预约演示
更新于:2025-07-05
IL-4R(Amgen, Inc.)
更新于:2025-07-05
概要
基本信息
原研机构 |
在研机构- |
非在研机构 |
权益机构- |
最高研发阶段终止临床2期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
关联
100 项与 IL-4R(Amgen, Inc.) 相关的临床结果
登录后查看更多信息
100 项与 IL-4R(Amgen, Inc.) 相关的转化医学
登录后查看更多信息
100 项与 IL-4R(Amgen, Inc.) 相关的专利(医药)
登录后查看更多信息
34
项与 IL-4R(Amgen, Inc.) 相关的文献(医药)2025-03-01·Dermatology and Therapy
Interleukin-4 and Atopic Dermatitis: Why Does it Matter? A Narrative Review
Review
作者: Mendes-Bastos, Pedro ; Torres, Tiago ; Duarte, Bruno ; Cruz, Maria J ; Filipe, Paulo ; Lopes, Maria J P ; Gonçalo, Margarida
Atopic dermatitis (AD) is a common chronic inflammatory skin condition that significantly impairs patients' quality of life as a result of intense itching and persistent eczematous lesions. Although AD has a multifaceted etiology-including genetic predisposition, environmental triggers, barrier dysfunction, and dysregulated immune responses-interleukin-4 (IL-4) has a recognized central role in its pathogenesis. This narrative review explores the role of IL-4 in the pathophysiology of AD, its contribution to the atopic march, and the therapeutic impact of IL-4 inhibition. IL-4 plays a critical role in skin barrier dysfunction, dysbiosis, pruritus, and inflammation, all of which contribute to the debilitating symptoms of AD. Moreover, IL-4 is implicated in other atopic conditions, such as asthma, allergic rhinitis, and food allergies, underscoring its role beyond AD and its importance in the atopic march. Recent advances in targeted therapies, particularly IL-4/IL-13 signaling inhibitors, have changed AD management. Dupilumab, an IL-4 receptor antagonist, has demonstrated significant efficacy in reducing AD symptoms and enhancing patient outcomes in both children and adults. In addition to symptomatic relief, suppressing IL-4 signaling may also offer potential for disease modification, altering AD's progression and possibly preventing the onset of other atopic conditions. This review highlights the crucial role of IL-4 as a therapeutic target in AD. By understanding the role of IL-4 in AD pathogenesis and exploring the therapeutic implications of targeting IL-4 pathways, this work can contribute to guide future research concerning treatment approaches and also emphasize the need for early and targeted interventions to mitigate disease impact and ultimately improve patient quality of life.
2023-10-01·Arthritis & rheumatology (Hoboken, N.J.)
Characterization of a Musculoskeletal Syndrome of Enthesitis and Arthritis in Patients With Atopic Dermatitis Treated With Dupilumab, an Interleukin-4/13 Inhibitor.
Article
作者: Woolf, Richard T ; Menon, Bina ; Kirkham, Bruce ; Smith, Catherine ; Nathan, Joseph ; Hughes, Catherine D ; Pink, Andrew E ; Mathew, Libin
OBJECTIVE:
To characterize the presentation and outcomes of patients with atopic dermatitis (AD) who developed musculoskeletal symptoms after treatment with dupilumab, a human IgG4 monoclonal antibody that blocks the functions of interleukin-4 (IL-4) and IL-13, key pathologic pathways in AD.
METHODS:
This article reports an observational cohort of patients receiving dupilumab who developed new-onset musculoskeletal symptoms after dupilumab therapy at our center. All patients had a comprehensive rheumatologic history and examination, with imaging by ultrasonography (US) or magnetic resonance imaging (MRI) in most patients.
RESULTS:
Between October 2018 and February 2021, we recorded 470 patients with AD commencing dupilumab treatment from routine clinical care records. Of 36 patients referred for rheumatologic assessment, we identified 26 patients (14 male, 12 female) with a musculoskeletal syndrome of inflammatory enthesitis, arthritis, and/or tenosynovitis. Clinical findings were confirmed by US and MRI. All patients had very good response to dupilumab treatment, and no specific predictors of musculoskeletal syndrome were noted. Symptoms were mild in 16 patients, moderate in 6 patients, and severe in 4 patients. Receipt of nonsteroidal antiinflammatory drugs or cyclooxygenase 2 inhibitors, reduction of dupilumab dose/frequency, and cessation of dupilumab therapy led to improvement, but moderate or severe symptoms persisted for many months.
CONCLUSION:
We report a new musculoskeletal syndrome of inflammatory enthesitis/arthritis/tenosynovitis in some patients receiving the IL-4 receptor antagonist dupilumab. This response to a cytokine-targeting therapy provides key insights into the pathogenesis of enthesitis.
2022-11-01·Explore (New York, N.Y.)4区 · 医学
Effects of meditation compared to music listening on biomarkers in breast cancer survivors with cognitive complaints: secondary outcomes of a pilot randomized control trial
4区 · 医学
Article
作者: Henneghan, Ashley M ; Harrison, Michelle L ; Fico, Brandon G ; Kesler, Shelli R ; Wright, Michelle L
CONTEXT:
We previously reported positive behavioral effects of both daily mantra meditation and classical music listening interventions in breast cancer survivors with cancer related cognitive complaints.
OBJECTIVE:
The objective of this pilot study was to compare the effects of the meditation intervention to a music listening intervention on biomarkers of inflammation and cellular aging (secondary outcomes) in breast cancer survivors.
DESIGN:
Randomized control trial, baseline data collection (time 1), post intervention data collection (time 2) SETTING: Community-based, Central Texas PARTICIPANTS: 25 breast cancer survivors (BCS) who were 3 months to 6 years post chemotherapy completion and reported cognitive changes.
