预约演示
更新于:2025-05-31
Allogeneic NK cell therapy(Hangzhou Qihan Biotech)
更新于:2025-05-31
概要
基本信息
药物类型 恒定NKT疗法 |
别名 QN-030A |
靶点- |
作用方式- |
作用机制 免疫细胞毒性、自然杀伤细胞替代物 |
在研适应症 |
非在研适应症 |
原研机构 |
在研机构 |
非在研机构- |
权益机构- |
最高研发阶段临床1期 |
首次获批日期- |
最高研发阶段(中国)临床1期 |
特殊审评- |
登录后查看时间轴
关联
2
项与 Allogeneic NK cell therapy(Hangzhou Qihan Biotech) 相关的临床试验NCT05665114
Clinical Study on the Safety and Efficacy of QN-030a in Acute Myeloid Leukemia
This is an open-label, Phase I study of QN-030a (allogeneic NK cell therapy) in relapse/refractory Acute Myeloid Leukemia (AML).
This clinical study is to evaluate the safety, tolerability and preliminary efficacy of QN-030a in patients with r/r AML, where a "3+3" enrollment schema will be utilized at dose escalation stage. Up to 18 patients will be enrolled.
This clinical study is to evaluate the safety, tolerability and preliminary efficacy of QN-030a in patients with r/r AML, where a "3+3" enrollment schema will be utilized at dose escalation stage. Up to 18 patients will be enrolled.
开始日期2022-12-24 |
申办/合作机构  浙江大学 浙江大学 [+1] |
NCT05601830
Clinical Study on the Safety and Efficacy of QN-030a in Acute Myeloid Leukemia With Minimal Residual Disease
This is an open-label, Phase I study of QN-030a (allogeneic NK cell therapy) in Acute Myeloid Leukemia Minimal Residual Disease(AML MRD).
This clinical study is to evaluate the safety, tolerability and preliminary efficacy of QN-020a in patients with AML MRD, where a "3+3" enrollment schema will be utilized at dose escalation stage. Up to 18 patients will be enrolled.
This clinical study is to evaluate the safety, tolerability and preliminary efficacy of QN-020a in patients with AML MRD, where a "3+3" enrollment schema will be utilized at dose escalation stage. Up to 18 patients will be enrolled.
开始日期2022-10-09 |
申办/合作机构- |
100 项与 Allogeneic NK cell therapy(Hangzhou Qihan Biotech) 相关的临床结果
登录后查看更多信息
100 项与 Allogeneic NK cell therapy(Hangzhou Qihan Biotech) 相关的转化医学
登录后查看更多信息
100 项与 Allogeneic NK cell therapy(Hangzhou Qihan Biotech) 相关的专利(医药)
登录后查看更多信息
1
项与 Allogeneic NK cell therapy(Hangzhou Qihan Biotech) 相关的新闻(医药)2024-11-11
NK cell therapy is gaining momentum as a promising solution to unmet medical needs in cancer treatment, offering safer, targeted options for patients unresponsive to traditional therapies. Increased R&D investment, regulatory support, and successful clinical trials have bolstered confidence in these therapies, while technological advances in cell production are set to meet growing demand. Rising cancer rates and heightened public awareness further amplify market potential, making NK cell therapy a pivotal area of growth in oncology.
LAS VEGAS, Nov. 11, 2024 /PRNewswire/ -- DelveInsight's
'
NK Cell Therapy Pipeline Insight 2024
' report provides comprehensive global coverage of pipeline NK cell therapies in various stages of clinical development, major pharmaceutical companies are working to advance the pipeline space and future growth potential of the NK cell therapy pipeline domain.
Key Takeaways from the NK Cell Therapy Pipeline Report
DelveInsight's NK cell therapy pipeline report depicts a robust space with
140+ active players working to develop
160+ pipeline NK cell therapies.
