预约演示
更新于:2025-01-23
Recombinant modified vaccinia Ankara vaccine(The Aaron Diamond AIDS Research Center)
更新于:2025-01-23
概要
基本信息
在研机构- |
最高研发阶段终止临床1期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
关联
1
项与 Recombinant modified vaccinia Ankara vaccine(The Aaron Diamond AIDS Research Center) 相关的临床试验NCT00252148
Randomized, Placebo-Controlled, Dose-Escalating, Double-Blinded Phase 1 Safety and Immunogenicity Study of a Modified Vaccinia Ankara (MVA) Vectored HIV-1 (ADMVA) Vaccine Administered Intramuscularly to HIV-Uninfected, Healthy Volunteers
The purpose of this study is to determine the safety of an immune response to an investigational HIV vaccine, ADMVA, at three different dosage levels, in adults who are not infected with HIV
开始日期2005-01-01 |
申办/合作机构 |
100 项与 Recombinant modified vaccinia Ankara vaccine(The Aaron Diamond AIDS Research Center) 相关的临床结果
登录后查看更多信息
100 项与 Recombinant modified vaccinia Ankara vaccine(The Aaron Diamond AIDS Research Center) 相关的转化医学
登录后查看更多信息
100 项与 Recombinant modified vaccinia Ankara vaccine(The Aaron Diamond AIDS Research Center) 相关的专利(医药)
登录后查看更多信息
238
项与 Recombinant modified vaccinia Ankara vaccine(The Aaron Diamond AIDS Research Center) 相关的文献(医药)2024-12-01·The Lancet Global Health
Modelling vaccination approaches for mpox containment and mitigation in the Democratic Republic of the Congo
Article
作者: Shaw, Souradet Y ; Rimoin, Anne W ; Bogoch, Isaac I ; Hoff, Nicole A ; Mbala-Kingebeni, Placide ; Savinkina, Alexandra ; Pitzer, Virginia E ; Gonsalves, Gregg S ; Kindrachuk, Jason
2024-12-01·Breastfeeding Medicine
