更新于:2024-11-06
Dermatophagoides Farinae Drops(Wolwo Bio-Pharma)
粉尘螨滴剂 (我武生物)
更新于:2024-11-06
概要
基本信息
原研机构 |
在研机构 |
非在研机构- |
最高研发阶段批准上市 |
首次获批日期- |
最高研发阶段(中国)批准上市 |
特殊审评- |
登录后查看时间轴
关联
3
项与 粉尘螨滴剂 (我武生物) 相关的临床试验舌下含服“粉尘螨滴剂”治疗特应性皮炎的初步临床有效性及安全性 Ⅲ 期临床试验
申办方希望通过扩大人群进行多中心、随机、双盲、安慰剂平行对照的III期临床研究工作,进一步考察“粉尘螨滴剂”对粉尘螨过敏引起的成人特应性皮炎的临床疗效及安全性,评价利益与风险关系,最终为药物注册申请的审查提供充分的依据。
开始日期2016-02-25 |
申办/合作机构 |
舌下含服“粉尘螨滴剂”治疗特应性皮炎的初步临床有效性及安全性II期临床试验
通过多中心的临床研究,初步评价“粉尘螨滴剂”用于治疗特应性皮炎的临床疗效及安全性,并为III期临床试验研究设计和给药剂量方案的确定提供依据
开始日期2011-11-10 |
申办/合作机构 |
Usefulness and Safety of Sublingual Dermatophagoides Farinae Drops in Patients With Atopic Dermatitis:A Randomized,Double-blind,Placebo-controlled Study.
To assess the effect and safety of sublingual Dermatophagoides Farinae Drops in adult patients with atopic dermatitis and allergic sensitization to house dust mites.240 adults age 18 to 60 years with atopic dermatitis(Objective Scoring Atopic Dermatitis, Objective SCORAD from 10 to 40) are going to be enrolled in a randomized,double-blind,placebo-controlled study.Sublingual Dermatophagoides Farinae Drops or placebo is given for 9 months in addition to standard therapy.SCORAD,average anesis interval,rescue medicine and DLQI will be recorded.
开始日期2011-10-01 |
申办/合作机构  浙江大学 浙江大学 [+5] |
100 项与 粉尘螨滴剂 (我武生物) 相关的临床结果
登录后查看更多信息
100 项与 粉尘螨滴剂 (我武生物) 相关的转化医学
登录后查看更多信息
100 项与 粉尘螨滴剂 (我武生物) 相关的专利(医药)
登录后查看更多信息
3
项与 粉尘螨滴剂 (我武生物) 相关的文献(医药)2015-04-01·Clinical and experimental allergy : journal of the British Society for Allergy and Clinical Immunology2区 · 医学
The efficacy of sublingual immunotherapy with Dermatophagoides farinae vaccine in a murine atopic dermatitis model
2区 · 医学
Article
作者: Guo, D. ; Ding, S. ; Wu, B. ; Liang, Q. ; Zhang, L. ; Liu, L. ; Li, Q.
Summary:
Background:
Sublingual immunotherapy (SLIT) is a potential treatment for house dust mite‐induced atopic dermatitis (AD). However, the mechanisms of action are not clear.
Objective:
To establish a mouse model of AD in order to study the effect and mechanisms of SLIT.
Methods:
Mice were sensitized and subsequently challenged by repeated cutaneous application of Der f allergen extract. Sensitized mice were SLIT‐treated with a Der f vaccine and analyzed for AD symptoms, histopathological and immunological parameters. This experiment was carried in the Laboratory Animal Center, Shanghai University of TCM.
Results:
Repeated application of Der f extract caused rapid increase in dermatitis scores. Clinical findings (scratching behaviour, dermatitis and oedema) and histological symptoms (inflammatory cell infiltration) in this model were very similar to those in human AD. Serum total and Der f‐specific IgE and IgG levels, and the expression level of T helper (Th)2 cytokine IL‐5, IL‐13; and Th1 cytokine IL‐12 and IFN‐γ were elevated compared with the control mice. SLIT treatment of sensitized mice markedly reduced the clinical and histopathological symptoms and decreased both Th1 and Th2 cytokine levels.
Conclusions and Clinical Relevance:
The mouse model induced by Der f allergen extract reflected the typical hallmarks of human AD. In the Der f allergens‐sensitized mice, SLIT treatment with Der f vaccine was shown to significantly inhibit AD symptoms through correction of Th2 and Th1 cytokine predominance. Our study suggested that SLIT could be considered as an attractive treatment for patients with extrinsic AD.
2011-04-01·The Journal of allergy and clinical immunology1区 · 医学
House dust mite sublingual immunotherapy: Results of a US trial
1区 · 医学
Article
作者: Cheri Swenson ; William W. Busse ; Robert K. Bush ; Michael D. Evans ; Beth Fahlberg ; Robert Esch
BACKGROUND:
Few trials of sublingual immunotherapy (SLIT) in the United States have been reported.
OBJECTIVE:
This randomized, placebo-controlled feasibility SLIT study compared the safety and physiologic effects of high- versus low-dose Dermatophagoides farinae vaccine.
METHODS:
Thirty-one D farinae-sensitive adults with allergic rhinitis with or without mild intermittent asthma were eligible for randomization to high-dose maintenance vaccine (n = 10, 4200 allergen units [approximately 70 μg of Der f 1/d]), low-dose maintenance vaccine (n = 10; 60 allergen units [approximately 1 μg of Der f 1/d]), or placebo (n = 11) over 12 to 18 months. Medication-symptom scores and adverse events were monitored, serum D farinae-specific IgE and IgG4 levels were measured, and bronchial reactivity to D farinae was determined at baseline and 6-month intervals.
RESULTS:
Of the 31 randomized subjects, 6 withdrew because of non-treatment-ascribed events. Four withdrew because of treatment-ascribed effects: high-dose group, 1 of 10 (gastrointestinal symptoms); low-dose group, 1 of 10 (gastrointestinal symptoms); and placebo group, 2 of 11 (headache and increased nasal symptoms). Thus 21 subjects completed the study: high-dose group, 9; low-dose group, 7; and placebo group, 5. Eleven of the 21 subjects experienced mild-to-moderate gastrointestinal symptoms, throat irritation, or both (high-dose group, 5/9; low-dose group, 4/7; and placebo group, 2/5). No severe systemic reactions were noted. No differences in symptom-medication scores were found. High-dose SLIT increased the bronchial threshold to allergen challenge and increased serum D farinae-specific IgG4 levels, whereas low-dose SLIT and placebo had no significant effect.
