A Prospective, Randomized, Multi-Center Study to Assess the Safety and Performance of MagnetOs Easypack Putty Standalone Compared to Demineralized Bone Matrix or Fibers (DBX or Grafton) Mixed With Local Autograft in Patients Undergoing One to Two-Level Instrumented Posterolateral Lumbar/Thoracolumbar Fusion (PLF)
This is a phase IV post-marketing study for MagnetOs Easypack Putty. MagnetOs Easypack Putty is a synthetic bone graft extender product that is routinely used by surgeons as a treatment for patients with degenerative disc disease or spinal trauma undergoing spinal fusion surgery.
In this study, MagnetOs Easypack Putty will be used according to the latest U.S. Instructions For Use, standalone in the posterolateral spine.
A Prospective, Randomized, Multi-center Study to Assess the Performance of MagnEtOs Flex Matrix Mixed with Local Autograft Compared to Trinity Elite Mixed with Local Autograft in Patients Undergoing Up to Four-level Instrumented PoSterolatEral Lumbar/thoraco-lumbar Fusion (PLF)
This is a phase IV post-marketing study for MagnetOs Flex Matrix. MagnetOs Flex Matrix is a synthetic bone graft extender product that is routinely used by surgeons as a treatment for patients with degenerative disc disease and undergoing spinal fusion surgery.
