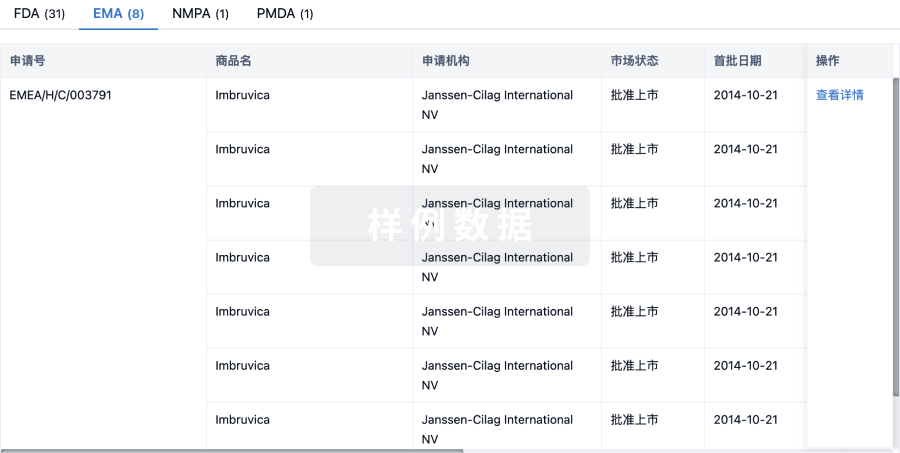
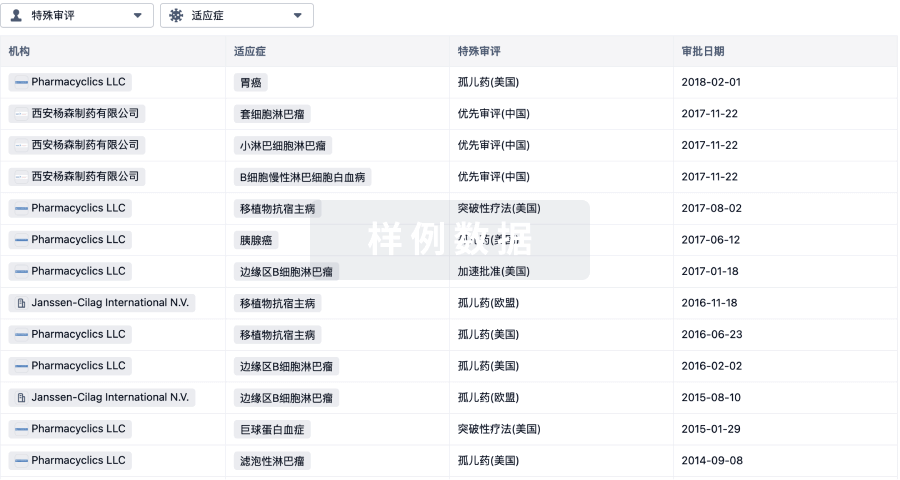
1
项与 Ocular Surface Immune Globulin (OSIG)(University of Illinois at Chicago) 相关的临床试验A Phase II Randomized Placebo-Controlled, Double Blind, Single-Center, Tolerability and Efficacy Clinical Trial of Ocular Surface Immunoglobulin (OSIG) Eye Drops In Patients With Dry Eye Disease
The main objective of this study is to assess the clinical and mechanistic effect of using Ocular surface immunoglobulin (OSIG) eye drops for treating Dry Eye Disease. Therefore, the investigator will perform a prospective, phase II, randomized, placebo-controlled, double-masked, tolerability and efficacy clinical trial using OSIG-eye drops in patients with Dry Eye Disease. This clinical trial will be powered to detect efficacy of the treatment.
This will be a Randomized controlled trial, in which a total of 40 subjects will be enrolled at one clinical site. Subjects will be randomly assigned to one of two groups (#1, #2), with 20 subjects per group. One group will be given placebo (Normal saline eye drops) and the other group will be given eye drops containing the study drug (OSIG). Treatment will be for eight weeks.
