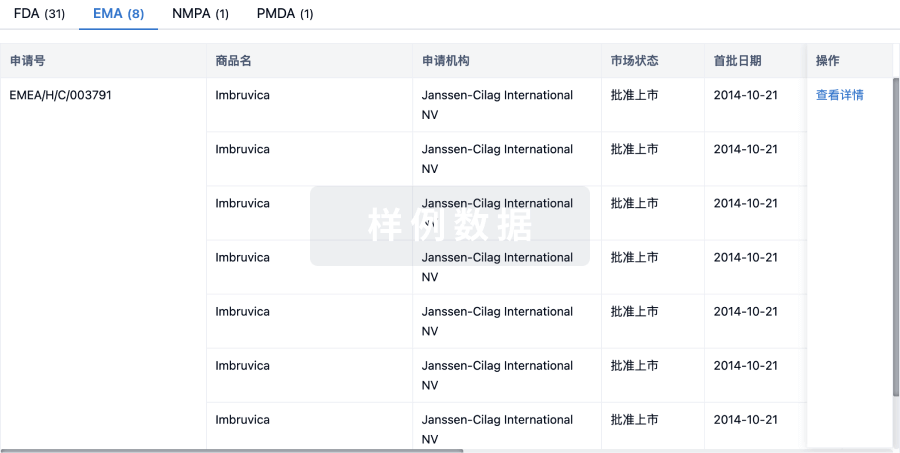
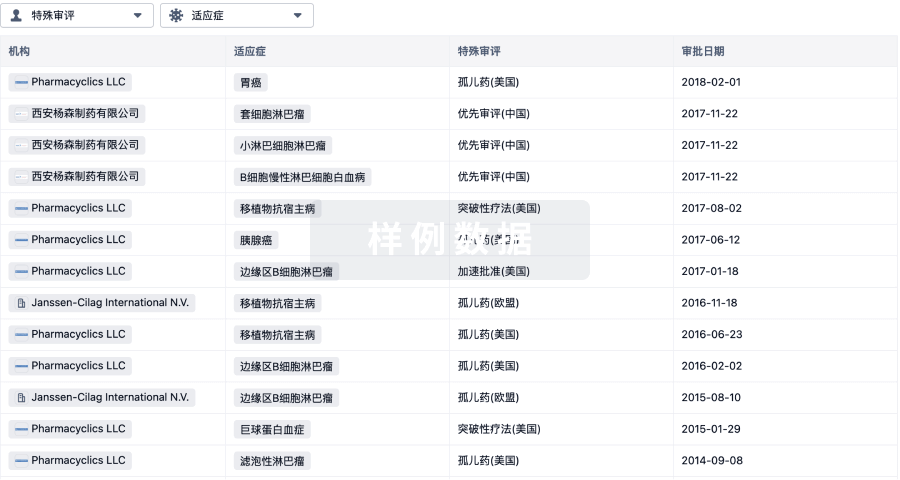
3
项与 Personalized neoantigen tumor vaccine(Ruijin Hospital) 相关的临床试验Clinical Study of XP-004 Personlaized mRNA Vaccine Combined With PD-1 Inhibitor as Adjuvant Therapy for Postoperative Pancreatic Cancer
This study primarily aims to assess the safety and tolerability of XP-004 personalized mRNA vaccines encoding tumor neoantigens combined with PD-1 inhibitor as adjuvant therapy for chemotherapy-intolerant patients following radical pancreatic cancer resection.
Secondary objectives focus on evaluating preliminary efficacy through three parameters: 1) XP-004-induced antigen-specific CD4+/CD8+ T cell activation levels, 2) recurrence-free survival (RFS), and 3) overall survival (OS) in post-operative pancreatic cancer patients receiving this combination therapy.
Clinical Study of Personalized Tumor Vaccines mRNA-0217/S001 and Pembrolizumab in Patients With Advanced Solid Tumors
The main objective of this study was to observe and evaluate the safety and tolerability of mRNA-0217/S001 vaccine encoding personalized tumor neoantigens alone/in combination with Pembrolizumab injection for the treatment of advanced solid tumors. The secondary objective was to observe the preliminary efficacy of mRNA-0217/S001 personalized tumor vaccine in the treatment of advanced solid tumors with neoantigen-specific CD4+ and CD8+ T lymphocyte responses, objective tumor response rate (ORR) and disease control rate (DCR), progression-free survival (PFS) and overall survival (OS) caused by mRNA-0217/S001 personalized tumor vaccine.