Key NK cell therapy companies such as
Amgen, Innate Pharma, Nektar Therapeutics, SMT bio Co., Ltd., Alphageneron Pharmaceuticals, XNK Therapeutics, ImmunityBio, Cellid, Cantargia, Affimed Therapeutics, Takeda, Artiva Biotherapeutics, Sanofi, Dragonfly Therapeutics, INmune Bio, NKGen Biotech, Asclepius Technology Company Group, Glycostem Therapeutics (IPD Therapeutic), Wugen, Celularity, VERAXA, GamidaCell, MiNK Therapeutics, Indapta Therapeutics, ImmunityBio, Inc., Allife Medical Science and Technology, Nkarta, Base Therapeutics, GT Biopharma, Athenex, Ambicion, Biohaven Pharmaceuticals, Acepodia, Bright Path Biotherapeutics, Nkarta Therapeutics, Qihan Biotech, Century Therapeutics, Fate Therapeutics, Chimeric Therapeutics, Senti Biosciences, GICELL, Deverra Therapeutics, Medigen Biotechnology Corporation, GlaxoSmithKline, CytoImmune Therapeutics, Nuwacell Biotechnologies Co., Ltd., and others are evaluating new NK cell therapies to improve the treatment landscape.
Promising NK cell therapies in the pipeline such as
Bemarituzumab, Monalizumab, NKTR-225, SMT-NK, Enkastim, CellProtect, ALT 803, BVAC-C, PD-L1.t-haNK, Nidanilimab, M ceNK, HER2 t-haNK, AFM-13, TAK-007, IPH4102, AB-101, KDS-1001, AB-201, DF9001, INKmune, IPH65, SNK01, IPH6101 (SAR443579), BCMA CAR-NK 92 cells, DF1001, Allogeneic Natural Killer Cell Therapy (oNKord), WU-NK-101, AFM24, CYNK-001, FLYSYN, GDA-201, AGENT-797, CYNK-101, SAR445514, IDP-023, DF6002, CD19.taNK, Anti-PSMA CAR NK cells, DF2001, NKX101, NK510, GTB-3550, KUR-501, RK-pulsed autologous antigen-presenting cells (APCs), BHV-1100, ACE1702, iPS NKT, Anti-CD19/CD22 CAR NK cell therapy, NKX019, KUR-502, BVAC-P, QN-019a, QN-030a, QN-023a, CNTY-101, FT576, CHM 0201, SENTI-202, AFM28, DF8001, FT522, SNK02, GIC-102, DVX201, Magicell®-NK, GSK4381562, CYTO NK-102, NCR300, and others are under different phases of NK cell therapy clinical trials.
In October 2024, Immunitybio announced that the first patients had been dosed in an initial trial studying the potential of the company's
CAR-NK cell therapy targeting CD-19 in the treatment of non-Hodgkin's lymphoma (NHL).
In July 2024, The U.S. Food and Drug Administration (FDA) gave Nkarta the green light to launch a clinical trial testing its cell therapy candidate
NKX019 in people with ANCA-associated vasculitis (AAV) and other autoimmune disorders.
In May 2024, KGen Biotech announced that its Safety Review Committee had cleared the Company's cryopreserved autologous, expanded, and enhanced
SNK01 to progress into Phase II clinical development.
In May 2024, Fate Therapeutics announced that a late-breaking abstract featuring preclinical data from its
FT522 program for autoimmune diseases will be featured at the American Society of Gene and Cell Therapy (ASGCT) 27th Annual Meeting.
In April 2024, Sanofi moved its natural killer (NK) cell engager candidate
SAR443579/IPH6101 to a Phase II trial evaluating the drug's use in treating a range of blood cancers.
In April 2024, the FDA granted orphan drug designation to the investigational therapy
IGNK001 (Gengleucel) for patients with acute myeloid leukemia (AML).
In February 2024, Indapta Therapeutics received FDA Fast Track Designation for its natural killer (NK) cell therapy
IDP-023 for patients with multiple myeloma (MM) and non-Hodgkin lymphoma (NHL).
Request a sample and discover the recent advances in the NK cell therapy segment @
NK Cell Therapy Pipeline Report
The NK cell therapy pipeline report provides detailed profiles of pipeline assets, a comparative analysis of clinical and non-clinical stage NK cell therapies, inactive and dormant assets, a comprehensive assessment of driving and restraining factors, and an assessment of opportunities and risks in the NK cell therapy clinical trial landscape.
NK Cell Therapy Overview
Natural Killer (NK) cell therapy is an innovative approach in immunotherapy that harnesses the power of NK cells, a vital component of the innate immune system. Unlike T cells, which require prior sensitization to target cancer cells, NK cells can recognize and kill abnormal cells without prior exposure. This unique ability makes NK cell therapy a promising option for treating various cancers, including hematologic malignancies like leukemia and lymphoma, as well as solid tumors. By isolating and activating NK cells from a patient's blood or using engineered NK cells from healthy donors, researchers aim to enhance the immune response against tumors, leading to improved patient outcomes. Recent advancements in genetic engineering, such as the introduction of chimeric antigen receptors (CAR) into NK cells, are further enhancing their specificity and efficacy against cancer cells.