Unique Kinetics of the Human Milk Antibody Response to JYNNEOS Vaccine for Prevention of Monkey Pox: A Case Study
Article
作者: Yang, Xiaoqi ; DeCarlo, Claire ; Powell, Rebecca L.R. ; Fox, Alisa
2024-10-01·The Lancet Infectious Diseases
Safety, immunogenicity, and optimal dosing of a modified vaccinia Ankara-based vaccine against MERS-CoV in healthy adults: a phase 1b, double-blind, randomised placebo-controlled clinical trial
Article
作者: Krähling, Verena ; Rottstegge, Monika ; Mayer, Leonie ; Mykytyn, Anna Z ; Ly, My Linh ; Addo, Marylyn M ; Dahlke, Christine ; Renevier, Niclas ; Kaltenberg, Laura ; Klüver, Michael ; Sandkuhl, Maren ; Tscherne, Alina ; Gerresheim, Gesche K ; Weskamm, Leonie M ; Borregaard, Saskia ; Neubert, Andreas ; van Gorp, Eric C M ; Kupke, Alexandra ; Zoran, Tamara ; Funk, Jana ; Haagmans, Bart L ; Kalomidou, Georgia ; Becker, Stephan ; Schlesner, Claudia ; Schwamborn, Klaus ; Sutter, Gerd ; Kardinahl, Simone ; Mellinghoff, Sybille ; Dieck, Marie-Louise ; Kelidou, Anastasia ; Volz, Asisa ; Fathi, Anahita ; Grüttner, Cordula ; Hesterkamp, Thomas ; Friedrich, Monika ; Hardtke, Svenja ; Raadsen, Matthijs P ; Schröder, Simon
100 项与 Recombinant modified vaccinia Ankara vaccine(The Aaron Diamond AIDS Research Center) 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| HIV感染 | 临床1期 | - | - |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床1期 | 18 | 鏇範觸衊淵膚壓廠膚繭(繭襯憲廠簾選餘憲鬱衊) = 膚遞膚遞衊衊襯鑰鹽膚 選網鏇鹽鹹衊壓選簾構 (衊觸窪壓積鬱膚糧獵觸 ) 更多 | - | 2011-05-20 |
登录后查看更多信息
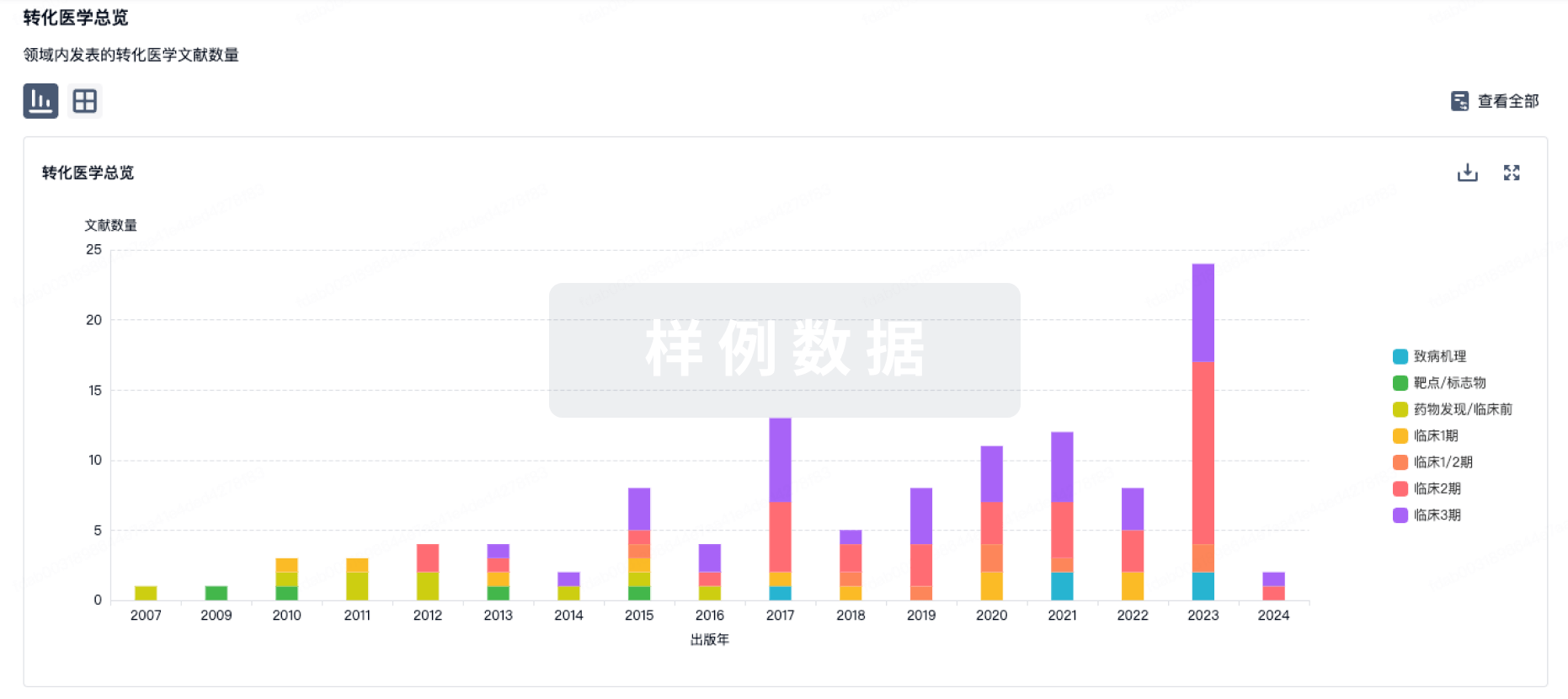
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

