1
项与 Autologous NY-ESO-1 CD4-TCR CD34+ HSC(Roswell Park Cancer Institute) 相关的临床试验A Phase I, Open Label Study Evaluating the Safety and Efficacy of Adoptive Transfer of Autologous NY-ESO-1 CD8-TCR Engineered T Cells and NY-ESO-1 CD4-TCR Engineered Hematopoietic Stem Cells (HSC) After a Myeloablative Conditioning Regimen, With Administration of IL-2 in Patients With Recurrent or Treatment Refractory Ovarian, Fallopian Tube or Primary Peritoneal Cancer
This phase I trial studies the best dose and side effects of NY-ESO-1 T cell receptor (TCR) engineered T cells and how well they work with NY-ESO-1 TCR engineered hematopoietic stem cells (HSCs) after melphalan conditioning regimen in treating patients with ovarian, fallopian tube, or primary peritoneal cancer that has come back (recurrent) or does not respond to treatment (refractory). The melphalan conditioning chemotherapy makes room in the patient's bone marrow for new blood cells and blood-forming cells (stem cells) to grow. Giving NY-ESO-1 TCR T cells and stem cells after the conditioning chemotherapy is intended to replace the immune system with new immune cells that have been redirected to attack and kill the cancer cells and thereby improve immune system function against cancer. Giving NY-ESO-1 TCR engineered T cells and HSCs after melphalan may work better in treating patients with ovarian, fallopian tube, or primary peritoneal cancer.
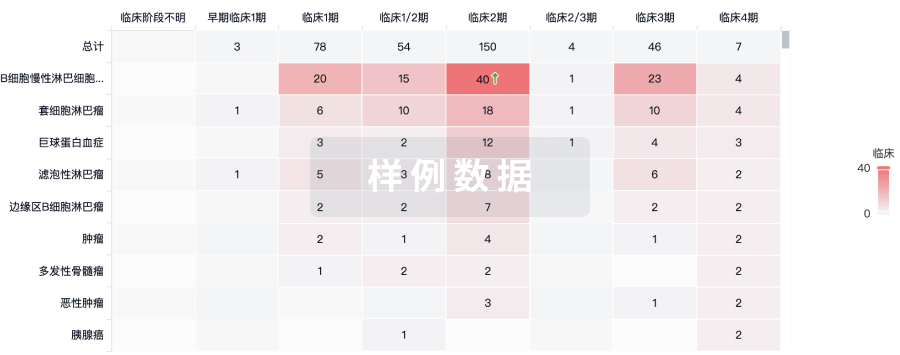
100 项与 Autologous NY-ESO-1 CD4-TCR CD34+ HSC(Roswell Park Cancer Institute) 相关的临床结果
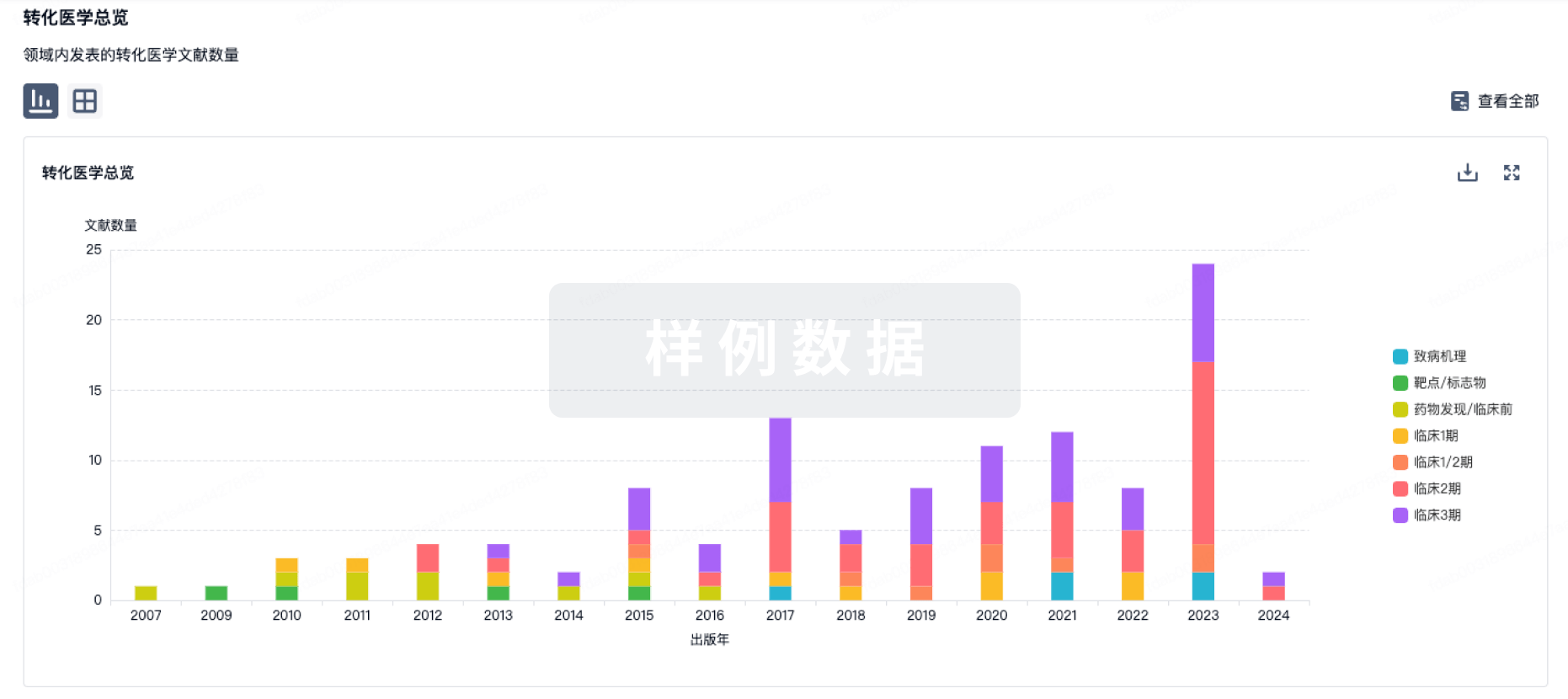
100 项与 Autologous NY-ESO-1 CD4-TCR CD34+ HSC(Roswell Park Cancer Institute) 相关的转化医学
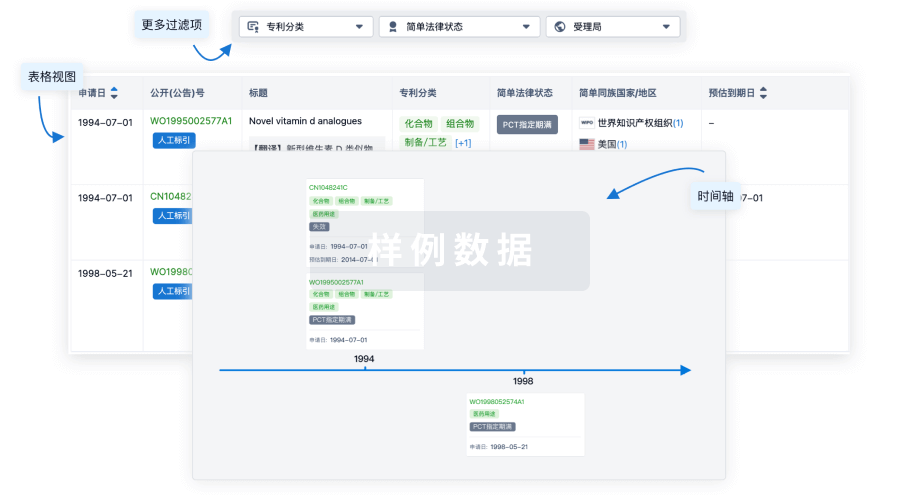
100 项与 Autologous NY-ESO-1 CD4-TCR CD34+ HSC(Roswell Park Cancer Institute) 相关的专利(医药)
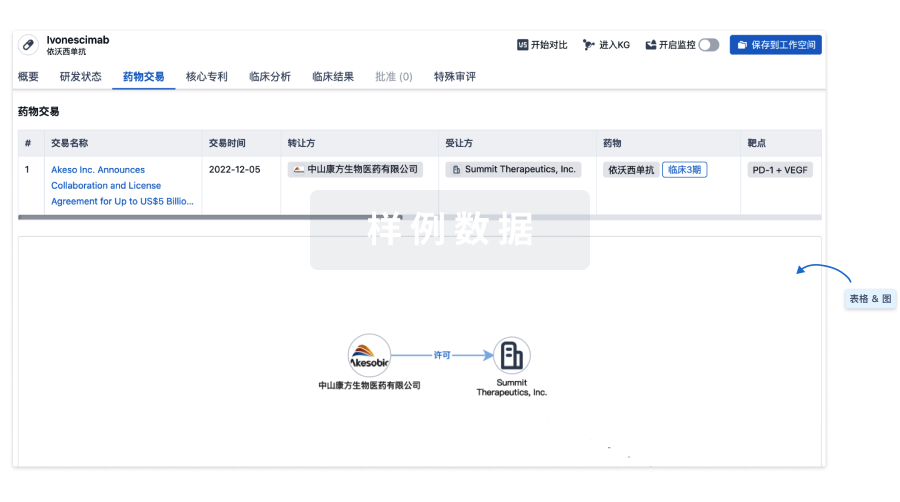
100 项与 Autologous NY-ESO-1 CD4-TCR CD34+ HSC(Roswell Park Cancer Institute) 相关的药物交易