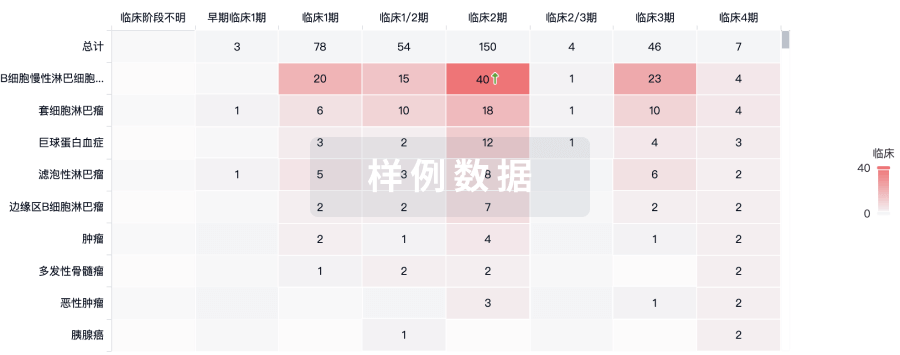
100 项与 Ocular Surface Immune Globulin (OSIG)(University of Illinois at Chicago) 相关的临床结果
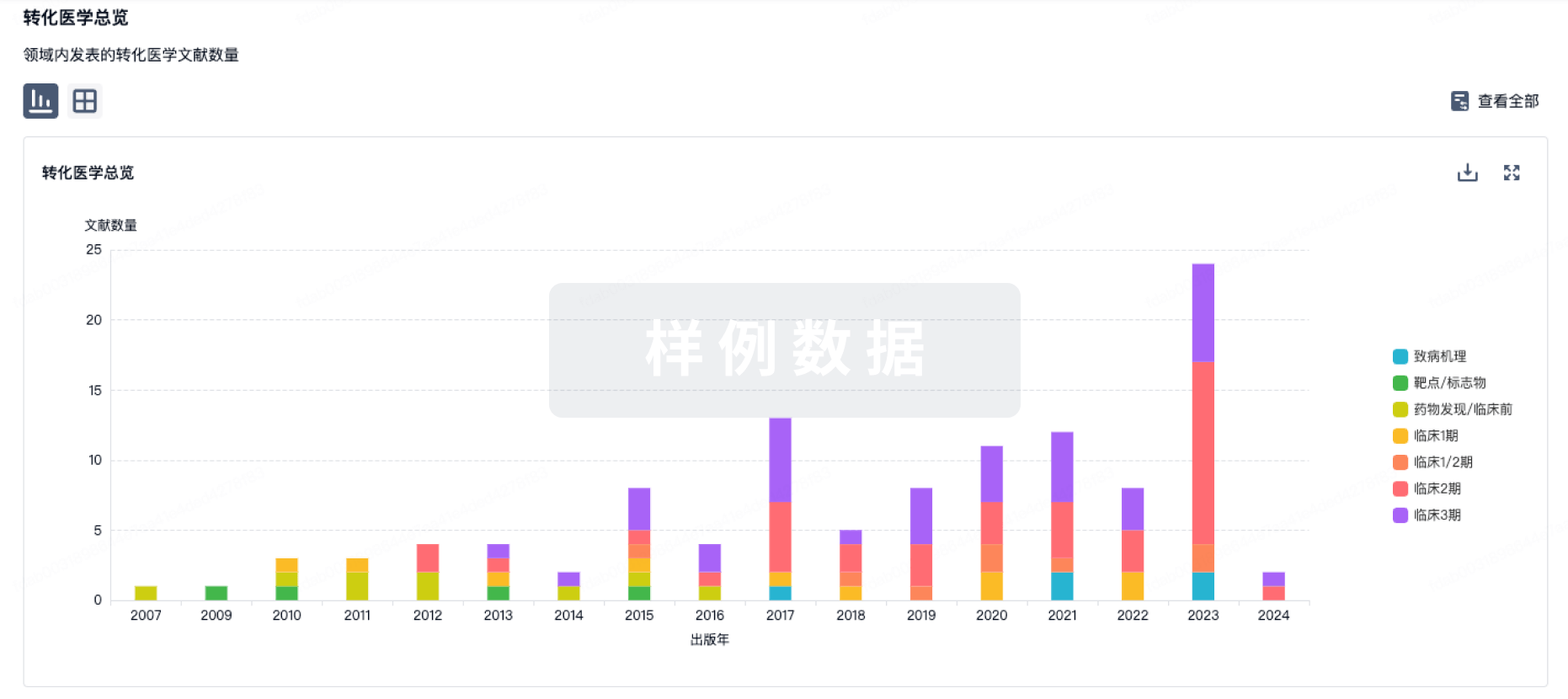
100 项与 Ocular Surface Immune Globulin (OSIG)(University of Illinois at Chicago) 相关的转化医学
