Mobius Therapeutics™ To Host Livestream Discussion on July 11th
2022-07-07
Arsham Sheybani, MD of Washington University and Lori Pacheco of Orbis to discuss Mitosol®
12:00 noon EDT - https://www.facebook.com/ophthalmicprofessional
ST. LOUIS, July 7, 2022 /PRNewswire/ -- Mobius Therapeutics™, LLC
, a St. Louis-based perioperative ophthalmic pharmaceutical company, will host a live stream discussion between Arsham Sheybani, MD of Washington University and Lori Pacheco, RN, CRNO of Orbis International. The stream may be accessed July 11 at 12:00 noon EDT at https://www.facebook.com/ophthalmicprofessional
Preview
来源: PRNewswire
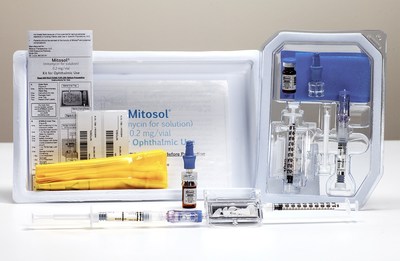
Mitosol (mitomycin for solution) 0.2 mg/vial, Kit for Ophthalmic Use

Preview
来源: PRNewswire
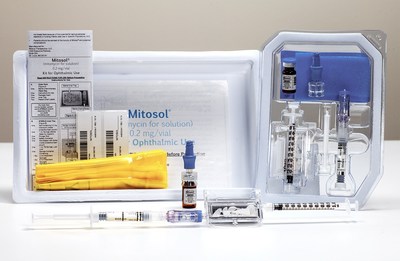
Mitosol (mitomycin for solution) Kit for Ophthalmic Use (PRNewsfoto/Mobius Therapeutics, LLC)
Preview
来源: PRNewswire

Preview
来源: PRNewswire
Mobius Therapeutics™ To Host Livestream on July 11th Arsham Sheybani MD and Lori Pacheco RN to discuss Mitosol®
Tweet this
"Mitosol® customers approach intraoperative use of antimetabolites from very different, yet closely allied positions," said Ed Timm, CEO of Mobius Therapeutics. "While their needs are distinct, resolution is found with the use of Mitosol®."
Mitosol® is the only FDA approved formulation of mitomycin-c bearing an ophthalmic indication. With room temperature storage and shelf life of up to 24 months, Mitosol® offers unique flexibility to providers, as its "shelf-ready" status permits on demand utility.
"Dr. Sheybani and Lori are renowned experts in their fields, offering impactful, real-world voices," continued Timm. "Having earned their advocacy and endorsement is a privilege. They tell us that Mitosol®, delivers value at every level: to the patient, the provider, and the American healthcare system."
About Mobius Therapeutics, LLC:
Mobius Therapeutics is a commercial stage venture focused on sterile perioperative ophthalmic pharmaceuticals. Mitosol® (mitomycin for solution) 0.2 mg/vial, Kit for Ophthalmic Use, is the only formulation of mitomycin-c bearing an ophthalmic indication. Mobius maintains additional perioperative formulations in commercialization, as well as an active product pipeline. Please see full prescribing information at https://mitosol.com/mitosol-package-insert/.
CONTACT:
Ed Timm
314-615-6932
[email protected]
SOURCE Mobius Therapeutics, LLC
更多内容,请访问原始网站
文中所述内容并不反映新药情报库及其所属公司任何意见及观点,如有版权侵扰或错误之处,请及时联系我们,我们会在24小时内配合处理。
机构
适应症
-靶点
-药物
热门报告
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。