INTERVENTION(S):
Kirtan Kriya meditation (KK) or classical music listening (ML), 8 weeks, 12 min a day MAIN OUTCOME: Telomerase activity [TA], c-reactive protein [CRP], soluble IL-2 receptor alpha [sIL-2Rα], soluble IL-4 receptor [sIL-4R], soluble IL-6 receptor [sIL-6R], soluble tumor necrosis factor receptor II [sTNF-RII], VEGF receptor 2 [sVEGF-R2], and VEGF receptor 3 [sVEGF-R3] RESULTS: Repeated measures analysis of variance models were analyzed from time 1 to time 2 by group for each biomarker. A pattern of greater telomerase activity across time in both groups (F (1,15) = 3.98, p = .06, ω2 = 0.04); significant decreases in sIL-4R across time for both groups (F (1,22) = 6.28, p = .02, ω2 = .003); group*time effect was nominally different but not statistically different for sIL-4R (F(1,22) = 3.82, p = .06, ω2 = .001); and a pattern for a group*time effect with ML group showing higher levels of sVEGF-R3 at time 2 (F (1,20) = 2.59, p = .12, ω2 = .009). No significant effects were found for CRP, sIL-2Rα, sIL-6R, sTNF-RII, or sVEGF-R2.
3
项与 IL-4R(Amgen, Inc.) 相关的新闻(医药)2024-11-18
Biosion Announces Exclusive, Global License Agreement with Aclaris Therapeutics on two potential First-in-Class and Best-in-Class Immunology Assets
- Biosion receives over $40 million cash payment as upfront license fee and reimbursement for certain development costs & drug product materials, and 19.9% shares of common stock of Aclaris Therapeutics; additional regulatory and sales milestone payments exceed $900M with tiered single digit sales royalty
- Aclaris Therapeutics will be responsible for development and commercialization of BSI-045B, a novel anti-TSLP monoclonal antibody, and BSI-502, a novel bispecific antibody that is directed against both TSLP and IL4R worldwide (excluding Greater China)
Newark, DE, U.S. and Nanjing, China
Nov. 18, 2024 -- Biosion, Inc. (Biosion), a global R&D stage biotechnology company, today announced that it has entered into an exclusive license agreement with Aclaris Therapeutics (NASDAQ: ACRS) for worldwide rights (excluding Greater China) to BSI-045B, a potential First-in-class, clinical-stage, novel anti-TSLP monoclonal antibody, and BSI-502, a potential best-in-class, pre-clinical stage, novel bispecific antibody that is directed against both TSLP and IL4R.
Under the terms of the agreement, Biosion shall receive over $40 million cash payment as upfront license fee and reimbursement for certain development costs & drug product materials. Biosion shall also receive 19.9% shares of common stock of Aclaris Therapeutics. Additional regulatory and sales milestone payments exceed $900M with tiered low-to-mid single digit royalty based upon a percentage of annual net sales.
“This transaction once again validates our proprietary antibody discovery platforms, BSI-045B and BSI-502 demonstrate the power of our discovery capabilities in generating potential first-in-class and best-in-class therapeutics.” said Mingjiu Chen, Ph.D., Founder and CEO of Biosion. “Together with our global partners through the complementary expertise and resources, we hope to bring more transformative medicines to patients worldwide.”
“This transaction marks Biosion’s ninth and tenth global partnered programs under our ‘discover-development-partnership’ model,” said Anthony Yeh, Ph.D., Chief Strategy Officer of Biosion. “This partnership accelerates the worldwide clinical development of two key assets while securing the financial resources necessary to advance our other innovative programs to IND filings.”
In a completed Phase 2a, single-arm, proof-of-concept trial in the United States in 22 patients with moderate to severe atopic dermatitis, BSI-045B was observed to have a pharmacodynamic, safety and efficacy profile that could position it as a potential best-in-class therapy. BSI-045B is also being advanced in multiple Phase 2 studies in China by Biosion's regional partner, Chia Tai Tianqing Pharmaceutical Group, Co., Ltd. (CTTQ), targeting both severe asthma and chronic rhinosinusitis with nasal polyps, accelerating the potential to show proof-of-concept across additional indications.
"This strategic transaction brings together Biosion’s innovative programs with our development capabilities in immunology, and we are excited to enhance our portfolio with these compelling assets," said Dr. Neal Walker, Interim CEO and Chair of the Board of Directors of Aclaris. "We look forward to further developing these assets in our quest to deliver exceptional therapies for patients worldwide."
About BSI-045B
BSI-045B is a humanized anti-thymic stromal lymphopoietin (TSLP) monoclonal antibody that specifically binds to human TSLP, blocking its interaction with the receptor complex and disrupting signal transduction. This mechanism prevents immune cells targeted by TSLP from releasing proinflammatory cytokines. BSI-045B is currently in clinical development, and its safety and efficacy have not been evaluated by regulatory authorities.
About BSI-502
BSI-502 is a humanized anti-TSLP and anti-interleukin 4 receptor (IL4R) bispecific antibody that blocks both the upstream TSLP receptor signal transduction and downstream IL4R activation thereby inhibiting this central proinflammatory pathway. BSI-502 is currently in pre-clinical development, and its safety and efficacy have not been evaluated by regulatory authorities.
About Biosion
Biosion is a global clinical-stage biotechnology company focused on developing innovative antibody-based therapies to improve outcomes for patients with immune and oncologic diseases. Leveraging its proprietary technologies such as the H³ antibody discovery platform, SynTracer® HT endocytosis platform, and Flexibody® bispecific platform, Biosion creates highly differentiated medicines to address significant unmet medical needs worldwide. With its “discover-development-partnership” model, the company has forged 10 global collaborations, advancing seven clinical-stage assets, including monoclonal antibodies, bispecific antibodies, and antibody-drug conjugates. Biosion operates across the US, China, and Australia. Learn more at www.Biosion.com.
