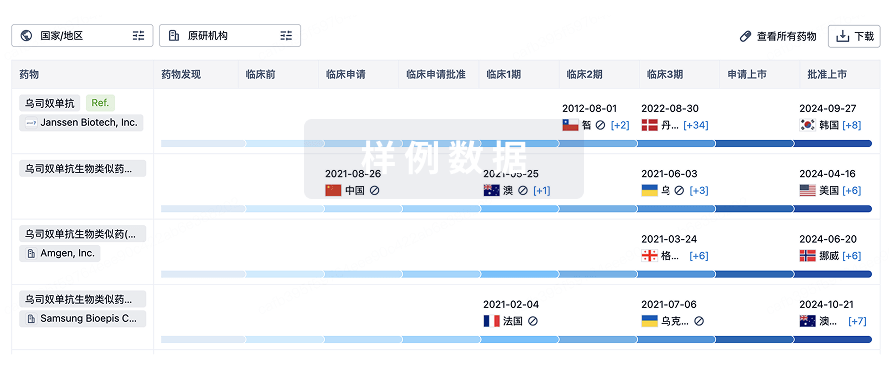
1
项与 Recombinant human alkaline phosphatase(Alloksys Life Sciences BV) 相关的临床试验A Single Dose Study to Assess the Peak Plasma Concentration of a Microdose of Recombinant Human Placental Alkaline Phosphatase (hRESCAP, Part 1) Followed by a Single Ascending Dose, FIH Study to Assess Safety and Tolerability of hRESCAP (Part 2).
In the present study human recombinant placental alkaline phosphatase (hRESCAP) will be investigated. Alkaline Phosphatase is naturally present in the body and reported to use lipopolysaccharde (LPS, bacterial endotoxins) and extracellular nucleotides leaking from damaged and ischemic cells as physiological substrates. The LPS-substrate prevalence makes alkaline phosphatase an interesting novel therapeutic agent in the treatment of LPS-mediated diseases. A bovine homologue of this protein (bovine intestinal alkaline phosphatase, BIAP) has previously been investigated for treatment of acute inflammatory responses such as sepsis, and was shown to be safe in humans. hRESCAP, which will be investigated in the current study, is expected to have a longer half-life in humans than the previously investigated BIAP, due to the fact that it is more sialylated. The possibility to increase the t1/2 to days instead of minutes enables treatment of chronic diseases.
100 项与 Recombinant human alkaline phosphatase(Alloksys Life Sciences BV) 相关的临床结果
100 项与 Recombinant human alkaline phosphatase(Alloksys Life Sciences BV) 相关的转化医学
100 项与 Recombinant human alkaline phosphatase(Alloksys Life Sciences BV) 相关的专利(医药)
100 项与 Recombinant human alkaline phosphatase(Alloksys Life Sciences BV) 相关的药物交易