预约演示
更新于:2025-05-07
Muscular Dystrophy Congenital, Merosin Negative
Merosin 缺陷性先天性肌营养不良症
更新于:2025-05-07
基本信息
别名 CMD1A、CMD1A - congenital muscular dystrophy type 1A、Congenital muscular dystrophy due to laminin alpha2 deficiency + [17] |
简介 An autosomal recessive inherited congenital muscular dystrophy caused by mutations in the LAMA2 gene. It is characterized by severe hypotonia, muscle weakness, elevated levels of serum creatinine kinase, and white matter abnormalities. |
关联
1
项与 Merosin 缺陷性先天性肌营养不良症 相关的药物靶点- |
作用机制- |
在研机构 |
原研机构 |
在研适应症 |
非在研适应症- |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
10
项与 Merosin 缺陷性先天性肌营养不良症 相关的临床试验NCT06503367
Establishing Clinical Trial Readiness in Patients Age 0-5 Years with LAMA2-related Congenital Muscular Dystrophy (READY CMD LAMA2)
The goal of this observational study is to understand how young children with LAMA2-related dystrophy move and change over time. We will also learn about how this condition impacts other body systems.
Participants will undergo:
* Neuromuscular assessments
* Blood collections
* Swallowing and breathing assessments
* Questionnaires
Participants will undergo:
* Neuromuscular assessments
* Blood collections
* Swallowing and breathing assessments
* Questionnaires
开始日期2025-03-01 |
申办/合作机构 |
NCT06354790
A Prospective, Longitudinal, Interventional Natural History Study of Children With LAMA2-related Dystrophies
The goal of this natural history study is to characterize the disease course, characteristics in paediatric population of LAMA2-RD (related dystrophies) patients.
The aim of the study is to establish a well-described cohort of patients in France with LAMA2-RD for prospective follow-up and recruitment for future clinical trials.
Participants will be follow up during a two years period regarding exhaustive aspects of the pathology:
* Muscular function
* Respiratory function
* Cognitive phenotyping
* Quality of life
* Growth parameters
* Biomarkers
The aim of the study is to establish a well-described cohort of patients in France with LAMA2-RD for prospective follow-up and recruitment for future clinical trials.
Participants will be follow up during a two years period regarding exhaustive aspects of the pathology:
* Muscular function
* Respiratory function
* Cognitive phenotyping
* Quality of life
* Growth parameters
* Biomarkers
开始日期2024-12-05 |
申办/合作机构 |
NCT06924125
Spanish Natural History Study for LAMA2 Muscular Dystrophy
The objective of this natural history study is to comprehensively characterize the disease progression and clinical features of LAMA2-related dystrophies (LAMA2-RD) in the pediatric population. The study aims to establish a well-defined cohort of patients in Spain, enabling long-term follow-up and facilitating recruitment for future clinical trials.
开始日期2021-07-27 |
申办/合作机构- |
100 项与 Merosin 缺陷性先天性肌营养不良症 相关的临床结果
登录后查看更多信息
100 项与 Merosin 缺陷性先天性肌营养不良症 相关的转化医学
登录后查看更多信息
0 项与 Merosin 缺陷性先天性肌营养不良症 相关的专利(医药)
登录后查看更多信息
403
项与 Merosin 缺陷性先天性肌营养不良症 相关的文献(医药)2025-01-01·International Orthopaedics
Systemic inhibition of bone morphogenetic protein 1.3 as a possible treatment for laminin-related congenital muscular dystrophy
Review
作者: Matic Jelic, Ivona ; Pecina, Marko ; Vukicevic, Slobodan ; Bordukalo Niksic, Tatjana ; Ivanjko, Natalia ; Stokovic, Nikola ; Kufner, Vera
2024-12-01·Life Science Alliance
Laminin-α2 chain deficiency in skeletal muscle causes dysregulation of multiple cellular mechanisms
Article
作者: Pita, Mafalda ; Zilhão, Rita ; Antonini, Dario ; Murtinheira, Fernanda ; Herrera, Federico ; Carlos, Ana Rita ; Paulino-Cavaco, Cláudia ; Rodrigues, Gabriela ; Thorsteinsdóttir, Sólveig ; Dayalan Naidu, Sharadha ; Melo, Catarina ; Ribeiro, Vanessa ; Fernandes, Diogo R ; Gameiro dos Santos, Pedro ; Fonseca, Inês ; Dinkova-Kostova, Albena T ; Saget, Bérénice ; Martins, Susana G
2024-12-01·Neuromuscular Disorders
Acute weakness and elevated creatine kinase levels associated with coxsackievirus infection in LAMA2-related muscular dystrophy
Article
作者: Webster, Richard ; Wong, Wui-Kwan ; Warner, Denise
7
项与 Merosin 缺陷性先天性肌营养不良症 相关的新闻(医药)2024-10-28
LAMA2-CMD is characterised by the absence of the LAMA2 protein, leading to severe, early-onset muscular dystrophy. Credit: Ines Nepo/Shutterstock.