引进/卖出
2024-07-25
·医药笔记
▎Armstrong
迈威生物成立于2017年,成立七年多以来,已有14款研发管线进入临床阶段,覆盖肿瘤、自免、代谢、眼科、感染五大治疗领域,药物形式覆盖单抗、双抗、ADC、重组蛋白等。
9MW2821:首款国产Nectin-4 ADC
9MW2821基于其IDDC定点偶联技术平台开发,经过充分的差异化设计,采用可专利的桥连子及经过优化的偶联反应条件,实现定点修饰。三阴性乳腺癌动物模型中显示其抗肿瘤活性优于已上市的Padcev,在食蟹猴、小鼠体内的安全性均优于Padcev。
9MW2821为首款进入三期临床的国产Nectin-4 ADC,2023年12月,迈威生物注册了Nectin-4 ADC新药9MW2821的三期临床试验,用于治疗局部复发性或转移性尿路上皮癌。
9MW2821在适应症拓展方面同样推进迅速。2024 年 2 月,获 FDA 授予快速通道认定用于治疗晚期、复发或转移性食管鳞癌;5 月,先后获孤儿药认定和快速通道认定用于治疗食管癌和既往接受过含铂化疗方案治疗失败的复发或转移性宫颈癌;7 月,获快速通道认定用于治疗局部晚期或转移性 Nectin-4 阳性三阴性乳腺癌。多项审评资质的获得也提示了美国在该些适应症的治疗新手段的稀缺。
临床表现方面,9MW2821 是全球首个在宫颈癌、食管癌以及乳腺癌适应症披露临床有效性数据的靶向Nectin-4 的治疗药物,截至2024年5月23日,目标剂量下累计入组已超过350 例。根据其在2024 ASCO大会发布的数据,II 期临床 1.25mg/kg 剂量组的 240 例患者中,37 例尿路上皮癌患者ORR和DCR分别为62.2%和91.9%,mPFS为8.8个月,mOS为14.2个月。53 例宫颈癌患者ORR和DCR分别为35.8%和81.1%,mPFS为3.9个月,mOS尚未达到。Nectin-4肿瘤细胞染色强度 3+ 的39例宫颈癌患者ORR为 43.6%。39 例食管癌患者ORR和DCR分别为23.1%和69.2%,mPFS 为3.9个月,mOS为8.2个月。20 例局部晚期或转移性三阴性乳腺癌患者ORR和DCR分别为50.0%和80.0%,mPFS 为 5.9 个月,mOS 尚未达到。
此外,9MW2821联合 PD-1 一线疗法已针对尿路上皮癌和三阴性乳腺癌分别开展临床试验。宫颈癌、食管癌、三阴性乳腺癌单药治疗均处于II期临床。四大适应症截止到今年年底前有望全面覆盖从一线到二三线患者群体的临床研究。
基于在ADC领域的持续探索,迈威生物后续还布局了Trop2 ADC、B7H3 ADC。两款产品均进入临床阶段,2023年9月完成首例患者给药,其中B7H3 ADC已获批在美国开展临床试验,并于2024年7月获 FDA授予孤儿药认定,用于治疗小细胞肺癌。考虑到其自有技术平台带来的源头创新研发能力,预计未来将有更多ADC产品进入临床试验阶段。
9MW1911:国内首款、全球第二款同靶点药物
9MW1911 是国内首家,全球进度处于第二梯队的靶向ST2单抗创新药,在慢性阻塞性肺疾病(COPD)适应症具有较大市场潜力,目前处于Ib/IIa期临床阶段。全球最领先开发进度为III期临床研究阶段。
临床前研究表明9MW1911 在分子水平上表现出与重组人 ST2 具有高亲和力,同时可以高效阻断 IL-33 与 ST2 的结合,抑制NF-kB 和 MAPK 信号通路的活化,进而抑制 Th2 细胞因子的产生;在细胞学水平上,9MW1911 可以抑制重组人 IL-33 促进 KU812 细胞生成 IL-5 的活性,表现出阻断IL-33/ST2 信号通路的作用;种属交叉试验显示,9MW1911 可以特异性结合人和食蟹猴ST2,与小鼠 ST2 不结合;非临床研究显示该品种动物体内作用机制清晰明确,与 ST2 特异性结合后,能够阻断细胞因子 IL-33 对 ST2 介导的信号通路的激活,抑制炎症反应的发生,从而实现对多种自身免疫性疾病的治疗。9MW1911所针对的 IL33/ST2 通路,系赛诺菲已上市药品Dupilumab(度普利尤单抗注射液,达必妥)所针对的IL-4/IL-13 信号传导通路的上游通路,具有更广泛的抗炎症反应。
目前全球尚无以ST2 或其配体 IL-33 为靶点的抗体药物上市。其2023年年度报告显示,9MW1911已完成健康受试者的 Ia 期临床试验,结果显示 9MW1911 注射液在剂量递增范围内安全且耐受性良好。
COPD赛道上,欧洲最新获批的度普利尤(IL-4R)所针对的COPD分型为II型炎症性患者,主要监测指标是嗜酸粒细胞大于等于300个,据报道这部分人群仅占到COPD全人群的31%左右。因此,COPD仍有巨大市场。目前有三款药物在COPD临床III期研究阶段:1)赛诺菲/再生元开发的IL-33抗体;2)MedImmune/阿斯利康共同开发的IL-33抗体;3)安进/基因泰克(Roche)开发的ST-2抗体。
9MW3011:国内首款TMPRSS6抗体
9MW3011由迈威生物自主研发,是一款针对全新靶点TMPRSS6的单抗新药。作用机制上,9MW3011通过结合肝细胞表面表达的TMPRSS6,上调肝细胞表达铁调素的水平,抑制铁的吸收和释放,从而达到调节体内铁稳态的作用。