The clinical applications of NK cell therapy are expanding rapidly, supported by a growing body of research and early-phase clinical trials. These studies have shown promising results, demonstrating the ability of NK cell therapy to induce durable remissions in patients with refractory cancers. Furthermore, the off-the-shelf nature of certain NK cell therapies allows for quicker treatment deployment compared to traditional CAR-T cell therapies, which require a personalized approach. Challenges remain, including optimizing NK cell expansion, persistence, and overcoming the immunosuppressive tumor microenvironment. However, ongoing research into combination therapies, such as pairing NK cell therapy with checkpoint inhibitors or monoclonal antibodies, is expected to enhance the therapeutic potential of NK cells and provide new hope for cancer patients.
Find out more about NK cell therapy @
Novel
NK Cell Therapies
A snapshot of the Pipeline NK Cell Therapies mentioned in the report:
Learn more about the emerging NK cell therapies @
NK Cell Therapy Clinical Trials
NK Cell Therapy Therapeutics Assessment
The NK cell therapy pipeline report proffers an integral view of the emerging NK cell therapies segmented by stage, product type, molecule type, and route of administration.
Scope of the NK Cell Therapy Pipeline Report
Coverage: Global
Therapeutic Assessment By Product Type: Mono, Combination, Mono/Combination
Therapeutic Assessment By Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
Therapeutics Assessment
By Route of Administration: Oral, Intravenous, Subcutaneous, Parenteral, Topical
Therapeutics Assessment
By Molecule Type: Vaccines, Monoclonal antibody, Peptides, Polymer, Small molecule
Key NK Cell Therapy Companies: Amgen, Innate Pharma, Nektar Therapeutics, SMT bio Co., Ltd., Alphageneron Pharmaceuticals, XNK Therapeutics, ImmunityBio, Cellid, Cantargia, Affimed Therapeutics, Takeda, Artiva Biotherapeutics, Sanofi, Dragonfly Therapeutics, INmune Bio, NKGen Biotech, Asclepius Technology Company Group, Glycostem Therapeutics (IPD Therapeutic), Wugen, Celularity, VERAXA, GamidaCell, MiNK Therapeutics, Indapta Therapeutics, ImmunityBio, Inc., Allife Medical Science and Technology, Nkarta, Base Therapeutics, GT Biopharma, Athenex, Ambicion, Biohaven Pharmaceuticals, Acepodia, Bright Path Biotherapeutics, Nkarta Therapeutics, Qihan Biotech, Century Therapeutics, Fate Therapeutics, Chimeric Therapeutics, Senti Biosciences, GICELL, Deverra Therapeutics, Medigen Biotechnology Corporation, GlaxoSmithKline, CytoImmune Therapeutics, Nuwacell Biotechnologies Co., Ltd., and others.
Key Pipeline NK Cell Therapies: Bemarituzumab, Monalizumab, NKTR-225, SMT-NK, Enkastim, CellProtect, ALT 803, BVAC-C, PD-L1.t-haNK, Nidanilimab, M ceNK, HER2 t-haNK, AFM-13, TAK-007, IPH4102, AB-101, KDS-1001, AB-201, DF9001, INKmune, IPH65, SNK01, IPH6101 (SAR443579), BCMA CAR-NK 92 cells, DF1001, Allogeneic Natural Killer Cell Therapy (oNKord), WU-NK-101, AFM24, CYNK-001, FLYSYN, GDA-201, AGENT-797, CYNK-101, SAR445514, IDP-023, DF6002, CD19.taNK, Anti-PSMA CAR NK cells, DF2001, NKX101, NK510, GTB-3550, KUR-501, RK-pulsed autologous antigen-presenting cells (APCs), BHV-1100, ACE1702, iPS NKT, Anti-CD19/CD22 CAR NK cell therapy, NKX019, KUR-502, BVAC-P, QN-019a, QN-030a, QN-023a, CNTY-101, FT576, CHM 0201, SENTI-202, AFM28, DF8001, FT522, SNK02, GIC-102, DVX201, Magicell®-NK, GSK4381562, CYTO NK-102, NCR300, and others.
