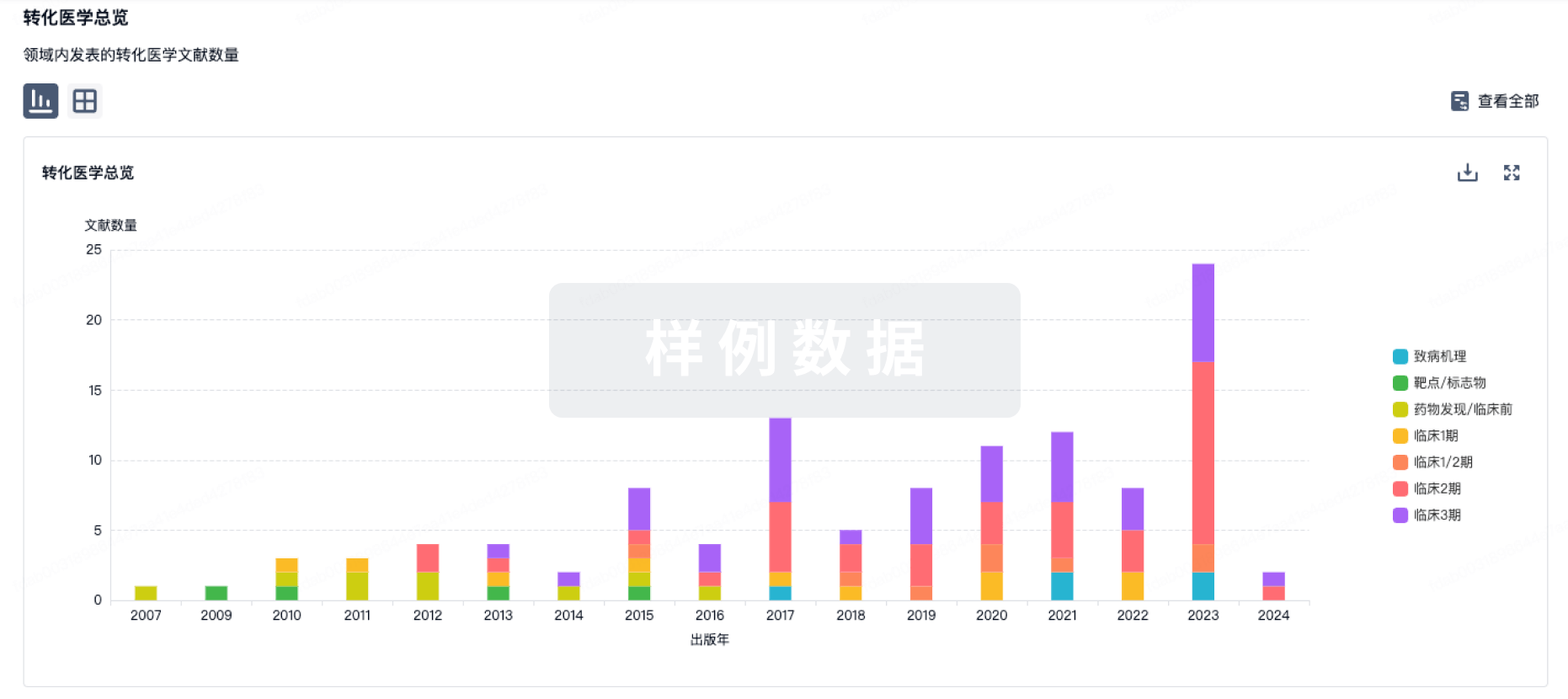
A review.Fox compares the multifunctionality of tRNA synthetases to mol. Swiss Army knives.These conserved mols. are similar to the aminoacyl-tRNA synthetases in other organisms, such as bacteria-but more complex organisms possess more complex aminoacyl-tRNA synthetases with addnl. domains that do not seem to be necessary for protein synthesis.In his lab, Fox and his colleagues study such seemingly extraneous domains of the enzyme glutamyl-prolyl-tRNA synthetase (EPRS), a bifunctional synthetase that attaches both proline and glutamine to their cognate tRNAs.They have discovered that EPRS' extra domains are crucial for the enzyme's noncanonical role of reigning in the immune response after acute inflammation.Several noncanonical roles for the cytoplasmic forms of these mols. (there are also mitochondrial tRNA synthetases) seem to involve the immune response and blood vessel growth.But their link to disease is particularly clear in Charcot-Marie-Tooth (CMT) disease, a peripheral neuropathy that is characterized by muscle weakness, especially in the legs and hands.More than 20 mutations across six different aminoacyl-tRNA synthetases have been linked to this disorder.Albena Jordanova at the University of Antwerp in Belgium linked mutations in the tyrosyl-tRNA synthetase to one particular subtype of this disorder, known as autosomal dominant-intermediate CMT neuropathy type C (DI-CMTC).Several years later, Eric First's lab investigated whether these mutations alter the protein building activities of tyrosyl-tRNA synthetase, or one of its noncanonical roles.Two of the mutations reduce the protein's ability to charge tRNA with tyrosine, but a third does not-it actually leads to a slight increase in enzymic activity relative to the wild-type form.First and his colleagues concluded that the link between mutations in tyrosyl-tRNA synthetase and DI-CMTC might not result from a loss of its ability to catalyze the attachment of tyrosine to its cognate tRNA-because not all the mutations impaired this ability.However, because multiple aminoacyl-tRNA synthetases are mutated in CMT, a noncanonical function might not explain the link either."That is why Xiang-Lei Yang's paper is so intriguing," First says in an e-mail, referring to a finding by one of Schimmel's former postdocs Xiang-Lei Yang, who is now also an investigator at Scripps.Yang found evidence that in another subtype of CMT called CMT2D, mutations in the glycyl-tRNA synthetase cause the degeneration of axons in peripheral nerves by enabling the mutant glycyl-tRNA synthetase to block a neuronal receptor from binding its ligand.Her team discovered that when glycyl-tRNA synthetase is mutated, it can still exit the cell as it normally does.But there, it seems to cling to a neuronal receptor called neuropilin 1 and disrupt the interactions between neuropilin 1and its binding partner that are thought to protect the motor neuron.When mouse models of CMT2D that produce mutated glycyl-tRNA synthetase are injected with more of this binding partner, that protective interaction is rescued."It was a long, long effort," Yang says."But at the end, it worked out beautifully.".In the case of cancer, the noncanonical functions of tRNA synthetase are even less clear.On the one hand, Sunghoon Kim's laboratory at Seoul National University discovered that macrophages secrete glycyl-tRNA synthetase, which reduces tumor growth in cellular and animal models.But on the other hand, an overabundance of another aminoacyl-tRNA synthetase might be connected with more severe disease.Christopher Francklyn-another former Schimmel lab postdoc-and Karen Lounsbury, professors at the University of Vermont, discovered that threonyl-tRNA synthetase is secreted during inflammation and can induce blood cell migration and blood vessel growth.Because cancers need to attract more blood flow as they grow, the scientists suspected that an overabundance of threonyl-tRNA synthetases might promote