2023年1月,迈威生物就 9MW3011 项目与DISC达成独家许可协议,迈威生物可获得合计最高达 4.125 亿美元的首付款及里程碑付款,另外可获得许可产品净销售额最高近两位数百分比的特许权使用费,其中 DISC向迈威生物支付一次性不可退还的首付款 1000 万美元。2023年9月和2024年2月获FDA授予快速通道认定和孤儿药认定,用于治疗真性红细胞增多症。
2024年1月31日,武田引进Protagonist Therapeutics在研的铁调素(Hepcidin)类似物Rusfertide,武田获得一半美国商业权益,以及美国外商业权益。根据协议,武田支付3亿美元预付款,额外的里程碑金额以及美国外的销售分成。同作为铁稳态调节药物,9MW3011也具有巨大的潜在临床价值和商业价值。
9MW3811:国内首款IL-11抗体
IL-11在多种疾病的发病机制中扮演重要角色,从1989年克隆其基因以来,科学界对该靶点研究的越来越深入。9MW3811已在澳、中、美三地获批开展临床试验,是国内企业开发的首个获批进入临床的抗IL-11 单抗。9MW3811可高效阻断 IL-11 下游信号通路的活化,抑制 IL-11 诱导的病理生理功能,从而达到对纤维化和肿瘤的治疗效果。
除上述管线外,迈威生物还有肿瘤免疫的PD-L1/CD47双抗、抗感染的α-toxin抗体9MW1411等品种处于II期临床阶段。
商业化阶段产品:地舒单抗、阿达木单抗
迈威生物在生物类似药领域布局颇多,地舒单抗、阿达木单抗均已获批上市,2023年12月继续纳入医保目录。地舒单抗还被纳入《中国临床肿瘤学会 (CSCO) 乳腺癌诊疗指南 2024》。国际化方面也在快速推进,地舒单抗在12个国家达成正式协议,在巴基斯坦提交上市申请;阿达木单抗在13个国家达成正式协议,并在印尼、巴基斯坦和埃及提交上市申请;阿柏西普眼内注射液在1个国家达成正式协议。
2023年12月,迈威生物长效G-CSF 8MW0511的上市申请也获得NMPA受理,用于成年非髓性恶性肿瘤患者在接受容易引起发热性中性粒细胞减少症的骨髓抑制性抗癌药物治疗时,使用本品降低以发热性中性粒细胞减少症为表现的感染发生率。
阿柏西普类似药9MW0813处于三期临床阶段,也已开展海外合作。在眼科领域,迈威生物还布局了VEGF-A特异性和高亲和力的抗体9MW0211,同样已经进入三期临床。
总结
迈威生物经过7年多发展建立起具有竞争力的差异化创新管线和生物类似药管线。创新药方面,重点布局ADC领域,在Nectin-4 ADC的部分适应症赛道已位居全球首位,逐渐完成从后线到前线的覆盖,尤其在三阴性乳腺癌领域具备先发优势;抗TMPRSS6单抗项目完成高额海外授权;重点品种抗ST2单抗、抗IL-11抗体目前全球均尚无优效的治疗手段获批,市场潜力巨大。随着9MW2821四大适应症的陆续明确和数据披露,以及对公司研发平台实力的验证,相信迈威生物将能够释放更多潜力。生物类似药方面,产品陆续上市,并广泛开拓海外国家合作。两条业务发展路线相辅相成,未来前景可期。
参考材料迈威生物官网;Iron metabolism and iron disorders revisited in the hepcidin era.
Armstrong技术全梳理系列
GPRC5D靶点全梳理;
CD40靶点全梳理;
CD47靶点全梳理;
补体靶向药物技术全梳理;
补体药物:眼科治疗的重要方向;
Claudin 6靶点全梳理;
Claudin 18.2靶点全梳理;
靶点冷暖,行业自知;
中国大分子新药研发格局;
被炮轰的“me too”;
佐剂百年史;
胰岛素百年传奇;
CUSBEA:风雨四十载;
中国新药研发的焦虑;
中国生物医药企业的研发竞争;
中国双抗竞争格局;
中国ADC竞争格局;
中国双抗技术全梳理;
中国ADC技术全梳理;
Ambrx技术全梳理;
Vir Biotech技术全梳理;
Immune-Onc技术全梳理;
亘喜生物技术全梳理;
康哲药业技术全梳理;
科济药业技术全梳理;
恺佧生物技术全梳理;
同宜医药技术全梳理;
百奥赛图技术全梳理;
腾盛博药技术全梳理;
创胜集团技术全梳理;
永泰生物技术全梳理;
中国抗体技术全梳理;
德琪医药技术全梳理;
德琪医药技术全梳理2.0;
和铂医药技术全梳理;
荣昌生物技术全梳理;
再鼎医药技术全梳理;
药明生物技术全梳理;
恒瑞医药技术全梳理;
豪森药业技术全梳理;
正大天晴技术全梳理;
吉凯基因技术全梳理;
基石药业技术全梳理;
百济神州技术全梳理;
百济神州技术全梳理第2版;
信达生物技术全梳理;
信达生物技术全梳理第2版;
中山康方技术全梳理;
复宏汉霖技术全梳理;
先声药业技术全梳理;
君实生物技术全梳理;
嘉和生物技术全梳理;
志道生物技术全梳理;
道尔生物技术全梳理;
尚健生物技术全梳理;
康宁杰瑞技术全梳理;
科望医药技术全梳理;
科望医药技术全梳理2.0;
岸迈生物技术全梳理;
礼进生物技术全梳理;
康桥资本技术全梳理;
余国良的抗体药布局;
荃信生物技术全梳理;
安源医药技术全梳理;
三生国健技术全梳理;
仁会生物技术全梳理;
乐普生物技术全梳理;
同润生物技术全梳理;
宜明昂科技术全梳理;
派格生物技术全梳理;
迈威生物技术全梳理;
Momenta技术全梳理;
NGM技术全梳理;
普米斯生物技术全梳理;
普米斯生物技术全梳理2.0;
三叶草生物技术全梳理;
贝达药业抗体药全梳理;
泽璟制药抗体药全梳理;
恒瑞医药抗体药全梳理;
齐鲁制药抗体药全梳理;
石药集团抗体药全梳理;
豪森药业抗体药全梳理;
华海药业抗体药全梳理;
科伦药业抗体药全梳理;
百奥泰技术全梳理;
凡恩世技术全梳理。