CONCLUSIONS:
High-dose D farinae SLIT was generally tolerable, increased serum D farinae-specific IgG4 levels, and improved the bronchial threshold to allergen challenge. Larger US trials are warranted.
2004-04-01·The Journal of allergy and clinical immunology1区 · 医学
Effect of specific immunotherapy added to pharmacologic treatment and allergen avoidance in asthmatic patients allergic to house dust mite
1区 · 医学
Article
作者: Zanolla, Luisa ; Maestrelli, Piero ; Pozzan, Marcella ; Fabbri, Leonardo M.
BACKGROUND:
Although several studies support the efficacy of specific immunotherapy in allergic asthma, its benefit compared with that of standardized pharmacologic intervention remains unknown.
OBJECTIVE:
A double-blind, placebo-controlled trial in 72 patients with mild-to-moderate asthma and allergy to house dust mite (HDM; Dermatophagoides species) was conducted to assess the effects of specific immunotherapy added to guideline-adjusted pharmacologic treatment and allergen avoidance.
METHODS:
After 1 observational year of pharmacologic treatment and standard measures of HDM avoidance, 2 groups of asthmatic subjects were randomly assigned to receive specific immunotherapy consisting of subcutaneous injections of either a mixture of Dermatophagoides pteronyssinus and Dermatophagoides farinae vaccine (n=41) or placebo (n=31) for 3 years. Medications were adjusted every 3 months according to the Global Initiative for Asthma guidelines.
RESULTS:
The adjustment of treatment was associated with a reduction in asthma symptom scores in all subjects. The addition of specific immunotherapy was associated with a decrease in the number of subjects requiring rescue bronchodilators, an increase in morning and evening peak expiratory flow, and a reduced skin sensitivity to HDM extracts. The addition of specific immunotherapy had no significant effects on the cumulative dose of inhaled corticosteroids, asthma symptoms, lung volumes, or bronchial responsiveness to methacholine.
CONCLUSION:
These results suggest that specific immunotherapy added to pharmacologic treatment and HDM avoidance provides marginal but statistically significant clinical benefits, possibly by reducing the allergic response of asthmatic patients sensitized to HDM.
3
项与 粉尘螨滴剂 (我武生物) 相关的新闻(医药)2024-10-17
·梅斯医学
杭州的秋天属于桂花香,馥郁的香气飘散在整座城市。然而,虽然眼下桂花飘香,但对不少人而言,不仅快乐不起来,更是噩梦一般的存在。
“桂花开了以后门诊数量急剧上升”,杭州市中医院耳鼻喉科梅显伟副主任医师表示,半天门诊看了40位患者,一半以上为过敏性鼻炎,多是因为花粉过敏,以中青年居多。