blood vessel growth and therefore cancer pathogenesis.When they analyzed databases and tissue from human ovarian cancer, they found that protein levels of threonyl-tRNA synthetase increased as disease severity increased-but, paradoxically, so did survival of the patients.The question, however, was whether threonyl-tRNA synthetase promoted blood vessel growth as a result of its canonical role in protein synthesis, or whether it did so while moonlighting.In 2015, Min Guo at the Scripps Research Institute in Florida, Francklyn, and Sunghoon Kim and established that a small mol. called borrelidin blocks the binding sites on threonyl-tRNA synthetase that would otherwise grab onto the amino acid threonine and its cognate tRNA-inhibiting the enzyme's protein synthesis activities, but also blocking angiogenesis.Around the same time, Francklyn, Guo, and Lounsbury published another paper showing that a less toxic borrelidin analog left threonyl-tRNA synthetase's protein building activities relatively intact while blocking its effects on angiogenesis-indicating that the enzyme was indeed moonlighting when it induced new blood vessels to grow.Given the paradoxical correlations between the amount of threonyl-tRNA synthetase, disease severity, and patient survival, it is not yet clear what potential the synthetase might have as a therapeutic target-so Francklyn and Lounsbury are conducting pilot trials with borrelidin and borrelidin analogs in a model of ovarian cancer.Francklyn notes that the therapeutic administration of these drugs will need careful calibration: too much could cause protein synthesis to shut down, and too little might have no effect.Companies are also turning to tRNA synthetases themselves as possible therapeutics.ATyr Pharma, founded by Schimmel and Yang, is examining whether the cytokine-like capabilities of histidyl-tRNA synthetase might make it an effective therapeutic agent for diseases that have an inflammatory component.In a group of autoimmune muscle diseases, autoantibodies frequently target histidyl-tRNA synthetase.Schimmel and his colleagues established that both histidyl-tRNA synthetase and alternatively spliced forms of the enzyme can be secreted when overexpressed in cells-and that the levels of one of these histidyl-tRNA synthetase splice variants are higher in the muscles of patients with one of these muscle conditions, called dermatomyositis.Schimmel and aTyr hypothesize that histidyl-tRNA synthetase is secreted in various forms in an attempt to combat the inflammation.The disorders that aTyr is going after first, facioscapulohumeral muscular dystrophy (FSHD) and limb girdle muscular dystrophy 2B (LGMD2B), also show signs of muscle inflammation secondary to the genetic mutations that cause these diseases.ATyr Pharma is testing a recombinant histidyl-tRNA synthetase named Resolaris that aTyr calls a first-in-class protein therapeutic, and received 'orphan drug' designation from the US Food and Drug Administration for treating FSHD.At the end of March, aTyr announced results of a Resolaris trial in 20 adult patients with FSHD, divided into three cohorts.In two of the three cohorts, one of the inclusion criteria was that the patient shows inflammation by MRI in one skeletal muscle in the legs.Although the researchers did not observe changes in inflammation in the MRIs of the placebo and treatment groups-and 40% of the treated patients developed anti-drug antibodies that did not apparently affect the pharmacokinetics of the drug according to aTyr-the majority of patients treated at the highest dose for one month and three months reported improved "individualized neuromuscular quality of life" in a questionnaire.Schimmel says that trying to translate these mols. into therapeutics for patients is his reward for spending a career on them: "Nature and these enzymes have been calling out to me and leading me down this path," he says."They have a whole other life-i.e., what we call the deep, dark biol.-that was missed.".