Clinical Study of Personalized Tumor Vaccines mRNA-0217/S001 and Pabolizumab in Patients With Advanced Pancreatic Cancer
The main objective of this study was to observe and evaluate the safety and tolerability of mRNA-0217/S001 vaccine encoding personalized tumor neoantigens alone/in combination with Pembrolizumab injection for the treatment of Advanced Pancreatic Cancer. The secondary objective was to observe the preliminary efficacy of mRNA-0217/S001 personalized tumor vaccine in the treatment of advanced solid tumors with neoantigen-specific CD4+ and CD8+ T lymphocyte responses, objective tumor response rate (ORR) and disease control rate (DCR), progression-free survival (PFS) and overall survival (OS) caused by mRNA-0217/S001 personalized tumor vaccine.
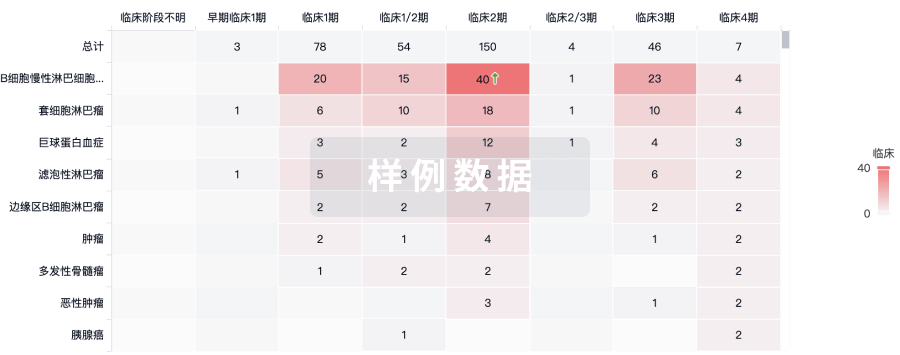
100 项与 Personalized neoantigen tumor vaccine(Ruijin Hospital) 相关的临床结果
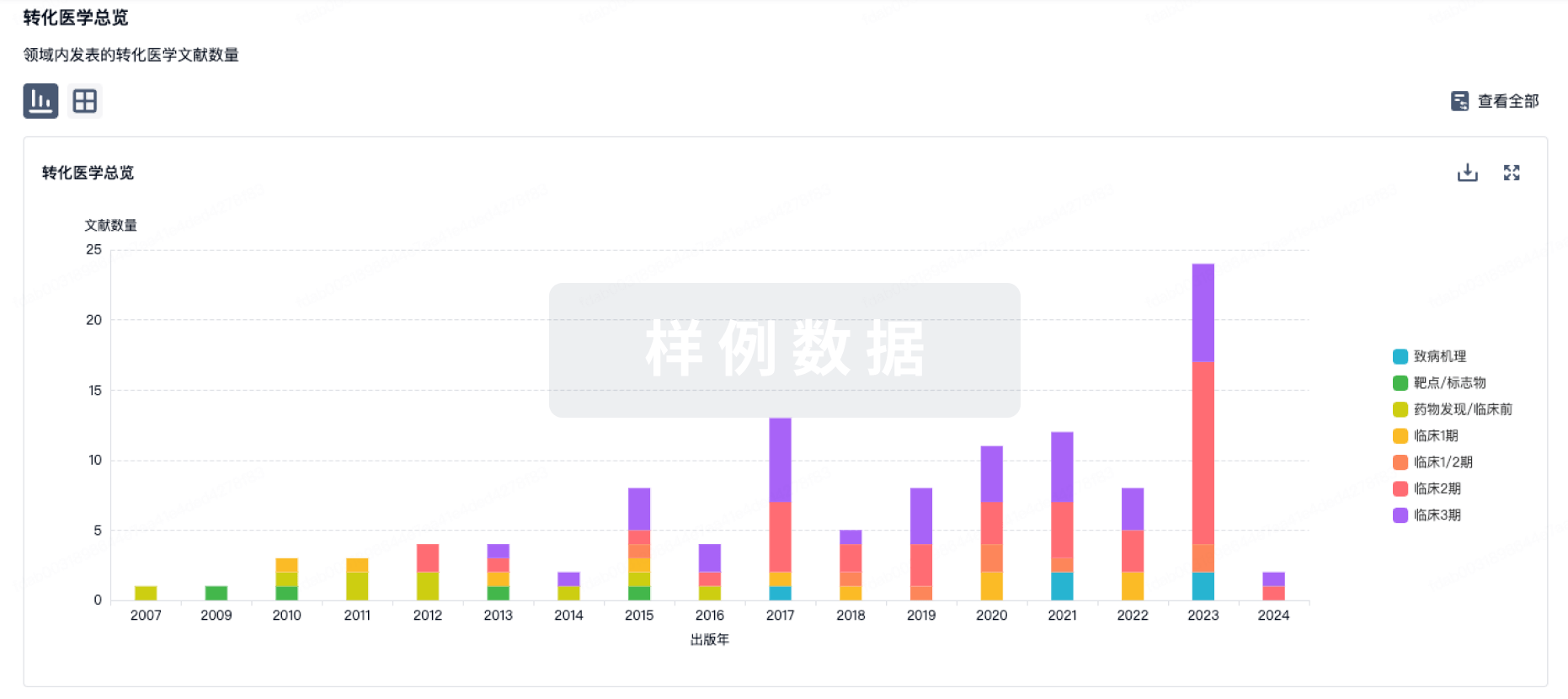
100 项与 Personalized neoantigen tumor vaccine(Ruijin Hospital) 相关的转化医学
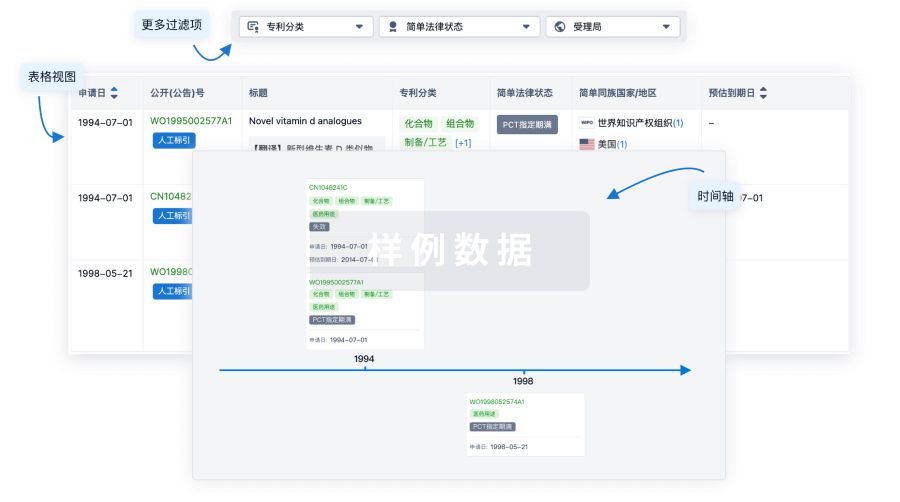
100 项与 Personalized neoantigen tumor vaccine(Ruijin Hospital) 相关的专利(医药)
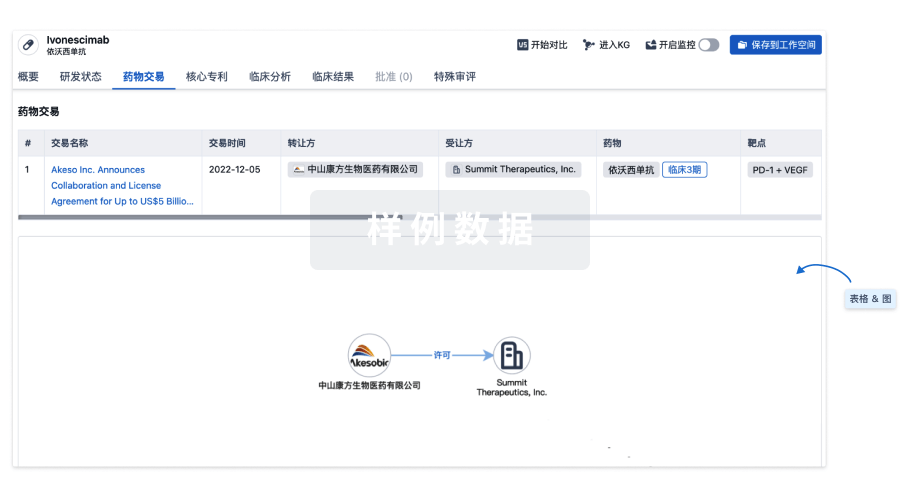
100 项与 Personalized neoantigen tumor vaccine(Ruijin Hospital) 相关的药物交易