Dive deep into rich insights for new NK cell therapies, visit @
NK Cell Therapy Drugs
Table of Contents
For further information on the NK cell therapy pipeline therapeutics, reach out @
NK Cell Therapy Treatment Drugs
Related Reports
NK Cell Therapy Market
NK Cell Therapy Market Insights, Epidemiology, and Market Forecast – 2032 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, market share of the individual therapies, and key NK cell therapy companies including
Sanofi, Dragonfly Therapeutics, INmune Bio, NKGen Biotech, Asclepius Technology Company Group, Glycostem Therapeutics (IPD Therapeutic), Wugen, Celularity, VERAXA, GamidaCell, MiNK Therapeutics, Indapta Therapeutics, among others.
Non-small Cell Lung Cancer Market
Non-small Cell Lung Cancer Market Insights, Epidemiology, and Market Forecast
– 2032 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key NSCLC companies, including
EMD Serono, Merck, Cellular Biomedicine Group, Inc., Celgene, CellSight Technologies, Inc., BeyondSpring Pharmaceuticals Inc., J Ints Bio, Forward Pharmaceuticals Co., Ltd., AstraZeneca, Bristol-Myers Squibb, Teligene US, Rain Oncology Inc, ReHeva Biosciences, Inc., Amgen, Novartis, RedCloud Bio, Parexel, Vitrac Therapeutics, LLC, Mythic Therapeutics, Instil Bio, Mirati Therapeutics Inc., Daiichi Sankyo, Inc., AstraZeneca, Precision Biologics, Inc, Promontory Therapeutics Inc., Palobiofarma SL, Regeneron Pharmaceuticals, Revolution Medicines, Inc., Cullinan Oncology, LLC, Iovance Biotherapeutics, Inc., Innate Pharma, among others.
Non-small Cell Lung Cancer Pipeline
Non-small Cell Lung Cancer Pipeline Insight – 2024 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key non-small cell lung cancer companies, including
BridgeBio Pharma, Daiichi Sankyo, EMD Serono, Merck, BridgeBio Pharma, Abbvie, Pfizer, Eli Lilly and Company BioNTech SE, Shenzhen TargetRx, Taiho Pharmaceutical, Chong Kun Dang, Bristol Myers Squibb, Innovent Biologics, Xuanzhu Biopharmaceutical, Bayer, GeneScience Pharmaceuticals, InventisBio, Apollomics, Imugene, Ono Pharmaceutical, Pierre Fabre, Jiangsu Hengrui Medicine Co., Bristol-Myers Squibb, Surface Oncology, Inhibrx, Sinocelltech, Mirati Therapeutics, REVOLUTION Medicines, Yong Shun Technology Development, Iovance Biotherapeutics, Galecto Biotech, among others.
Multiple Myeloma Market
Multiple Myeloma Market Insights, Epidemiology, and Market Forecast
– 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key multiple myeloma companies, including
Johnson & Johnson (Janssen), Pfizer, AbbVie and Roche (Genentech), Regeneron Pharmaceuticals, Bristol-Myers Squibb, Celgene, Roche (Genentech), Arcellx, Novartis, Regeneron Pharmaceuticals, BeiGene, CARsgen Therapeutics, Cartesian Therapeutics, C4 Therapeutics, Heidelberg Pharma, Bristol-Myers Squibb, RAPA Therapeutics, AbbVie (TeneoOne), Takeda
, among others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve
.