过敏性鼻炎也称为变应性鼻炎(AR),是临床常见的慢性鼻病,因特应性个体暴露于过敏原(变应原)后主要由免疫球蛋白E(IgE)介导的鼻黏膜非感染性慢性炎性疾病,已成为主要的呼吸道慢性炎性疾病,影响着全世界10%~20%的人口。
AR 的发病与环境因素直接相关,不同地区的过敏原也有所不同。北京地区儿童常见吸入过敏原为尘螨、真菌、杂草花粉和动物毛等;西北和东北地区主要过敏原为杂草花粉;华东、华中 和 华 南地 区 过 敏 原 以 粉 尘 螨 和 屋 尘 螨 为主。近年来,饲养宠物导致儿童对猫毛和狗毛过敏以及某些食物过敏原与花粉过敏原之间存在交叉抗原性的问题也受到关注。AR 的遗传特征较为明显,父母罹患变应性疾病会增加儿童AR的发病风险。
临床分类
1、按过敏原种类分类
①季节性AR:常见过敏原为花粉、真菌等季节性吸入过敏原。
②常年性AR:常见过敏原为尘螨、蟑螂、动物皮屑等室内常年性吸入过敏原,以及某些职业性过敏原。
2、按症状发作时间分类
①间歇性AR:症状发作<4d/周,或<连续4周。
②持续性AR:症状发作≥4d/周,且≥连续4周。
3、按疾病严重程度程度分类
①轻度AR:症状轻微,对生活质量(包括睡眠、日常生活、工作学习)未产生明显影响。
②中-重度AR:症状较重或严重,对生活质量产生明显影响。
临床表现
1、症状
AR的典型症状为阵发性喷嚏、清水样涕、鼻痒和鼻塞;可伴有眼部症状,包括眼痒、流泪、眼红和灼热感等,多见于花粉过敏患者。随着致敏花粉飘散季节的到来,花粉症患者的鼻、眼症状发作或加重。如果致病因素以室内过敏原(尘螨、蟑螂、动物皮屑等)为主,症状多为常年发作。
40%的AR患者可合并支气管哮喘,在有鼻部症状的同时还可伴喘鸣、咳嗽、气急、胸闷等肺部症状。
2、体征
AR发作时最主要的体征是双侧鼻黏膜苍白、肿胀,下鼻甲水肿,鼻腔有多量水样分泌物。眼部体征主要为结膜充血、水肿,有时可见乳头样反应。伴有哮喘、湿疹或特应性皮炎的患者有相应的肺部、皮肤体征。
临床诊断
①症状:阵发性喷嚏、清水样涕、鼻痒和鼻塞等症状出现2个或以上,每天症状持续或累计在1h以上,可伴有流泪、眼痒和眼红等眼部症状;
②体征:常见鼻黏膜苍白、水肿,鼻腔水样分泌物;
③过敏原检测:至少1种过敏原SPT和/或血清特异性IgE阳性,或鼻激发试验阳性。AR的诊断应根据患者典型的过敏病史、临床表现以及与其一致的过敏原检测结果而作出。
过敏原检测通常需要将体内和体外检测相结合,且充分结合临床病史,以判断患者是由何种过敏原致敏,以及致敏的程度与疾病症状的关系。
鉴别诊断
1、急性鼻炎:亦称普通感冒,早期可有喷嚏、鼻塞、清水样涕等症状,流感引发的急性鼻炎多突然起病,常有咳嗽、流涕或鼻塞症状,其主要症状为发热,体温可达39~40℃,伴头痛、肌肉酸痛、乏力等全身症状。
2、非变应性鼻炎伴嗜酸粒细胞增多综合征:该病临床表现与AR难以区分,但症状较重,可伴嗅觉减退。其主要特征是高倍镜下鼻分泌物嗜酸粒细胞占比>20%,伴外周血嗜酸粒细胞增多,过敏原检测为阴性。
3、血管运动性鼻炎:其症状与儿童 AR 相似,主要包括鼻塞及清水样涕,少数患者亦有喷嚏及鼻痒。诱发因素包括温度湿度变化、刺激性气味及运动等。鼻腔检查鼻黏膜一般可呈充血或苍白,外周血和鼻分泌物嗜酸粒细胞数量正常,过敏原检测为阴性。
4、鼻窦炎:鼻窦炎可与 AR 有相似的症状,如鼻塞、流涕、咳嗽、头痛、嗅觉减退等,两者也可能合并存在。但鼻窦炎患儿鼻腔分泌物通常为黏脓性,可伴面部疼痛或压痛。鼻内镜检查及必要时的鼻窦CT检查有助于鼻窦炎的诊断。
5、脑脊液鼻漏:儿童脑脊液鼻漏多为先天性,临床症状多表现为单侧清水样涕,量可因头位变化而改变。鼻漏出液的糖定量或 β2 转铁蛋白检查有助于鉴别,必要时鼻内镜或影像学检查可帮助识别漏口位置。
6、抽动障碍:指突然、无目的、快速、刻板的肌肉收缩,临床症状多样,可表现为皱鼻、吸气、眨眼等,与部分AR患儿症状相似。抽动症状不由某些物质、药物或其他医疗事件引起,鼻腔检查及过敏原检测均为阴性。
7、其他疾病:儿童先天性后鼻孔闭锁、鼻腔狭窄、鼻腔异物、鼻中隔偏曲和腺样体肥大等疾病可引起鼻塞症状,也需要与AR进行鉴别。
治疗
AR的治疗原则为“防治结合,四位一体”,包括环境控制、药物治疗、免疫治疗和健康教育。
1、环境控制
避免或尽可能减少接触相关过敏原。以花粉为例,一天中花粉漂浮相对密集时间是 14∶ 00 ~16∶ 00,有些植物在 20∶ 00 ~ 22∶ 00 还会形成第 2 次花粉漂浮高峰。因此,尽量在花粉漂浮高峰时段减少外出,关闭门窗以避免或减少花粉进入室内。
2、药物治疗
AR常用治疗药物分为一线用药和二线用药。一线治疗药物包括鼻用糖皮质激素(简称鼻用激素)、第二代口服和鼻用抗组胺药、口服白三烯受体拮抗剂;二线治疗药物包括口服糖皮质激素、口服和鼻用肥大细胞膜稳定剂、鼻用减充血剂、鼻用抗胆碱能药。
其中,鼻用糖皮质激素可在鼻腔局部发挥抗炎、抗过敏和抗水肿特性,并具有免疫抑制作用,是变应性鼻炎治疗中最重要的一线用药。
第二代鼻用激素——丙酸氟替卡松,具有强效局部抗炎与抗过敏作用,可减轻鼻腔炎症水肿、渗出与毛细血管扩张,有效控制患者鼻痒、鼻塞与流涕等变应性症状;采用鼻腔局部给药,可显著提升鼻黏膜吸收率,药物浓度较高,起效较快,且安全性较高。
3、免疫治疗
免疫治疗是AR的一线治疗方法,目前临床常用的免疫治疗方法有皮下注射法(皮下免疫治疗)和舌下含服法(舌下免疫治疗),分为剂量递增和剂量维持两个阶段,总疗程为3年。
皮下免疫治疗通常在5岁以上的患者中开展,舌下免疫治疗可以放宽到3岁,具体需遵循药品说明书中的年龄规定。
(1)适应证:免疫治疗适用于过敏原特异性IgE介导的疾病,包括AR、变应性结膜炎和变应性哮喘。根据临床症状、体征、SPT和/或血清特异性IgE检测结果阳性确诊疾病者,适宜进行免疫治疗,在疾病初期即可开展,无需以药物治疗失败为前提。
免疫治疗尤其适用于以下患者:
①用常规药物治疗和过敏原回避等措施不能有效控制症状者;