抗体药物偶联物孤儿药临床3期ASCO会议快速通道
2024-05-13
摘要:全球超过3亿人饱受哮喘困扰,且发病率逐年增加,作为最常见的慢性疾病之一,哮喘激发机制复杂,异质性强,是一类免疫介导的炎症疾病。随着基于生理、病理等机制深入研究,陆续推出的治疗性小分子和激素药物已实现对大多数患者的控制和治疗,但仍有5%~10%的患者为难控制、难治疗性哮喘,即重度哮喘。近十年来,得益于生物医药的迅猛发展,蛋白、抗体类药物凭借高有效性、高特异性、高安全性成为关键的重度哮喘治疗药物,然而由于生物制剂通常经注射给药,不可无创、快速地递送药物直接入肺迅速吸收起效,故而临床中亟需起效快、便捷、经济、安全的吸入式生物制剂。本综述通过对哮喘发病机制的阐释,概括现行的小分子、激素类和生物类治疗药物,并总结吸入式哮喘生物制剂的研发历程,对其未来前景进行分析,旨在加深对大分子吸入药物研究方向的认知,更新该领域前沿动态,以期为更多吸入式生物制剂的开发提供参考。关键词:哮喘;给药途径;吸入给药;生物制剂_正文_支气管哮喘简称哮喘,以呼吸急促、咳嗽和胸闷等多种症状为特征,与慢性气道炎症,广泛、可逆性呼气性气流阻塞和气道高反应性等症状有关[1]。全球超过3亿人饱受哮喘困扰,且发病率逐年增加。根据全球哮喘指南全球哮喘防治创议(global initiative for asthma,GINA)建议,需对患者采用阶梯式治疗方案,尤其推荐个性化治疗方案。随着生物制剂的发展,抗体类生物制剂已成功应用至哮喘治疗,为中重度哮喘患者带来了更有效、更安全的药物选择,但其注射的给药方式,缓慢的起效速度,频繁的入院用药、高昂的用药成本在很大程度上限制了药物的推广,故而吸入式抗体药物的临床呼声日益高涨。本综述对哮喘机制、现有治疗药物及吸入抗体药物的开发历程进行了调查研究,以期加深对哮喘发病机制及治疗药物的全面认识,同时对吸入式抗体药物的开发必要性、开发难度、研发现状进行汇总更新。1哮喘发病机制哮喘传统上被分为两类,外源性哮喘和内源性哮喘,外源性哮喘又被称为过敏性哮喘,由过敏原引起,主要归因于异常的辅助性T细胞(helper T lymphocyte,Th)2型炎症。内源性哮喘是由多种因素引发的,如阿司匹林、肺部感染、运动、寒冷、压力、肥胖等[2]。根据Th2炎症的状态,哮喘疾病又被分为高Th2和低Th2哮喘[3]。高Th2哮喘的特征是嗜酸性气道炎症,这与血液中嗜酸性粒细胞数目的增加或呼出一氧化氮(fractional exhaled nitric oxide,FeNO)分数升高有关,而低Th2哮喘包括中性粒细胞哮喘和寡粒细胞性哮喘。哮喘的病理机制是复杂的,新研究表明,哮喘的发病机制也可不依赖于非免疫应答/炎症发生,但相关研究有限[4]。2哮喘标志物由于丰富的异质性外源、内源表型特征,哮喘非常适合精准治疗方案,因而非侵入性生物标志物的鉴别将有助于其诊断和治疗。Th2和2型固有淋巴细胞(innate lymphoid cells,ILC2)通路的激活是2型炎症哮喘发生的核心,Th2型细胞因子主要有一系列白介素(interleukin,IL),如IL-4、IL-5、IL-9、IL-13、IL-31,而ILC2s产生的2型细胞因子主要有IL-5、IL-9和IL-13,上述因子均为哮喘有效标志物。嗜酸性粒(eosinophil,EOS)细胞、免疫球蛋白(immunoglobulin,Ig)E、表达ILC2s的循环趋化因子受体(cyclic chemokine receptor,CCR)10、血浆CCL(CC chemokine ligand)27水平和Fe-NO是哮喘诊断和预后的常见生物标志物。此外,如图1所示,IgE、IgG亚族,血清IgE抑制活性,嗜碱性粒细胞活化,趋化因子和细胞因子,调节性T(regulatory T,Treg)细胞,调节性B(regulatory B,Breg)细胞和树突状细胞等标记物已被推荐作为跟踪过敏原耐受诱导治疗的生物标记物[5]。3哮喘现有小分子、激素治疗药物GINA指南推荐使用药物种类包括糖皮质激素、β2-受体激动剂、胆碱能受体阻断药物、茶碱类药物、白三烯受体拮抗剂和生物靶向制剂[6]。除上述药物外,基于创新机制的新型小分子药物研发处于临床研究不同阶段,如正大天晴的前列腺素D2受体拮抗剂(又称为CRTH2拮抗剂)TQC3564正在中国临床I期研究阶段;针对磷酸二酯酶4(PDE4)抑制剂的NCT04108377正处于临床I期患者招募阶段。此外,基于蛋白激酶抑制剂(NCT00676572)、选择性糖皮质激素受体调节剂(AZD7594)、他汀类药物(NCT02433535)均以控制哮喘为目标并处于临床开发中[7]。新型小分子哮喘治疗药物的有效开发离不开对哮喘机制以及哮喘的不同表型和内型的理解,但从临床效果上看,针对中重度哮喘,生物制剂展现出更加明确的应用前景,因此在另一方向上,生物靶向药物的发展也在高歌猛进,力求在新维度上实现哮喘疾病的控制及治疗。4哮喘现有靶向治疗药物随着病情严重,在以糖皮质激素为基础治疗后仍会出现控制不佳、反复重症发作等情况,此时患者即可被认定是重度患者,高达10%的成人和2.5%的儿童哮喘群体患有重度哮喘,因固定气流受限、病情恶化、常年就医、住院和死亡风险的增加等因素致使生活质量严重下降。因此,在整个哮喘患者谱系中,重症哮喘患者是临床中最迫切的未满足医疗需求群体,目前推荐的有效控制方案是生物制剂添加疗法。