Contact Us
Shruti Thakur
[email protected]
+14699457679
Logo:
SOURCE DelveInsight Business Research, LLP
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED

细胞疗法孤儿药快速通道免疫疗法临床2期
100 项与 Allogeneic NK cell therapy(Hangzhou Qihan Biotech) 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 难治性急性髓细胞白血病 | 临床1期 | 中国 | 2022-12-24 | |
| 急性髓性白血病 | 临床1期 | 中国 | - | 2022-10-09 |
| 残留肿瘤 | 临床1期 | 中国 | - | 2022-10-09 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
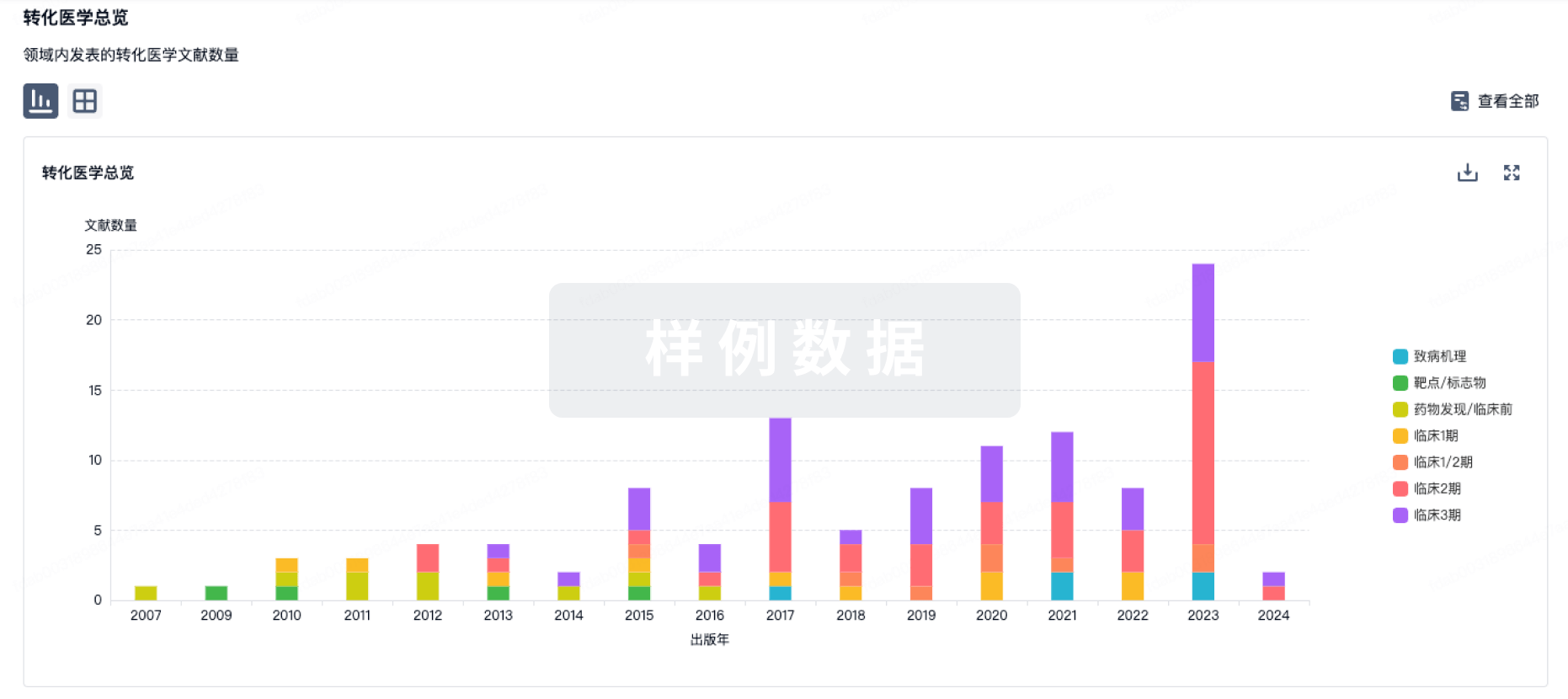
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
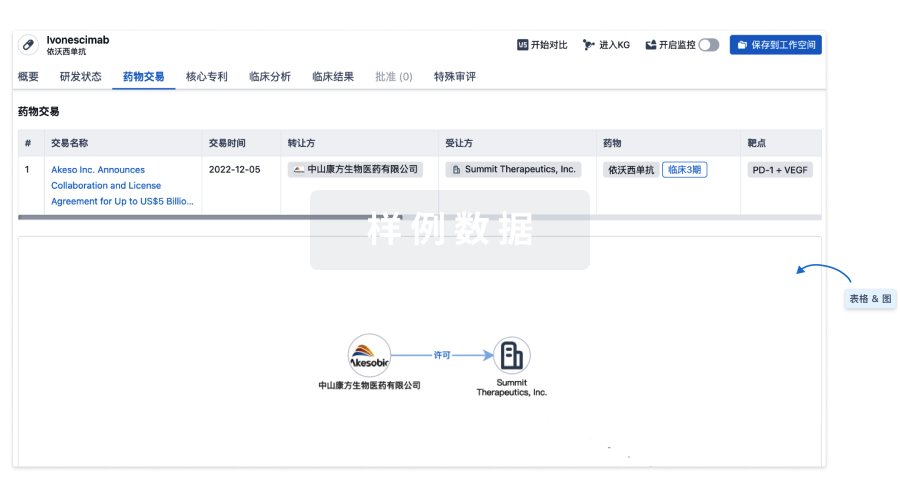
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
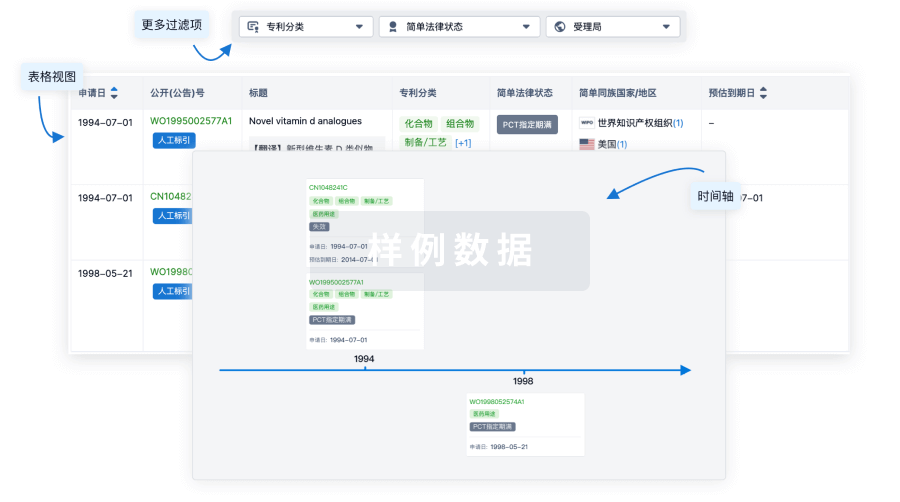
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或

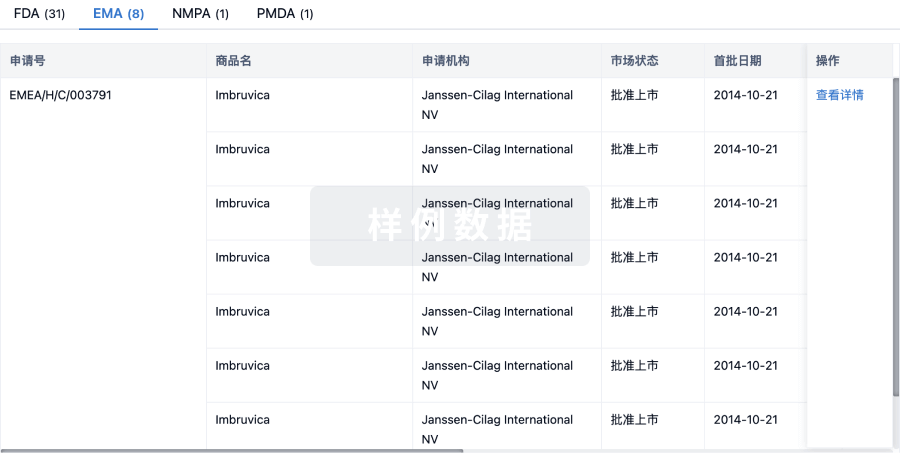
批准
利用最新的监管批准信息加速您的研究。
登录
或
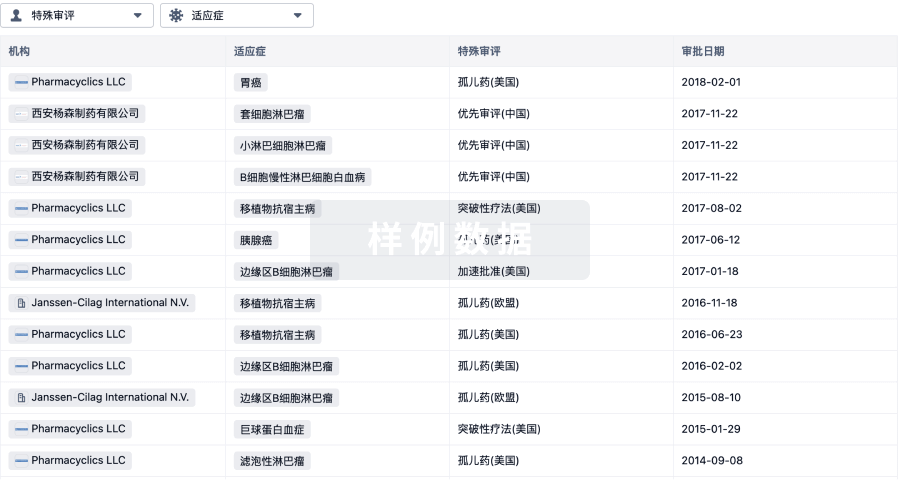
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