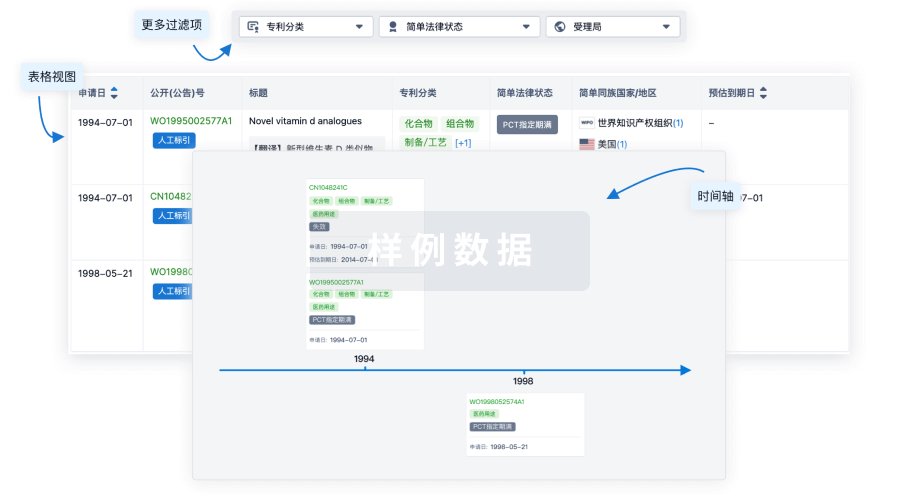
100 项与 Ocular Surface Immune Globulin (OSIG)(University of Illinois at Chicago) 相关的专利(医药)
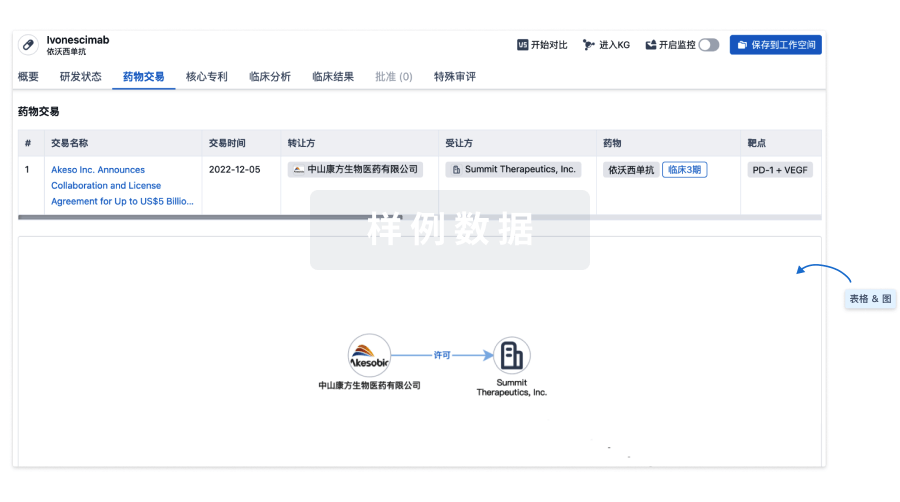
100 项与 Ocular Surface Immune Globulin (OSIG)(University of Illinois at Chicago) 相关的药物交易