The US Food and Drug Administration (FDA) has granted orphan drug designation (ODD) to Modalis Therapeutics’ investigational therapy, MDL-101, aimed at treating congenital muscular dystrophy type 1A (LAMA2-CMD).
An epigenetic editing therapy, MDL-101 includes a guide nucleotide targeting the LAMA1 gene — a close homolog of the disease-causing LAMA2 gene. It utilises an enzyme-null Cas9 (dCas9) fused with a trans-activating domain, driven by a muscle-specific promoter and delivered via a muscle-specific AAV [adeno-associated virus] vector.
MDL-101 works by upregulating LAMA1 gene products in patients’ muscle tissue to compensate for loss of function resulting from LAMA2 mutations.
This approach has the potential to offer a one-time, long-lasting treatment option for individuals with LAMA2-CMD.
ODD status facilitates the development of treatments for conditions affecting fewer than 200,000 patients in the US.
See Also:
UAE’s clinical trials space matures with experience from Covid-19
FDA approves Iterum’s ORLYNVAH NDA for uUTI treatment
Under the ODD status for MDL-101, Modalis will receive benefits including exemption from application fees on filing new drug applications, tax reductions for clinical development and preferential treatment for the development and promotion of the asset in the US.
On receipt of regulatory approval, MDL-101 would also receive seven years of exclusive sales rights in the US market.
The US regulator previously granted
rare paediatric disease designation
for gene therapy for patients with LAMA2-CMD.
LAMA2-CMD is characterised by the absence of the LAMA2 protein, a critical component comprising more than 3,000 amino acids.
This leads to severe, early-onset muscular dystrophy, and due to the protein’s size, conventional gene therapy approaches such as using an AAV vector are challenging.
Modalis focuses on the development of precision genetic therapies leveraging its epigenome editing technology, CRISPR-GNDM. This technology enables modulation of gene expression without causing double-strand DNA breaks.
Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva
.
Editorial content is independently produced and follows the
highest standards
of journalistic integrity. Topic sponsors are not involved in the creation of editorial content.
Free Whitepaper
Cell and gene therapies: Pipe dream to pipeline
The cell and gene industry is gaining momentum, with a new wave of therapies promising to transform the way doctors treat, and even cure, disease. In this report, Cytiva and GlobalData have collaborated to explore the rise of the cell and gene therapy industries, the current state of the market, present and future opportunities for advancement, and the challenges that lie ahead.
Thank you.
You will receive an email shortly. Please check your inbox to download the Whitepaper.
By Cytiva Thematic
By downloading this Whitepaper, you acknowledge that GlobalData may share your information with
Cytiva Thematic
and that your personal data will be used as described in their
Privacy Policy

孤儿药基因疗法寡核苷酸临床研究上市批准
2024-09-30
LAMA2-CMD is an early onset congenital muscular dystrophy caused by the absence of the LAMA2 protein. Image credit: Shutterstock/ Dan76.
Modalis Therapeutics has been awarded a rare paediatric disease designation by the US Food and Drug Administration (FDA) for its congenital muscular dystrophy type 1a (LAMA2-CMD) gene therapy.