②需要大剂量药物和/或多种药物联合使用方能控制症状者;
③药物治疗引起不良反应者;
④希望避免长期使用药物者;
⑤希望预防AR或哮喘发病者。
对出现严重不良反应、无法频繁至医院等不能进行皮下免疫治疗的患者,可考虑舌下免疫治疗。
(2)禁忌证:包括绝对禁忌证和相对禁忌证。
绝对禁忌证包括:
①未控制的或重症哮喘[第一秒用力呼气容积(FEV1)<70%预计值]和不可逆的呼吸道阻塞性病变;
②免疫性疾病活动期;
③恶性肿瘤。
相对禁忌证包括:
①哮喘得到部分控制;
②正在使用β受体阻滞剂或血管紧张素酶抑制剂;
③严重的心血管系统疾病,发生严重不良反应时可能增加使用肾上腺素的风险;
④自身免疫性疾病缓解期;
⑤严重的精神系统疾病或依从性差(包括不能理解治疗的风险和局限性)、经常不能按时返院就诊者;
⑥原发性或继发性免疫缺陷;
⑦免疫治疗过程中曾发生过严重不良反应;
⑧不推荐在妊娠期或计划妊娠期开始免疫治疗,在维持治疗阶段出现妊娠的,应充分告知患者风险并征得同意,再决定是否继续治疗,发生不良反应时应终止免疫治疗;⑨几种特殊情况:花粉过敏引起的季节性AR患者在花粉播散期禁止开始免疫治疗;急性感染、发热或接种其他疫苗(如新型冠状病毒疫苗)等情况下,应推迟或暂停免疫治疗,必要时调整剂量;皮下免疫治疗注射治疗当天应避免剧烈运动和饮酒,口腔溃疡或外伤时不应给予舌下免疫治疗。相对禁忌证在评估获益大于风险时可以谨慎使用,在治疗期间应严密观测。
(3)皮下免疫治疗
根据剂量累加阶段注射频率的不同,皮下免疫治疗可分为常规免疫治疗和加速免疫治疗,后者又可分为集群免疫治疗和冲击免疫治疗。目前国内临床应用较多的是常规免疫治疗和集群免疫治疗。
出现下列情况之一时,应考虑推迟免疫治疗:
①1周内有发热或急性呼吸道感染病史;
②近期肺功能显著下降;
③注射前有过敏反应发作;
④特应性皮炎或湿疹发作期;
⑤最近接触过较多过敏原;
⑥1周内注射了其他疫苗;
⑦正在使用β受体阻滞剂或血管紧张素酶抑制剂;
⑧3~4 d内有全身性的并发疾病及哮喘发作。
不良反应:
(4)舌下免疫治疗
国内目前可供临床使用的舌下含服标准化过敏原疫苗有粉尘螨滴剂和黄花蒿花粉滴剂。与皮下免疫治疗相比,舌下免疫治疗操作相对简便,具有无创性、耐受性和安全性好等特点,但需要每天给药,治疗的依从性有待通过患者教育等各种策略进行提高。
来源 | 中国变应性鼻炎诊断和治疗指南(2022版)
撰文|Anna.AN
编辑 | Swagpp
● 哀悼!近期两名顶级科主任突然离世!享年均为59岁!15岁考上大学、天才医生、风湿免疫专家、糖尿病防治大咖!我们无比惋惜悲痛
● 每一口外卖、瓶装水都影响生殖健康!柳叶刀子刊:微塑料恐侵入并损伤男性生殖系统,尤其是不粘锅涂层
● 震撼!超百家医院、诊所、药店主动退出医保!国家医保局通报无锡骗保案,举报有奖!“上网比药价”实施,留给药店的时间不多了
版权说明:梅斯医学(MedSci)是国内领先的医学科研与学术服务平台,致力于医疗质量的改进,为临床实践提供智慧、精准的决策支持,让医生与患者受益。欢迎个人转发至朋友圈,谢绝媒体或机构未经授权以任何形式转载至其他平台。
点击下方「阅读原文」 立刻下载梅斯医学APP!
2024-07-06
我武生物,一家稀缺的“三好”药企——业绩、风险、投资回报率,全都跑赢全球标杆公司,但在投资人买单这件事上却输得很惨。
资深分析师| 苏叶
图文编辑| 静静
最敏锐的投资者会知道,2021年2月,才是A股医药下行的起点。根据同花顺数据统计,有三分之二的公司从此开启长跌之路。曾经的香饽饽转眼成“烫手山芋”,我武生物尤为突出。从跌速上,第一波出货药企他排前15%;从幅度上,跌幅榜他排前5%。
尽管这期间,我武生物市值跌去四分之三,约300亿。但谁也不会否认,我武生物仍是国内脱敏治疗的龙头,且是唯一的上市龙头。业务稀缺性上,与恒瑞、兴齐眼药这样的龙头比,有过之无不及。
财务指标上,我武生物一直也表现不俗。自2014年上市来,我武生物收入年复合增长率(CAGR)超过15%,归母净利润CAGR约13.5%,投资人回报率维持在15%-25%。
那么问题来了,我武生物从巅峰到低谷,是被高估还是被错杀?他的底价会在哪?带着问题,CM10医药研究中心将从行业稀缺值、财务健康度、业务健康度、综合建议,来一一回答。
小而美脱敏赛道:独狼屹立
过敏不是小赛道。根据沙利文数据,2023年,中国仅过敏性鼻炎、哮喘、特应性皮炎、慢性阻塞性肺疾病四类基数最大的过敏疾病,对应的患者群体就超过了5亿。该机构预计,抗过敏药全球市场总规模将在2024年达到630亿美元,其中中国82亿美元(约合600亿元),五年CAGR超过13%。
脱敏是过敏赛道里的吃力不讨好型。现行的过敏治疗主要有三种:对症治疗、脱敏治疗、回避过敏原。
对症治疗“治标”,见效快,但不持久,一直是药物研发的主流——从抗组胺药物(如氯雷他定、西替利嗪)、白三烯(如孟鲁司特)、糖皮质激素,到小分子(如JAK抑制剂乌帕替尼),再到生物靶向药(如度普利尤、奥马珠),迭出重磅大单品。在国内,氯雷他定2023年销售额达16亿元,度普利尤单抗2023年达30亿元。
脱敏治疗,有百年历史,以“治本”为标签,见效慢,治疗周期长达3-5年,从全球来看都是小众赛道。在全球,玩家只有ALK、Allergo pharma和Stellargenes Greer等企业;在国内,相当长时间里只有我武生物,品种基本上只有尘螨、黄花蒿变应原滴剂等较为成熟的低致敏原性制剂。
综合来看,对症治疗占了过敏赛道的主流市场,其中以“疗效更优、安全性高、重症及耐药患者起效明显”为特点的生物制剂,如度普利尤,来势汹汹,唯一阻碍是价格。据开源证券报告,2023年过敏领域里生物制剂的渗透率已经超过了15%,到2030年或将达到50%。如今该领域是过敏赛道最卷的方向,包括IL-4R、TSLP、IL-33等均有恒瑞、正大天晴等强企参与。而脱敏治疗,由于多方面因素,耗时长、价格高等,在国内发展了十多年渗透率仍不足1%,短期内受困因素并未解除。