目前共6款针对Th2通路的哮喘治疗性单克隆抗体获批,包括靶向IgE的奥马珠单抗(omali-zumab)、靶向IL-5的瑞替珠单抗(reslizumab)、IL-5受体药物贝那利珠单抗(benralizumab)、针对IL-4Rα的度普利尤单抗(dupilumab)以及靶向TSLP的特泽鲁单抗(tezepelumab)。目前针对低Th2型、中性粒细胞为主的重度哮喘发病机制相关分子靶点的试验性药物非常稀缺,因此,针对不同表型基础生物学机制的深入研究将对临床有关重度哮喘治疗方案的开发起到关键性驱动作用[8]。抗体类生物制剂的获批为重度哮喘病人带来了更新、更安全、更有效的治疗方式,表1对Th2通路相关靶点哮喘治疗药物关键信息进行统计,以期对哮喘生物制品有更为系统的理解。上述靶向药物已在临床试验及疾病用药过程中验证了有效性和安全性,多种潜在疾病治疗靶点药物研发正在进展中,包括细胞因子、膜分子和细胞内信号通路,如IL-37、IL-11,基质相互作用分子1(STIM1),CD200R、OX40L和CD30L、HMGB等[9]。5吸入式大分子哮喘治疗药物发展现状5.1 药物通过吸入途径递送的优势吸入给药(肺给药)可快速、无创地输送至呼吸道并传导至肺部,肺内巨大的表面积和广泛的血管分布能使药物快速吸收并发挥治疗效果,其剂量对身体其他部位的暴露量较低,可最大限度地减少全身性不良反应。吸入给药是一种无创的局部和全身给药方式,在治疗多种疾病方面具有巨大的潜力,已应用于包括哮喘、慢性阻塞性肺疾病、呼吸道病毒感染和糖尿病、囊性纤维化等多种疾病领域[10]。吸入给药作为治疗呼吸系统疾病优选的给药方案,可满足如下几项对给药方式的特殊需求[11]:(1)药物通过系统性给药在肺内很难达到有效富集的药物浓度,生物利用度低,而吸入给药具有肺内高浓度的优势。(2)当以达到肺部有效治疗浓度为目标给药时,药物剂量通常较大并呈现全身性毒性,吸入给药用药量低,规避胃肠降解作用和肝脏首过效应,安全性更好。(3)需要快速起效的肺部疾病治疗药物,吸入给药可快速实现肺部药物沉降,并由于肺部表面积大、血管化丰富、组织通透性好,药物可以快速吸收并起效。(4)在确保肺部疾病治疗药物药效的同时,若对用药成本关注,亦通过吸入给药的方式改进。5.2 吸入制剂的临床应用吸入型产品可分为雾化溶液吸入、干粉吸入和软雾吸入三大种类,吸入制剂被广泛应用于多种呼吸系统疾病领域[11]:如(1)糖皮质激素、β2受体激动剂、毒蕈碱乙酰胆碱受体拮抗剂等激素和小分子类吸入制剂(组合)用于哮喘和慢性阻塞性肺疾病治疗。(2)妥布霉素、氨曲南赖氨酸盐、左氧氟沙星、阿米卡星、多粘菌素、神经氨酸酶抑制剂、利巴韦林、喷他脒等吸入制剂在抗感染领域的应用。(3)吸入重组人脱氧核糖核酸酶在囊性纤维化适应症的治疗应用。(4)吸入甘露醇在气道黏液清除、囊性纤维化及支气管扩张中的应用。(5)吸入前列腺环素发挥血管扩张机制用于肺动脉高压的治疗。(6)吸入肺表面活性剂用于呼吸窘迫综合征的治疗。(7)吸入核酸药物如重组新型冠状疫苗在抗病毒感染领域的开发应用。吸入制剂主要应用在呼吸系统疾病领域,但从口鼻吸入通过呼吸道将药物递送至肺部并交换进入体循环,也为系统性疾病的治疗提供了创新性的方案[11]。在中枢神经系统领域,尼古丁吸入剂(器)已在尼古丁替代疗法中得到成功应用以减轻或摆脱尼古丁依赖。全球首款左旋多巴吸入粉雾剂INBRIJA®已在美国和欧盟获批上市用于帕金森的吸入治疗。糖尿病领域,辉瑞与Mannkind曾先后获批两种吸入胰岛素药物Exu-bera®与Afrezza®,为糖尿病治疗提供了新的治疗选择。5.3 吸入大分子药物在哮喘治疗领域的开发现状抗体具有高特异性、高亲和力和高安全性等优势,已被广泛用于治疗肺部疾病,如肺癌和严重哮喘。目前共有6款针对重度哮喘的单克隆抗体上市,并取得了优异的临床治疗效果。然而,已上市产品均以皮下注射或静脉输注方式递送,其局限性包括侵入性、系统毒性(如细胞因子风暴),需专业医师操作等因素使得大量患者难以院外治疗。因此,吸入给药凭借直接呼吸道给药,无创、局部效应增强、全身暴露最小和易于自我给药等优点,已在多个吸入性单克隆抗体及其衍生物在临床试验中得到治疗潜力的评估[12],表2汇总了吸入式生物制剂在哮喘领域的简要总结。5.3.1 吸入单克隆抗体/片段诺和诺德及诺华曾在轻度过敏性哮喘患者中开展了奥马珠单抗自我雾化给药疗效研究,虽然33名受试者在分别连续雾化用药2周和8周后血清中均检测到奥马珠单抗,但血清IgE水平无明显变化,变应原诱导的气道反应不受抑制,推测无效的原因是肺组织中覆盖IgE效应细胞的奥马珠单抗浓度不足,无法中和肺部产生的IgE。此外,一名受试者产生了抗奥马珠单抗抗体,这也令人担心吸入单抗可能比注射给药免疫原性更强[13]。2014年,来自比利时的研究团队发表了关于雾化吸入抗原结合片段(fragment of antigen binding,Fab)在哮喘小鼠模型(NMRI)中的研究结果[14],该研究通过聚乙二醇化延长了IL-17A Fab2和IL-13Fab双抗在呼吸道局部停留时间,肺内聚乙二醇化抗体含量在给药后4h达到平台期,而未成功偶联聚乙二醇的抗体则入肺后就开始清除,研究阐释了聚乙二醇化修饰对治疗性蛋白局部递送到肺部后的长效具有显著效果。此后,该团队又相继针对不同分子量聚乙二醇分别对IL-17A Fab和IL-13Fab长效后肺部沉积进行了探索,上述研究主要聚焦肺部药物长效化机制研究,并未对抗体靶点组合的药效及成效性进行深入探索,但初步论证了吸入抗体给药途径的可行性。