As per the designation, the Japan-based company will be eligible to receive a priority review voucher if the drug candidate, MDL-101, is approved by the FDA.
Recipients can
redeem priority review vouchers
themselves for any chosen drug or instead sell them to other pharma companies. Having the ability to slash four months off FDA review time, vouchers can sell for about $100m.
The designation comes after a period of uncertainty for the vouchers’ future, with the programme facing a tight deadline on sunset provisions. The US House of Representatives ultimately passed a bill last week to reauthorise the rare paediatric priority review voucher program through September 2029.
Modalis said it also pursuing orphan drug status for its candidate, with the FDA currently reviewing the company’s application. Orphan drug status provides benefits such as tax credits for US-based clinical trials and market exclusivity.
See Also:
FDA approves PureTech’s KarXT to treat schizophrenia in adults
FDA greenlights Regeneron’s Dupixent for the treatment of COPD
LAMA2-CMD is an early-onset congenital muscular dystrophy caused by the absence of the LAMA2 protein. Affected infants can have severe muscle weakness, hypotonia, and joint deformities, amongst others. As those with the disease grow, spinal curvature and speech issues can also develop. There are no approved therapies to treat LAMA2-CMD.
Gene therapies for LAMA2-CMD using an adeno-associated virus (AAV) vector are difficult to develop as LAMA2 is made up of 3,000 amino acids, meaning it cannot be loaded onto a vector, according to Modalis.
The company is therefore using its clustered regularly interspaced short palindromic repeats (CRISPR)-based epigenome editing technology to develop the therapy. MDL-101 works by inducing the expression of the sister gene LAMA1 in muscle tissues, which compensates for the deficient function of LAMA2.
Modalis’s CEO Haru Morita said: “We have received many requests for our efforts from children and families around the world suffering from this disease for which there is currently no treatment, and we feel a mission to respond to the expectations of patients who are eagerly awaiting the start of clinical trials as soon as possible.”
Modalis expects to file an investigational new drug (IND) application in 2025, according to a Q2 2024
financial results
press release.

基因疗法孤儿药
2024-03-04
BOSTON, March 4, 2024 /PRNewswire/ -- Constant Therapeutics LLC, a biopharmaceutical company focused on the development of treatments impacting the Alternative Renin-Angiotensin System, today announced that the first patient was dosed March 3, 2024, in the Company's phase 2 clinical trial of TXA127, the Company's lead peptide product under development as a potential treatment for Duchenne Muscular Dystrophy-associated (DMD-associated) Cardiomyopathy.
The phase 2 trial is an open-label, multi-center trial designed to evaluate the safety and efficacy in non-ambulatory patients with DMD-associated Cardiomyopathy who are 16 years and older and who are receiving systemic glucocorticoids. Patients will receive 6 months of treatment and have the option to enter a 12-month extension study. The study is being conducted in Israel at Sheba Medical Center and Hadassah Medical Center and is expected to enroll 10 patients.
"This is a significant milestone for Constant and for the DMD community," said Rick Franklin, CEO of Constant Therapeutics. "There are few treatments for DMD patients with cardiomyopathy, and heart failure is a leading cause of death in this disease. We are excited to initiate this carefully designed trial, and we look forward to sharing the data with the DMD community upon completion of the trial."
About Constant Therapeutics LLC
Constant Therapeutics LLC is a private biopharmaceutical company focused on the development of treatments that effect the Alternative Renin-Angiotensin System. The lead compound, TXA127, is a pharmaceutical formulation of the naturally occurring peptide Angiotensin (1-7) and is being developed for the treatment of stroke recovery and DMD-associated Cardiomyopathy. For more information on Constant Therapeutics, please visit our website at .
About TXA127
TXA127 is a pharmaceutical formulation of the naturally occurring human peptide angiotensin-(1‐7). In addition to its specific effects in cardiac dysfunction, TXA127 has shown therapeutic activity in animal models of chronic stroke, Duchenne Muscular Dystrophy (DMD), Limb‐Girdle Muscular Dystrophy (LGMD), Congenital Muscular Dystrophy (MDC1A), Marfan Syndrome and Epidermolysis Bullosa.