中国脱敏治疗赛道的独狼。我武生物的差异在于,一是给药方式,他目前是国内唯一做舌下脱敏的;二是治疗费用相对更友好,粉尘螨滴剂(商品名:畅迪)3年治疗费用约为6638元,而竞品(屋尘螨变应原制剂、螨变应原注射液)需要8400-10360元,由于治疗全自费,所以粉尘螨滴剂价格优势相对明显)。截至2023年报披露日,我武生物已上市产品有7个(如下),其中畅迪和畅晧为治疗产品。
数据来源:公司年报
A股稀缺龙头,跑赢全球标杆公司。自上市以来,至6月13日,我武生物股价累计上涨412.85%,跑赢全球标杆ALK(对应期间300%)。跑赢,除了在A股略显珍贵外,也有业绩数据支撑,比如上市9年(2014-2023年)营收CAGR超过15%,ALK为7.9%;净利润CAGR为13.5%,ALK为11.6%;ROE更是跑赢。
要注意,两家公司并不在同一发展阶段,在基数上有差别,可以通过看ALK来看我武生物的未来。ALK公司是全球脱敏领域的先驱和标杆,成立于1923年,开发了全球首款治疗牧草花粉过敏的舌下过敏免疫治疗片剂(SLIT片剂),之后又推出了治疗豚草和尘螨过敏的SLIT片剂。目前已经投入市场的产品有牧草花粉、豚草花粉、屋尘螨、日本柳杉花粉、桦木科树花粉五大产品,2023年收入达到48.24亿丹麦克朗(约合50亿元),覆盖了新型舌下AIT(过敏原特异性免疫治疗) 约40%的市场份额,大约240万患者。过去五年间,片剂销售年复合增长29%。
增长困境:必须自己打败自己
从行业的维度里,很难想象,我武生物为何会走向这样的“沉沦”。成也萧何败也萧何。上市的9年里,我武生物的“成败”有两大推力:一是机构投资者,二是畅迪。
机构投资者涌入推动估值泡沫产生。上市期间无论从盈利能力,还是从风险指标上,我武生物都是一只无可指摘的股票。也正因此,我武生物从上市开启长牛轨道。
数据来源:同花顺Find
2016年走出A股“大震荡”后,二级市场的机构开始重构投资逻辑。彼时,我武生物因业绩平平,加上上市仅3年,仍是一只“隐形股”。
直至2017年,我武生物在净利润低基数下创造了44%的同比增长,加上解禁原因,持股的机构一下子翻倍不止,自此被机构投资者盯上。
2018年股价回调后,又迎来新一轮基金重仓,直至2020年。在机构投资者的推动下,我武生物一年半时间股价上涨超200%,迎来市值接近500亿元的巅峰。彼时它的净利润仍不足3亿元,PE高达168倍。
后来的结果也证明了估值泡沫的存在。从2021年开始,我武生物率先走进资本“寒冬”的估值调整期。
市场总在过度乐观和过度悲观后,迎来反转时刻。在反转来临前,必须要认知到我武生物自身的诸多无奈。
无奈一,畅迪进入生命周期后期,面临自己打败自己。与众多药企相同,我武生物所面临的新旧产品迭代危机已经无法掩盖。畅迪是我武生物最核心的收入来源,长期以来营收贡献比例不低于98%。2023年畅迪销售额约为8.21亿元,首次出现同比下滑。值得重视的是,畅迪的增速已经连续放缓,增速的高峰为2018年。
更让人无奈的是,比起大多产品,或受集采冲击,或被竞品蚕食,畅迪放缓的核心是市场的天花板。畅迪早在2006年便在国内获批上市,国内竞品仅有ALK的安脱达、Allergo的阿罗格,但只有畅迪的给药方式为舌下含服,其余为皮下注射。所以,从2012年起,畅迪作为市场一哥,一路卖到超过80%的天花板份额。而ALK 又在近期宣布,决定撤回其屋尘螨舌下片剂在中国的生物制品许可申请(BLA)。也就是说,接下来,我武生物要自己打败自己,销售冲破10亿(在国内,10亿是判断大单品的一个门槛),才有望迎来重估。今年第一季度2.16亿元的营收开了个好头。
数据来源:同花顺Find
无奈二,接棒产品畅皓“远水”解不了近渴。作为我武生物重注的接力产品,黄花蒿花粉变应原舌下滴剂(商品名:畅皓)被寄予众望——
市场空间大。据东吴证券研报,2021年国内过敏性鼻炎患者人数超2.4亿,蒿属花粉点刺阳性率高达28.6%,潜在人群达0.63亿。
竞争格局好。与畅迪相比,畅皓的市场环境更好,是国内唯一获批的黄花蒿脱敏治疗产品,完全没有竞品。
可观的定价。畅皓的年化治疗费用,是畅迪的6倍,约为12000元。
畅皓上市后迅速放量,但短期贡献占比仍小。2021年6月,畅皓在国内获批上市,年底畅皓销售额367万元;2022年,销售额831万元;2023年,销售额1531万元,销量约5.7万支,收入贡献占比不足2%。另一方面,我武生物在北方没有销售积累,在国内尘螨过敏原主要在南方,而花粉主要在北方,有知情人称“在北方开拓市场困难”。
无奈三,“躺平”多年销售人效亟待提升。我武生物在销售上有三大痛点:一是医院拓展,目前九百多人的团队覆盖了大约1000家医院,还有近2万家医院等待开拓;二是销售人效,2023年人均创收不足50万元,对比来看,兴齐眼药74万,恒瑞医药116万。三是营销压力,销售费率创历史新高约为38%,过去五年都低于35%,未来几年压力不会减。压力主要来自新产品推广支出,同时老产品陷入增长瓶颈。
总体来说由于过度依赖单一产品,我武生物的归母净利润增速早已步入下滑通道。进一步,ROE(投资回报率)也从2019年起进入下跌通道,2023年成上市以来的最低点,不足15%,不过比起很多公司并不算低。
数据来源:同花顺Find
变数一,核心高管退居“幕后”。6月13日,我武生物公告,创始人胡赓熙因个人原因辞去总经理职务,原任期到2025年12月27日,胡赓熙将继续担任董事长及董事会专门委员会相关委员职务。胡赓熙一直是我武的灵魂人物,提前退居或增添不确定性。据新浪证券,两年前胡赓熙因个人资金需求,曾累计减持1045.2万股,套现金额5亿元,不过后来提前终止了减持计划。
估值空间:在哪?