比利时布鲁塞尔的优时比制药(UCB Pharma)在食蟹猴过敏性哮喘模型中布局了针对雾化吸入IL-13靶点Fab抗体CDP7766的研究,该研究发现通过振动网筛雾化器产生的气溶胶仍保持优异的分子活性及纯度,没有明显的降解和聚体产生,并且在动物模型中表现出良好的耐受性,未观察到局部刺激相关的不良反应,并显著抑制了肺泡灌洗液中过敏原诱导的细胞因子和趋化因子的上调,临床上可快速持久地抑制FeNO,健康和哮喘受试者均表现出良好的耐受性(NCT02473939),证明了吸入性抗IL-13抗体片段药物治疗过敏性哮喘的潜力[15]。Faghihi等通过喷雾干燥将肿瘤坏死因子α(tumor necrosis factorα,TNFα)单克隆抗体Infliximab开发成干燥粉剂,用于局部炎症性哮喘治疗,探讨通过吸入给药途径抑制肺部分泌TNFα的可行性,但其粉剂分子与TNFα结合降低了21%,在卵清蛋白刺激小鼠哮喘模型中表现出的TNFα分子抑制活性相对于单抗降低了2个数量级,虽然抗体干粉开发成功与否主要取决于喷雾干燥条件、配方成分及抗体雾化稳定性,但该研究初步提示了单克隆抗体并不合适用于吸入制剂开发[16]。CSJ117是诺华布局的一款针对TSLP靶点的Fab拮抗剂(Ecleralimab),CSJ117采用粉雾剂药物开发方式用以治疗成人轻度特应性哮喘,在其I期临床研究(NCT03138811)中,CSJ117耐受性良好,并减轻过敏原激发后的支气管收缩[17]。2020年诺华启动了一项II期试验,目的是研究CSJ117治疗未得到控制的重度哮喘的药效和安全性研究(NCT04410523),II期临床研究已完成,但暂未数据揭盲,并且由于诺华产业战略调整,该项目暂停了后续开发,正在积极寻求合作转让。安进及阿斯利康在携手成功推出TSLP单克隆抗体Tez-epelumab商业化上市后,快速对上市药物分子进行工程化改造将单克隆抗体皮下注射液开发成为Fab干粉吸入剂型,项目代码为AMG104/AZD8630,该项目已于2022年启动临床I期研究,以评估安全性、耐受性和药代动力学(NCT05110976)。5.3.2 吸入纳米抗体除了单克隆抗体及抗体片段外,诸如Nanobodies®、Anticalin®、Affibody®等新型蛋白骨架类型在基于蛋白质的吸入治疗领域引起了轰动。与全长单克隆抗体相比,新型蛋白骨架体积较小、结构简单,更容易结合隐蔽结合位点、组织渗透面积更大、药物聚集更方便、热稳定性更强[18]。纳米抗体是由驼科动物体内重链抗体(95kDa)可变区扩增而来的单域抗体,又称为VHH抗体(variable domain of heavy chain of heavy-chain antibody,VHH),从属于基因工程抗体范畴。纳米抗体晶体尺寸直径2.5nm,长4nm,是目前可与抗原结合的最小片段。2018年,首创的Nano-body®caplacizumab(Cablivi®)获得欧洲药品管理局的首个上市许可,用于治疗获得性血栓性血小板减少性紫癜,虽然caplacizumab是肠外用药,但吸入型Nanobody®平台及产品一直在临床开发中。吸入型抗合胞病毒纳米抗体药物ALX-0171的安全性和耐受性已在一项首次婴儿I/IIa期临床试验中得到证实,并观察到抗病毒活性[19]。尽管IIb期剂量范围研究已经完成(NCT02979431),但在下呼吸道感染和炎症发生的基础上,继续雾化ALX-0171并不能改善临床治疗结果,所以该项目未达预期而被搁置研究,但该研究表明ALX-0171在早期的使用干预可产生重要的临床抗病毒作用。随着新冠疫情爆发,许多纳米抗体也被开发成吸入制剂以对抗SARS-CoV-2感染,如洛启生物的Nb11-59表现出强效的体外真病毒中和活性[20]。在哮喘类领域,目前有两款来自洛启生物的纳米抗体吸入产品处于中国临床I期开发阶段,洛启生物靶向IL-4Rα的LQ036产品适应症为嗜酸性粒细胞哮喘,LQ043H靶向TSLP,其适应症为非嗜酸性粒细胞哮喘。LQ036与LQ043H是目前全球推进速度最快的同靶点吸入纳米抗体产品。LQ036及LQ043H均具有优异的体外阻断活性,并按照吸入制剂开发标准进行了完整的空气动力学研究,具有雾化后粒径均匀、生物活性完整保留、药物纯度稳定等优势,同时两款产品均采用毕赤酵母生产系统表达,产量高,工艺简单,药物成本优势明显。LQ036已完成针对安全性、耐受性、免疫原性和药代动力学的澳洲临床I期研究(NCT04993443),安全性、耐受性优异,无药物相关免疫原性发生;目前该项目正处于中国临床I期开发阶段,评价雾化经口吸入LQ036在中国健康受试者体内的耐受性、安全性、免疫原性和药代动力学(CTR20223011)。洛启LQ043H项目同期处于中国临床I期研究阶段,评价LQ043H单域抗体雾化液在中国健康受试者中单次给药后的耐受性、安全性、药代动力学和免疫原性(CTR20230092)。此外,LQ036的慢性阻塞性肺疾病适应症IND申请获得国家药监局药品审评中心(CDE)受理(受理号:CXSL2300435)。LQ036和LQ043H的顺利推进将为纳米抗体在吸入给药方向上的临床应用提供有力支持,并创新性地为哮喘和慢性阻塞性肺疾病患者提供更有效、更安全、更便捷、更经济的治疗方案。