In this study, MagnetOs Flex Matrix will be used according to the latest U.S. Instructions For Use, specifically as a bone graft extender mixed with autograft in a 1:1 vol.% in the posterolateral spine.
Trinity Elite will also be used according to its latest IFU approved in the US. Specifically, this cellular based allograft is an allograft intended for the treatment of musculoskeletal defects.
A Prospective, Randomized, Single-center Study to Assess the Performance of MagnetOs Putty Compared to Local Autograft in Patients Undergoing up to Three-level Instrumented Posterolateral Lumbar Fusion (PLF).
This is a phase IV post-marketing study for MagnetOs Putty. MagnetOs Putty is a synthetic bone graft extender product that is routinely used by surgeons as a treatment for patients with leg pain and/or back pain and undergoing spinal fusion surgery.
In this study, MagnetOs Putty will be used according to the latest U.S. Instructions For Use, specifically as a bone graft extender mixed with autograft in a 1:1 vol.% in the posterolateral spine.
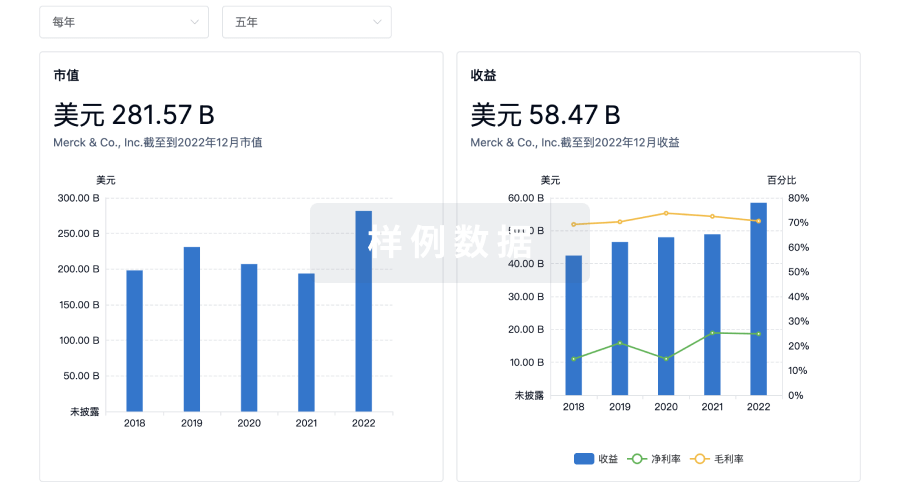
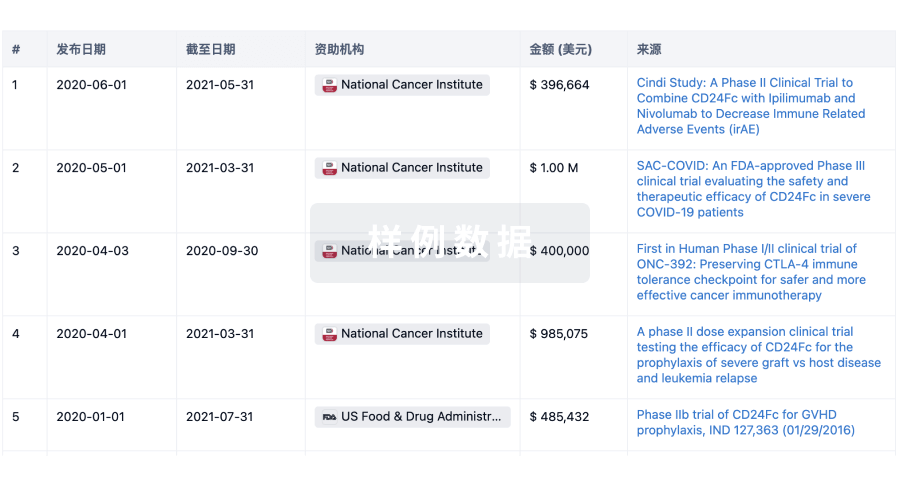
100 项与 Kuros Biosurgery AG 相关的临床结果
0 项与 Kuros Biosurgery AG 相关的专利(医药)
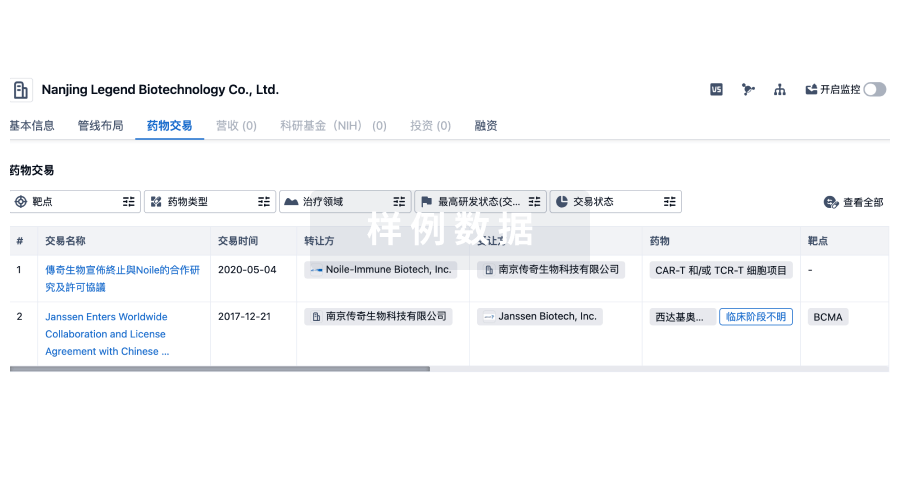
100 项与 Kuros Biosurgery AG 相关的药物交易
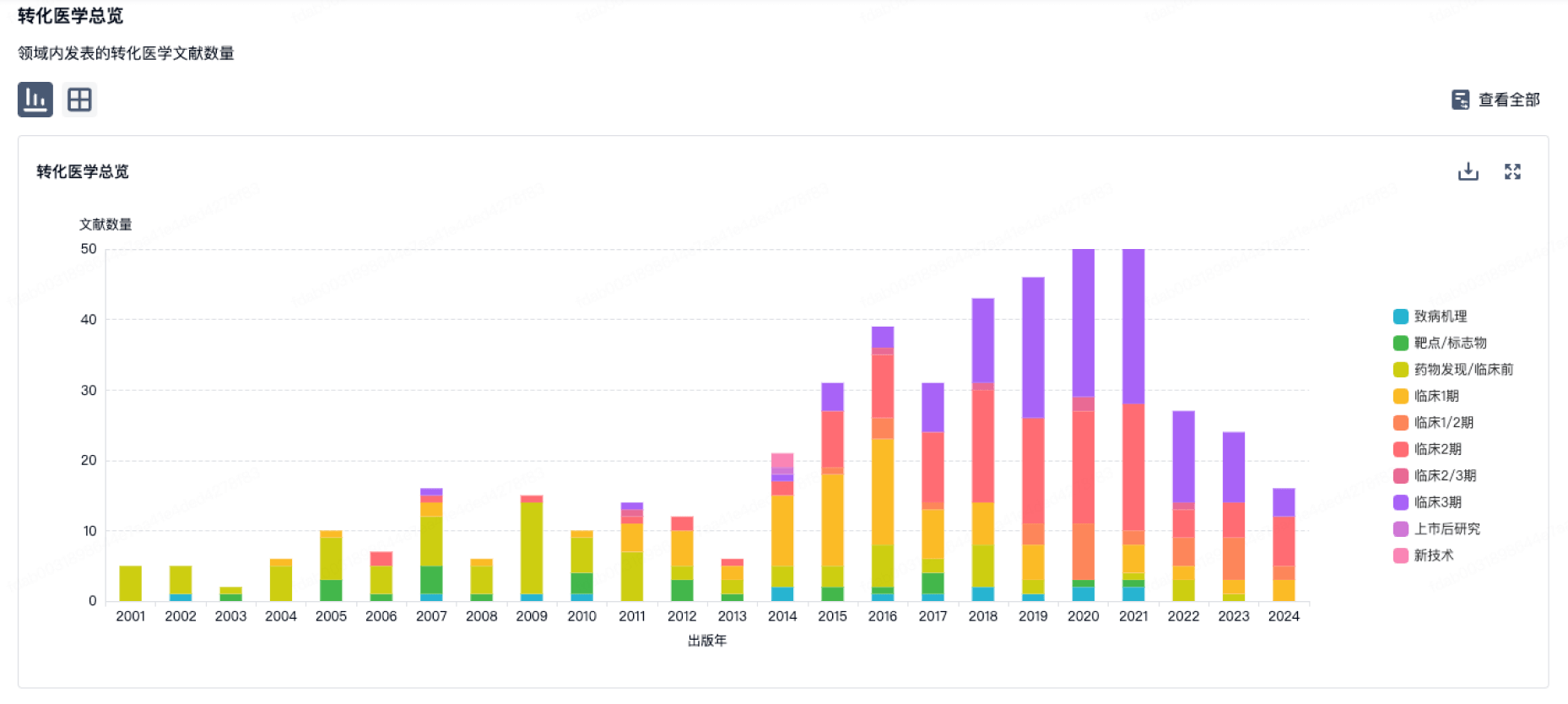
100 项与 Kuros Biosurgery AG 相关的转化医学