我武生物还有空间吗?答案是肯定的。但空间需要他打开天花板,才能走出成长性,给到新的估值空间。
CM10医药研究中心认为有三大抓手:
第一,聚焦并扩大现有产品组合的覆盖范围,包括畅迪在地域上的横向覆盖,畅皓的新市场覆盖,适应证有针对性地扩展到新群体。在销售拓展上我武生物有很大拓展空间,已经毋庸赘述。新群体建议往儿科拓展,一方面,儿科领域市场空间大,全球约有4-5亿人受过敏性鼻炎困扰,其中40%为儿童;另一方面,从脱敏治疗的特点来说,儿童市场的需求和依从性都好于成人。据了解,我武生物的黄花蒿花粉变应原舌下滴剂扩展儿童适应证人群补充申请已于2023年5月获批。
我武生物先前也提出聚焦主业的发展策略,将继续巩固在现有过敏性疾病诊疗及延伸产品群中的优势,继续在该领域中发展相关及互补产品群:包括脱敏治疗产品及过敏诊断产品,不断完善“诊断+治疗”、“对因+对症”的过敏性疾病诊疗解决方案,确保公司在该领域的持续竞争优势,2023年审批上市的药品争取培养为爆款。
第二,创新合理延伸,最大限度地发挥现有核心产品的价值,并将产品组合多样化。尽管尘螨和黄花蒿花粉是中国需求量最大的两种过敏原,能够满足大多患者的需求,但从品类上标准化脱敏制剂仍然缺乏。我武生物在临床反应和过敏领域深耕多年,研发创新可往相邻疾病领域拓展,例如食物过敏、过敏反应等。我武生物在研管线中有望于近期上市的,还有葎草花粉点刺液、烟曲霉点刺液、德国小蠊点刺液、猫毛皮屑点刺液、悬铃木花粉点刺液等产品。
我武生物拓展新领域的现有策略是进入市场潜力巨大、且可能建立明确技术优势的领域,力争在下一个十年内,在2-3个医药领域形成新的产品集群。该公司已进入干细胞治疗药物、天然药物(抗耐药抗生素)这两个领域,正在推进抗耐药结核药物、干细胞治疗药物等产品的临床前研究工作。
不可否认,两个新领域的市场空间。但从科学维度上,新领域与我武生物先前所在的脱敏治疗赛道方向、机制完全不同,可谓任重道远。另一方面,我武生物过往的经历平顺无虞,欠缺成功的革命基因,加上大跨度的变革,又给其未来蒙上一层纱。
“ 任何伟大的变革都是痛苦的。我们已经见证了,诸多仿制药企走向创新药企历经的黑暗与迷茫岁月。我们已经看见了,一些蜕变,和新价值的诞生。CM10医药研究中心致力于上市公司的价值发现、挖掘与诊断。”
免责声明:本文章是基于上市公司的公众公司属性、以上市公司公开披露的信息(包括但不限于临时公告、定期报告和官方互动平台等)为核心依据的独立第三方研究;CM10医药研究中心力求报告(文章)所载内容及观点客观公正,但不保证其准确性、完整性、及时性等:本报告(文章)中的信息或所表述的意见不构成任何投资建议,CM10医药研究中心不对因使用本报告(文章)所采取的任何行动承担任何责任。
回复“逆商”,了解电子期刊详情
精彩推荐
CM10 | 集采 | 国谈 | 医保动态 | 药审 | 人才 | 薪资 | 榜单 | CAR-T | PD-1 | mRNA | 单抗 | 商业化 | 国际化 | 猎药人系列专题 | 出海
启思会 | 声音·责任 | 创百汇 | E药经理人理事会 | 微解药直播 | 大国新药 | 营销硬观点 | 投资人去哪儿 | 分析师看赛道 | 药事每周谈 | 医药界·E药经理人 | 中国医药手册
创新100强榜单 | 恒瑞 | 中国生物制药 | 百济 | 石药 | 信达 | 君实 | 复宏汉霖 |翰森 | 康方生物 | 上海医药 | 和黄医药 | 东阳光药 | 荣昌 | 亚盛医药 | 齐鲁制药 | 康宁杰瑞 | 贝达药业 | 微芯生物 | 复星医药 |再鼎医药|亚虹医药
跨国药企50强榜单 | 辉瑞 | 艾伯维 | 诺华 | 强生 | 罗氏 | BMS | 默克 | 赛诺菲 | AZ | GSK | 武田 | 吉利德科学 | 礼来 | 安进 | 诺和诺德 | 拜耳 | 莫德纳 | BI | 晖致 | 再生元
财报
2023-05-16
·医药笔记
创业传奇,与IgE靶点的不解之缘1986年,一家名为Tanox的生物技术公司在美国休斯顿成立,正是这家起初名不见经传的华人生物技术公司诞生了日后的全球重磅新药,第一代抗IgE抗体奥马珠单抗(Xolair®,Omalizumab)。Tanox是华人生物制药领域的先行者和开拓者,创造过多个第一和唯一:成功开发了第一也是唯一上市的抗IgE抗体奥马珠单抗,华人开发的第一个年销售金额超过30亿美元的抗体药,第一也是唯一被Genentech公司收购的企业。Tanox联合创始人是两对在生物医药领域颇具传奇的华人夫妇,孙乃超、周若芸夫妇和张子文、唐南栅夫妇。在Tanox期间,孙乃超博士与夫人周若芸主要负责药物的开发工作,是奥马珠单抗的主要发明人。开发靶向IgE抗体的主意最初便是由他们提出,也正是从那一刻开始,孙乃超博士与IgE靶点和抗IgE抗体结下了不解之缘。左图:IgE在多种过敏疾病中扮演核心作用 右图:B淋巴细胞结合IgE免疫球蛋白的一个独特抗原结合表位通过专利查询,可以找到一篇1994年发表,题为“Antigenic
