预约演示
更新于:2025-01-23

The Gordon Schanzlin New Vision Institute, Inc.
更新于:2025-01-23
概览
关联
2
项与 The Gordon Schanzlin New Vision Institute, Inc. 相关的临床试验NCT06176651
Evaluation of Miebo (Perfluorohexyloctane) Eyedrops in Habitual Contact Lens Wearers
Miebo (Perfluorohexyloctane) is a novel, non-aqueous, single entity, preservative free, ophthalmic drop. This drop was recently approved by the Food and Drug Administration (FDA) for the treatment of Meibomian Gland Disease (MGD).
All published data on Miebo has been done in non-contact lens wearers. As contact lens dropout rates seem to be an ongoing problem for practitioners, we are performing this study utilizing this novel new drug with contact lens patients to determine if the drops assist comfort in typical soft contact lens wearing patients. If the study determines that Miebo assists in the overall comfort of contact lens patients this could be a possible way to help keep patients in their contact lenses longer.
All published data on Miebo has been done in non-contact lens wearers. As contact lens dropout rates seem to be an ongoing problem for practitioners, we are performing this study utilizing this novel new drug with contact lens patients to determine if the drops assist comfort in typical soft contact lens wearing patients. If the study determines that Miebo assists in the overall comfort of contact lens patients this could be a possible way to help keep patients in their contact lenses longer.
开始日期2023-09-21 |
NCT05624320
Improved Near Vision and Intermediate Without Loss of Binocular Distance Vision Using AGN-190584 in Subjects Utilizing Single -Use Daily, Contact Lenses for Distance Vision Correction in Both Eyes
AGN-190584 has gone through many clinical trials showing its safety and effectiveness in individuals from 40-55 years of age. There has been no published data on the use of this product in individuals corrected with contact lenses for distance vision. There are many other applications AGN-190584 could be used for and further studies will show the effectiveness for each.