Contact:
[email protected]
+1-617-245-0289
SOURCE Constant Therapeutics
临床2期
分析
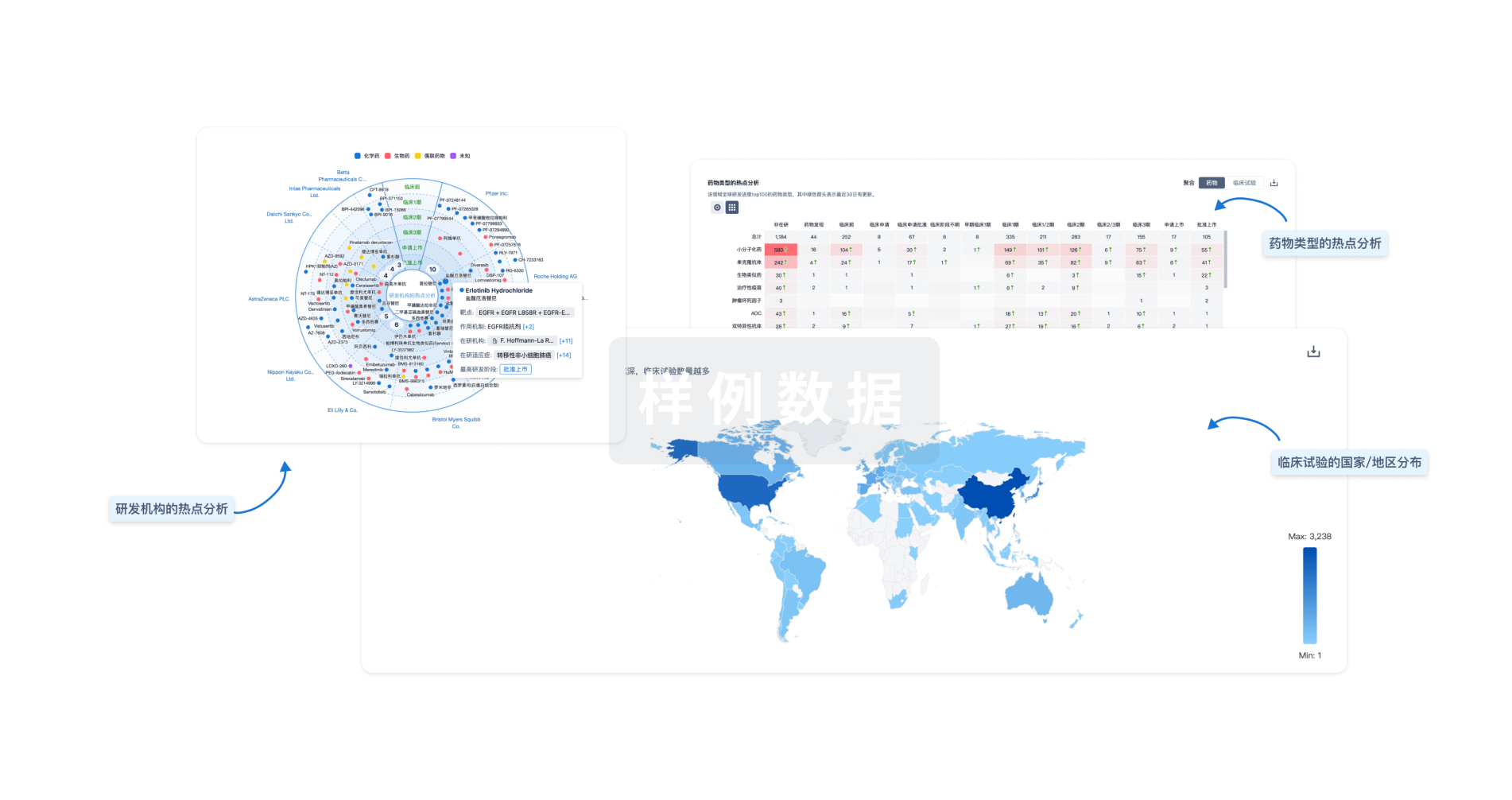
对领域进行一次全面的分析。
登录
或
Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