epitopes on B cell-bound IgE Immunoglobulins”的欧洲专利,其中孙乃超、周若芸夫妇和张子文位列发明人一栏。该专利对B淋巴细胞结合IgE免疫球蛋白的一个独特抗原结合表位(ige.bl)进行了保护。据介绍,该表位为外源性支气管哮喘,变应性鼻炎,花粉热以及食物和药物过敏在内的多种由IgE介导的过敏性疾病的治疗和诊断提供了依据,对抗IgE抗体的研发与发展有着十分深远的影响。目前,奥马珠单抗由罗氏与诺华进行全球推广(罗氏负责美国市场,诺华负责美国外市场),已经于2017年8月在我国获批上市,商品名茁乐。财报显示,奥马珠单抗2022年度销售额达到36.8亿美元,位列2022全球药品销售额TOP100第41位,上市以来累计销售额超过300亿美元,是一款名副其实的“超级重磅炸弹”。在Tanox期间,孙乃超博士已致力于第二代抗IgE抗体的研发。2007年Tanox被Genentech收购,2009年Genentech被Roche收购,由于Roche和Novartis千丝万缕的关系,Tanox/Genentech的某些项目由Novartis 承接,并继续开发,其中就包括第二代抗IgE抗体/Ligelizumab。在一项治疗难治性CSU患者的头对头IIb期临床研究中,Ligelizumab显示出优于奥马珠单抗的疗效,临床结果发表在顶级医学杂志《新英格兰医学杂志》上。目前处于III 期临床试验中,被Novartis作为潜在“10亿美金的重磅候选药物”重点开发。续写传奇,打造IgE靶点下一代“重磅药物”2020年,孙乃超博士联合刘恒博士创建天辰生物(原龙行生物),并基于其三十多年在IgE靶点的技术积累,再度开发了全新一代抗IgE 抗体(LP-003),比第一代和第二代IgE抗体有更高的生物学活性,更高的亲和力,更长的半衰期,并最大限度的去除了其副作用。根据NMPA药物临床试验登记与信息公示平台信息,LP-003目前已进入II期临床试验(登记号#CTR20231145)LP-003选择过敏性鼻炎为首个适应症,颇具策略性和看点。首先过敏性鼻炎是典型的由IgE介导的过敏性疾病,其次奥马珠单抗的过敏性鼻炎适应症在日本已经获批,验证了IgE靶点在这个适应症上的可行性。但由于奥马珠单抗在中国尚未申请此适应症,其它在研生物制剂中仅有康诺亚的IL-4Ra抗体申报过此适应症(尚未查到临床开展的信息),LP-003有望成为国内首个用于过敏性鼻炎的生物制剂,竞争格局相对较好。天辰生物的LP-003 同时具有高生物学活性,高亲和力,和较长的半衰期,可以在保证有效性/疗效的基础上,覆盖完整的过敏季,同时降低剂量和不良反应,大幅度提高患者的依从性,具有较强的差异化和竞争优势。过敏性鼻炎分为季节性过敏性鼻炎和常年性过敏性鼻炎。季节性过敏性鼻炎发病率较高、症状发作呈季节性,多在春秋两季发生,又以秋季为多。常见过敏原为花粉、真菌等季节性吸入过敏原。常年性过敏性鼻炎症状发作呈常年性,常见过敏原为尘螨、蟑螂、动物皮屑等室内常年性吸入过敏原,以及某些职业性过敏原。针对常年性过敏性鼻炎,目前主流的治疗手段为脱敏治疗:其代表公司为我武生物,和丹麦的ALK。左图为我武生物粉尘螨滴剂(畅迪),右图为ALK屋尘螨变应原制剂(安脱达)我武生物(SZ300357)月线走势图(截至2023年5月11日,公司市值224.2亿元人民币)我武生物是中国医药市场中一个非常成功的细分领域的隐形冠军,在二级市场表现也是可圈可点。然而由于脱敏治疗本身的作用特点(剂量、药效和副作用),且脱敏的周期较长(平均治疗周期1-3年),导致其依从性有较大的提高空间(约40%-70%)。因此国际和国内变态反应的权威专家都在探讨包括抗IgE抗体在内的药物与脱敏治疗联合用药。在去年12月举行的中华医学会2022年全国变态反应学术大会上,中华医学会变态反应分会副主任委员兼秘书长,中国医师协会变态反应医师分会副会长,北京协和医院变态反应科支玉香教授在主会场发表了题为“过敏原特异性免疫治疗的更新迭代”的专题报告,提及抗IgE抗体与脱敏治疗的联合用药带来重要的临床意义,会上还有其他专家分享了“变应性鼻炎的超级冲击免疫治疗”等抗IgE 抗体与脱敏治疗联用的临床实践经验,可望解决目前脱敏治疗未满足的临床需求,进一步扩展脱敏市场应用与商业价值。中国的过敏性鼻炎患者人群基数大(患病率17.6%,约2亿人),目前除了传统的抗组胺药物,鼻喷激素和白三稀受体拮抗剂以外,并无太多其他有效治疗手段,且未有获批的生物制品(脱敏治疗除外),有调查表明现有治疗手段仍不能控制疾病症状的患者占25%-35%,因此存在着巨大的未满足的临床需求。