开始日期2022-12-01 |
申办/合作机构 |
100 项与 The Gordon Schanzlin New Vision Institute, Inc. 相关的临床结果
登录后查看更多信息
0 项与 The Gordon Schanzlin New Vision Institute, Inc. 相关的专利(医药)
登录后查看更多信息
5
项与 The Gordon Schanzlin New Vision Institute, Inc. 相关的文献(医药)2024-02-01·Ophthalmology and Therapy
Phase IV Multicenter, Prospective, Open-Label Clinical Trial of Cenegermin (rhNGF) for Stage 1 Neurotrophic Keratopathy (DEFENDO)
Article
作者: Schanzlin, David ; Mantelli, Flavio ; Massaro-Giordano, Mina ; Goisis, Giovanni ; Pasedis, Georgea ; Berdy, Gregg ; Hamrah, Pedram ; Holland, Edward
Journal of Cataract & Refractive Surgery
Effectiveness and safety of a thermo-mechanical action device versus thermal pulsation device in the treatment of meibomian gland dysfunction
Article
作者: Berdy, Gregg J. ; Verachtert, Anthony ; Parkhurst, Gregory D. ; Echegoyen, Julio ; Sadri, Ehsan ; Klein, Ifat ; Agmon, Yael G.
Clinical Ophthalmology
A Randomized, Controlled Trial Comparing Tearcare® and Cyclosporine Ophthalmic Emulsion for the Treatment of Dry Eye Disease (SAHARA) [Response To Letter]
Article
作者: Saenz, Bobby ; Perez, Victor L ; Chester, Thomas ; Ayres, Brandon D ; Ehegoyen, Julio ; Loh, Jennifer ; Bloomenstein, Marc R ; Kannarr, Shane R ; Rodriguez, Tomasita C ; Dickerson, Jaime E
100 项与 The Gordon Schanzlin New Vision Institute, Inc. 相关的药物交易
登录后查看更多信息
100 项与 The Gordon Schanzlin New Vision Institute, Inc. 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2025年03月31日管线快照
无数据报导
登录后保持更新
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
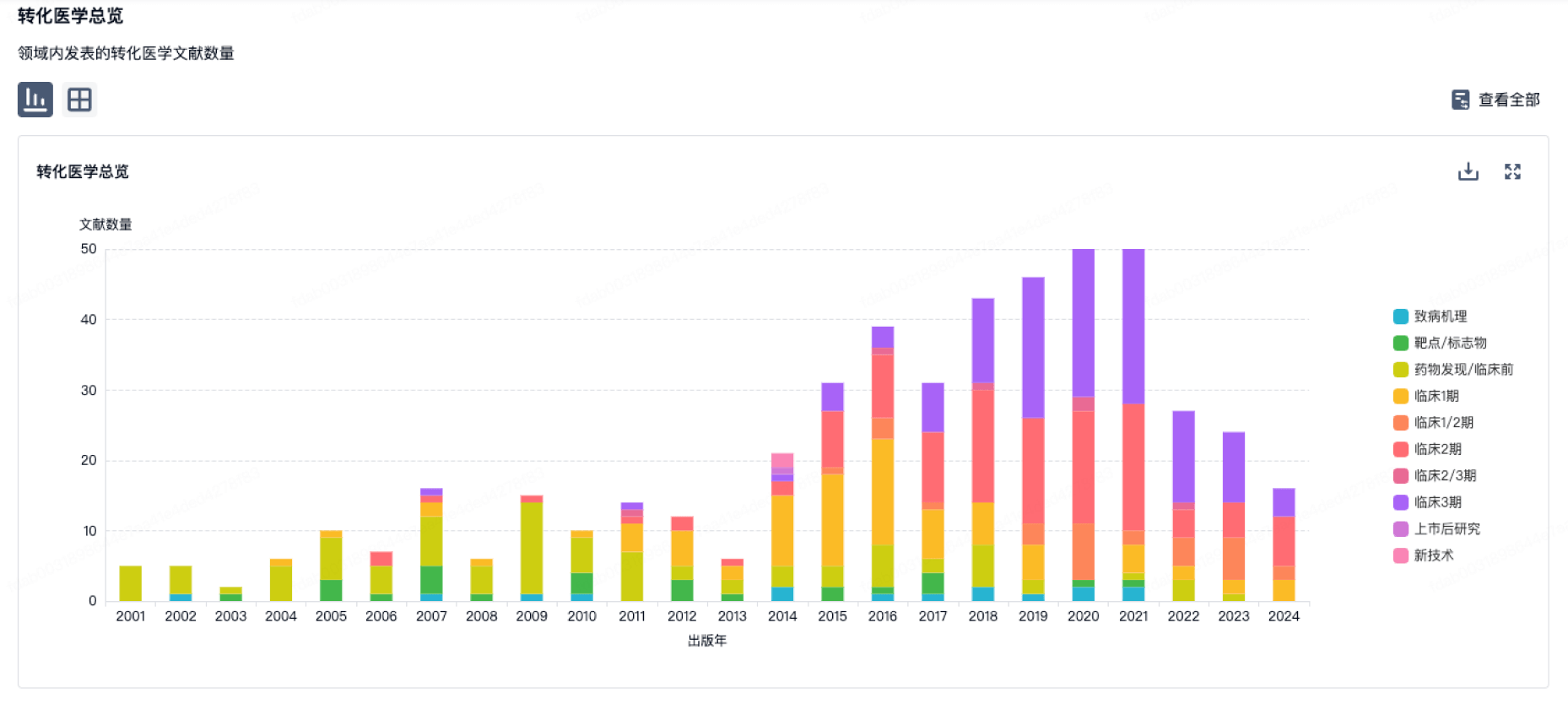
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
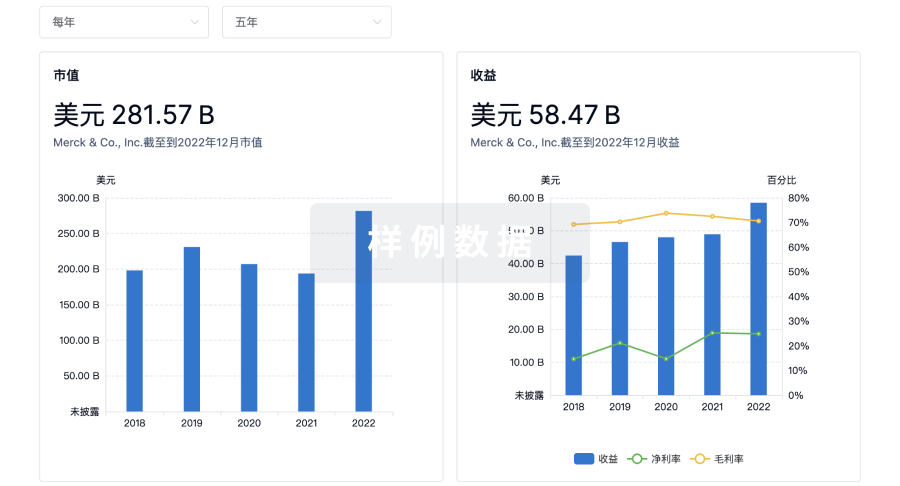
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
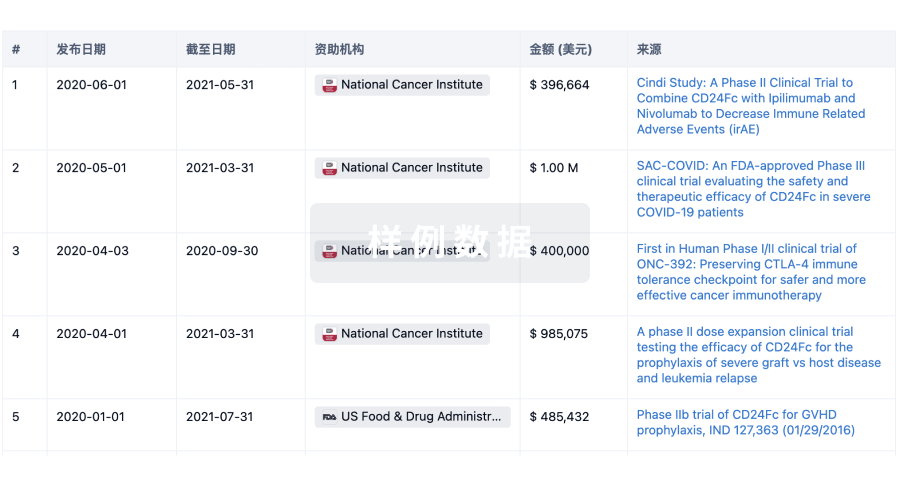
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或
投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或
融资
发掘融资趋势以验证和推进您的投资机会。
登录
或
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用