5.3.3 吸入类抗体骨架蛋白Anticalin®是Astra Zeneca与Pieris公司蛋白研发平台推出的一类基于丰富血浆及体液来源脂质运载蛋白骨架修饰开发而成的工程化蛋白,通过对脂质运载蛋白1进行定向蛋白质突变,经过系统性修饰推出了Elarekibep项目(PRS-060),Elarekibep是一款靶向IL-4Rα的高亲和力、缓慢解离、长效拮抗剂,用于治疗中度至重度哮喘。Elarekibep的小尺寸和稳定性使其可以被直接吸入肺部,其临床I期以雾化液方式开展,试验显示轻度哮喘患者的FeNO水平显著降低(NCT03384290,NCT03574805)。在其干粉吸入制剂临床II期向3mg及1mg更低剂量组进行药效探索时,其非人类灵长类动物长期毒性研究出现了非药物剂量依赖的炎症介导的肺组织损伤,随即其合作方阿斯利康终止了该项目后续临床开发(NCT04643158)。该项目的失败原因更多指向于Anticalin®平台分子自身结构毒性问题,这一结论为类抗体支架蛋白Anticalin®的未来研发应用蒙上了阴影[21]。Affibody®是Affibody公司推出的一类新型抗体模拟物,该类药物是由58个氨基酸残基组成的新型亲和配体,相对分子质量约为6500,由3个螺旋结构组成,功能与抗体相似,但具备相对分子质量小、折叠速度快、选择性和亲和力高、结构稳定等优势。Affibody最近宣布接受Chiesi投资,使用Affibody专有技术布局开发呼吸系统疾病吸入性创新疗法,根据公司最新官网更新,ABY-062作为一种靶向TSLP的候选药物,很有可能是按照吸入式治疗药物方向开发,这也意味着吸入大分子药物开发行列迎来了新抗体骨架Affibody®的加入,我们期待其在吸入性创新疗法上的表现。5.3.4 吸入重组蛋白除吸入抗体类药物之外,重组蛋白也曾尝试开发成吸入制剂。拜耳医药曾尝试过IL-4蛋白突变体雾化吸入开发方向,Pitrakinra(AER-001,BAY-16-9996)是IL-4蛋白的变体,具有2个氨基酸变化,使其能够结合IL-4Rα链并抑制IL-4Rα与γC或IL-13Rα1的相互作用。在Pitrakinra的一项II期临床研究中,吸入Pitrakinra可减弱过敏原激发后哮喘受试者的晚期哮喘反应和气道炎症,但对哮喘患者应对过敏原挑战的肺功能没有显著影响[22]。虽然在一项药物遗传学研究中,在携带IL-4Rα基因特异性单核苷酸多态性的患者中,Pitrakinra显著降低哮喘急性发作的频率,但最终其临床III期不明原因并未继续推进。Altrakincept是一款通过雾化给药开发的竞争性重组人可溶性IL-4Rα蛋白,已在临床试验中进行了测试,并证明可预防中重度哮喘患者因停用皮质类固醇而导致的肺功能恶化,但进一步的长期研究结果令人失望,该项目也未再进一步推进[23]。6小结在过去的15年里,生物制剂的开发和临床应用显著扩大,但这些药物的给药途径仍主要以局部或系统性注射为主,吸入抗体是一种明显的无创替代方案,为多种疾病的局部治疗提供了机会,尤其对于呼吸系统疾病治疗,同时也尝试应用于系统性疾病的治疗。但由于抗体三维结构复杂,三级结构是其作用的核心,在吸入给药过程中遭受各种应力下容易产生降解和聚体,这种结构的破坏严重影响产品有效性和安全性等性能的评估,所以维持其结构完整性和生物活性对于成功开发吸入性生物制剂至关重要,这无疑对分子特性、配方设计、辅料选择、加工工艺和吸入装置等方面提出了系统性挑战。后续基础研究对上皮细胞转运机制的理解,辅助抗体稳定性、吸收和雾化性能的相容性赋形剂的开发,适配性更高、便携性更好、操作更便捷吸入装置的开发等都会对吸入抗体药物的开发提供更坚实的开发基础。并且随着纳米抗体、抗体片段、抗体骨架等新形式生物制剂的诞生为更多创新药物的开发提供了保障,未来我们期待更多吸入相关创新技术的诞生。总而言之,吸入型生物制剂的开发是一个系统过程,需要全面、充分考虑发病机理、患者群体、药物分子类型选择及其与肺内生物屏障的相互作用等因素,并合理地选择制剂、赋形剂和装置,同时也涉及上述学科和领域的共同发展。我们希望未来会有治疗效果更佳、安全数据更好、自行用药更便捷、药物价格更低廉的吸入生物制剂产品的诞生以满足日渐复杂、日益壮大的临床未满足需求。参考文献详见《中国临床药理学与治疗学》 2024 Apr; 29(4)识别微信二维码,添加生物制品圈小编,符合条件者即可加入生物制品微信群!请注明:姓名+研究方向!版权声明本公众号所有转载文章系出于传递更多信息之目的,且明确注明来源和作者,不希望被转载的媒体或个人可与我们联系(cbplib@163.com),我们将立即进行删除处理。所有文章仅代表作者观点,不代表本站立场。
100 项与 IL-4R(Amgen, Inc.) 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 哮喘 | 临床2期 | 美国 | - | - |
| 哮喘 | 临床2期 | - | - | |
| 移植物抗宿主病 | 临床前 | 德国 | - | - |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

生物类似药
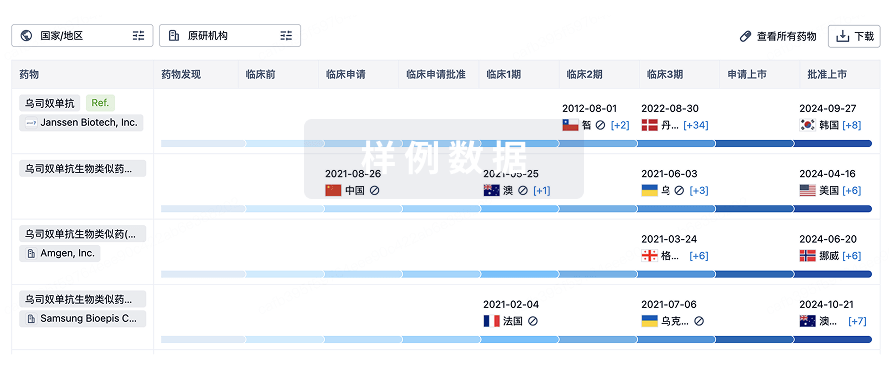
生物类似药在不同国家/地区的竞争态势。请注意临床1/2期并入临床2期,临床2/3期并入临床3期
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或

Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