2022年10月,《抗IgE单抗治疗变应性鼻炎专家共识(2022,南昌)》发布,进一步证实了临床专家对该治疗方式的认可。新一代抗IgE抗体LP-003作为国内过敏性鼻炎进展最快的生物制剂,可望填补这一临床空白。孙乃超博士作为抗IgE抗体领域的开山鼻祖,对IgE靶点和第一代及第二代抗IgE抗体具有独到且深刻的理解。根据天辰生物公开资料,LP-003是孙乃超博士基于过敏性疾病临床上的特点和未满足的临床需求,再次开发的一款活性更高,用量更低,且半衰期更长的新一代抗IgE抗体。我们非常期待LP-003在临床上的表现,并希望孙乃超博士带领天辰生物续写其在过敏领域的传奇故事,给中国的生物医药发展写下浓墨重彩的一笔。参考资料:[1] 张燕影等. 变应性鼻炎患者皮下免疫治疗和舌下免疫治疗的临床依从性对比及分析[J]. 国际耳鼻咽喉头颈外科杂志, 2022, 46(4):5.[2] Wang XD, Zheng M, Lou HF, Wang CS,
Zhang Y, Bo MY, et al. An increased prevalence of self-reported allergic
rhinitis in major Chinese cities from 2005 to 2011. Allergy 2016;71:1170-80[3] 中华医学会2022年全国变态反应学术大会[4] https://www.sohu.com/a/536450935_121118803孩子得了过敏性鼻炎怎么办?耳鼻喉科专家为您支招Armstrong技术全梳理系列GPRC5D靶点全梳理;CD40靶点全梳理;CD47靶点全梳理;补体靶向药物技术全梳理;补体药物:眼科治疗的重要方向;Claudin 6靶点全梳理;Claudin 18.2靶点全梳理;靶点冷暖,行业自知;中国大分子新药研发格局;被炮轰的“me too”;佐剂百年史;胰岛素百年传奇;CUSBEA:风雨四十载;中国新药研发的焦虑;中国生物医药企业的研发竞争;中国双抗竞争格局;中国ADC竞争格局;中国双抗技术全梳理;中国ADC技术全梳理;Ambrx技术全梳理;Vir Biotech技术全梳理;Immune-Onc技术全梳理;科济药业技术全梳理;恺佧生物技术全梳理;同宜医药技术全梳理;百奥赛图技术全梳理;腾盛博药技术全梳理;创胜集团技术全梳理;永泰生物技术全梳理;中国抗体技术全梳理;德琪医药技术全梳理;德琪医药技术全梳理2.0;和铂医药技术全梳理;荣昌生物技术全梳理;再鼎医药技术全梳理;药明生物技术全梳理;恒瑞医药技术全梳理;豪森药业技术全梳理;正大天晴技术全梳理;吉凯基因技术全梳理;基石药业技术全梳理;百济神州技术全梳理;百济神州技术全梳理第2版;信达生物技术全梳理;信达生物技术全梳理第2版;中山康方技术全梳理;复宏汉霖技术全梳理;先声药业技术全梳理;君实生物技术全梳理;嘉和生物技术全梳理;志道生物技术全梳理;道尔生物技术全梳理;尚健生物技术全梳理;康宁杰瑞技术全梳理;科望医药技术全梳理;科望医药技术全梳理2.0;岸迈生物技术全梳理;礼进生物技术全梳理;康桥资本技术全梳理;余国良的抗体药布局;荃信生物技术全梳理;安源医药技术全梳理;三生国健技术全梳理;仁会生物技术全梳理;乐普生物技术全梳理;同润生物技术全梳理;宜明昂科技术全梳理;派格生物技术全梳理;迈威生物技术全梳理;Momenta技术全梳理;NGM技术全梳理;普米斯生物技术全梳理;普米斯生物技术全梳理2.0;三叶草生物技术全梳理;贝达药业抗体药全梳理;泽璟制药抗体药全梳理;恒瑞医药抗体药全梳理;齐鲁制药抗体药全梳理;石药集团抗体药全梳理;豪森药业抗体药全梳理;华海药业抗体药全梳理;科伦药业抗体药全梳理;百奥泰技术全梳理;凡恩世技术全梳理。
并购临床3期财报
100 项与 粉尘螨滴剂 (我武生物) 相关的药物交易
登录后查看更多信息
外链
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| - | - | - |
研发状态
批准上市
10 条最早获批的记录, 后查看更多信息
登录
| 适应症 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|
| 超敏反应 | 中国 | - |
未上市
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 特应性皮炎 | 临床3期 | 中国 | 2016-02-25 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
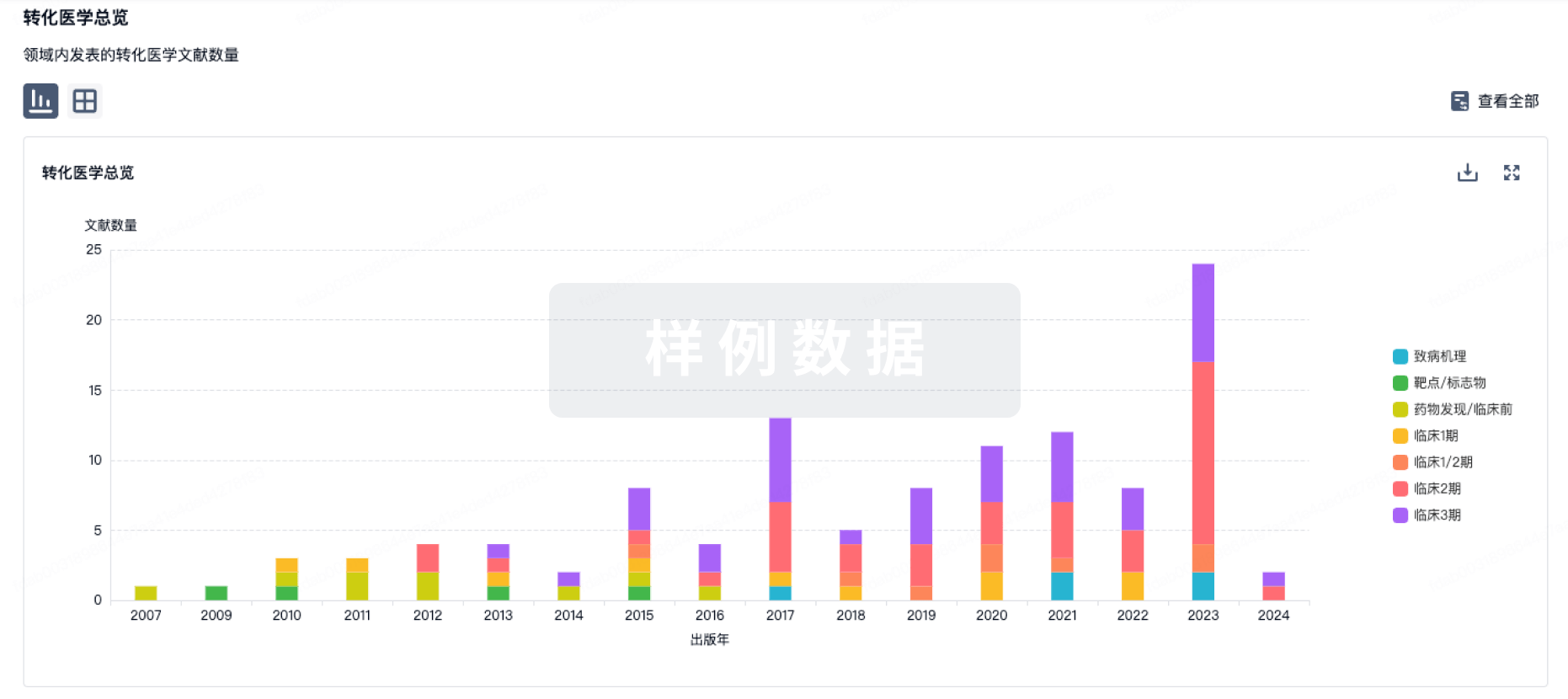
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


