or in saturated fat and cholesterol and have been shown to help prevent and reverse heart disease, as well as diabetes, high blood pressure, stroke, and cancer.”
Comments
on the
proposed rule
will be accepted through Feb. 16, 2023.
100 项与 Physicians Committee for Responsible Medicine, Inc. 相关的药物交易
登录后查看更多信息
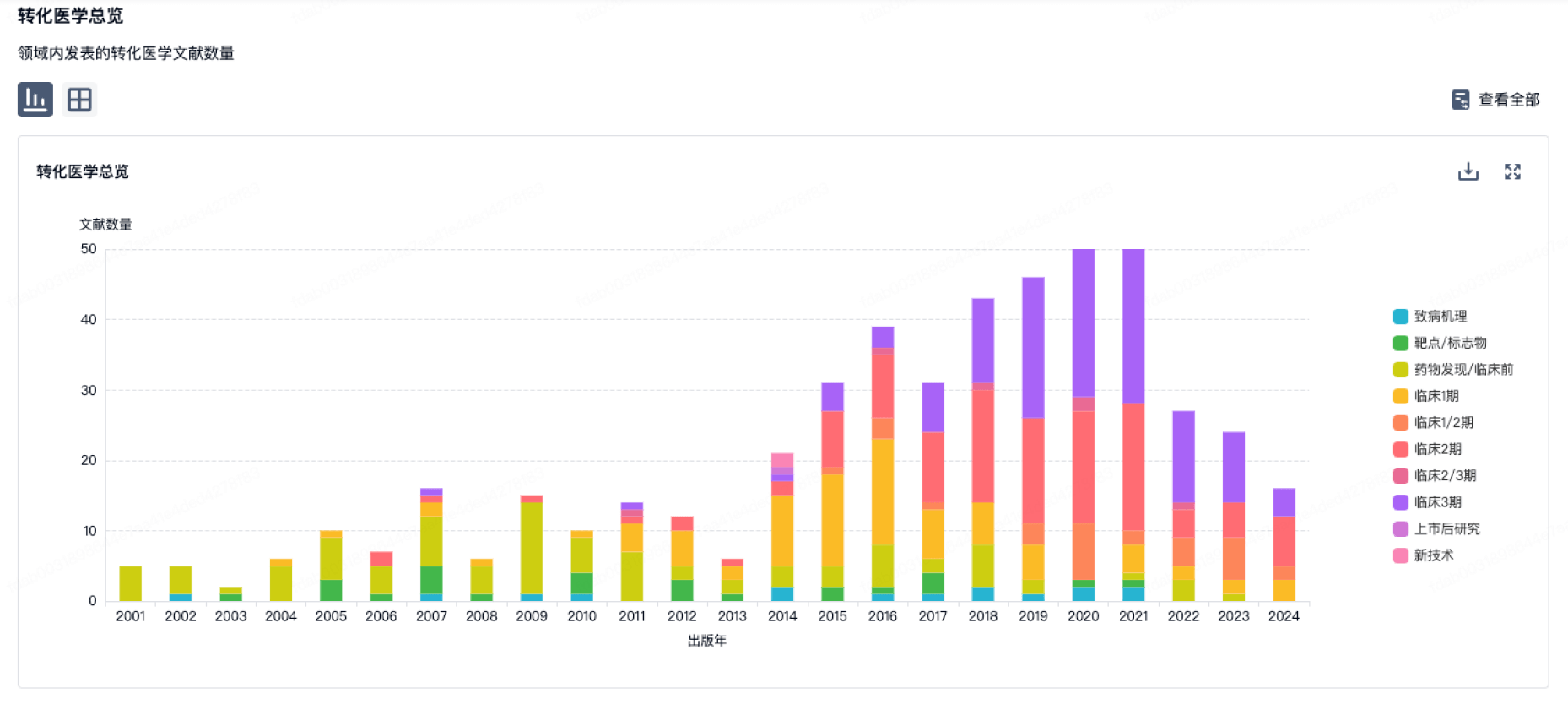
100 项与 Physicians Committee for Responsible Medicine, Inc. 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2024年11月19日管线快照
无数据报导
登录后保持更新
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用