更新于:2024-09-19
PROX1
更新于:2024-09-19
基本信息
别名 Homeobox prospero-like protein PROX1、prospero homeobox 1、Prospero homeobox protein 1 + [2] |
简介 Transcription factor involved in developmental processes such as cell fate determination, gene transcriptional regulation and progenitor cell regulation in a number of organs. Plays a critical role in embryonic development and functions as a key regulatory protein in neurogenesis and the development of the heart, eye lens, liver, pancreas and the lymphatic system. Involved in the regulation of the circadian rhythm. Represses: transcription of the retinoid-related orphan receptor RORG, transcriptional activator activity of RORA and RORG and the expression of RORA/G-target genes including core clock components: BMAL1, NPAS2 and CRY1 and metabolic genes: AVPR1A and ELOVL3. |
关联
100 项与 PROX1 相关的临床结果
登录后查看更多信息
100 项与 PROX1 相关的转化医学
登录后查看更多信息
0 项与 PROX1 相关的专利(医药)
登录后查看更多信息
1,174
项与 PROX1 相关的文献(医药)2106-07-05·Oncotarget
Estrogen receptor beta reduces colon cancer metastasis through a novel miR-205 - PROX1 mechanism
Article
作者: Nguyen-Vu, Trang ; Mesmar, Fahmi ; Saxena, Ashish ; Gustafsson, Jan-Åke ; Wang, Jun ; Mukhopadhyay, Srijita ; McCollum, Catherine W. ; Bondesson, Maria ; Williams, Cecilia
2024-09-01·Arteriosclerosis, Thrombosis, and Vascular Biology
FOXC1 and FOXC2 Ablation Causes Abnormal Valvular Endothelial Cell Junctions and Lymphatic Vessel Formation in Myxomatous Mitral Valve Degeneration
Article
作者: Tan, Can ; Kurup, Shreya ; Dyakiv, Yaryna ; Kume, Tsutomu ; Muller, William A ; Ge, Zhi-Dong ; Muller, William A. ; Liu, Ting
2024-08-06·Human Molecular Genetics
Lymphatic endothelial cell-specific NRAS p.Q61R mutant embryos show abnormal lymphatic vessel morphogenesis
Article
作者: Ohnishi, Hidenori ; Niihori, Tetsuya ; Abe, Taiki ; Ozeki, Michio ; Aoki, Yoko ; Nozawa, Akifumi
4
项与 PROX1 相关的新闻(医药)2023-11-28
·今日头条
本文为转化医学网原创,转载请注明出处
作者:Jerry
导读:
肿瘤异质性及其驱动因素影响肿瘤进展和癌症治疗。单细胞RNA测序用于研究肿瘤生态系统的异质性。
近日,浙江大学郭国骥、梁廷波等研究人员在权威期刊《Advanced Science》上发表了题为“Pan-Cancer Single-Nucleus Total RNA Sequencing Using snHH-Seq”的研究论文,本研究中,
研究人员开发了一种高通量、高灵敏度的方法snHH-seq,该方法将随机引物与液滴微流控平台的预索引策略相结合。
这种创新的方法允许从临床冷冻样品中检测单个细胞核中的总RNA。并且建立了一个强大的流水线,以促进全长RNA-seq数据的分析。snHH-seq应用于来自32例不同肿瘤类型患者的73万多个单核。泛癌症研究使其能够全面分析肿瘤转录组数据,包括表达水平、突变、剪接模式、克隆动态等。
https://onlinelibrary-wiley-com.libproxy1.nus.edu.sg/doi/10.1002/advs.202304755
研究背景
01
癌细胞及其驱动因素的异质性在肿瘤发生和恶性进展中起着重要作用,并对癌症治疗有重要影响。单细胞RNA测序(scRNA-seq)能够在单细胞水平上测量转录信息,从而准确地解决肿瘤异质性。
许多研究利用scRNA-seq来阐明肿瘤微环境的多样性,揭示治疗策略的机制,并据此提出新的分子标记和治疗靶点。
然而,scRNA-seq在处理临床肿瘤样本,特别是档案材料(如冷冻组织)方面具有独特的后勤和技术挑战。首先,临床肿瘤样本的scRNA-seq需要实施快速组织解离程序,目前大多数医院的常规病理实验室不存在这种程序。其次,大多数scRNA-seq方法使用oligo-dT引物来捕获和扩增聚腺苷化转录本,这一过程高度依赖于组织样品的质量。此外,寡聚dt捕获导致缺乏对许多生物过程很重要的非聚腺苷化转录本。第三,高通量scRNA-seq方法只能检测转录本3 '或5 '端的短片段,这限制了临床样本的突变和剪接分析,特别是对肿瘤。
为了克服这些挑战,研究人员开发了“高通量高灵敏度单核总RNA测序”(snHH-seq),将随机引物和预索引策略结合在液滴微流控平台上。单核RNA-seq (snRNA-seq)减少了对样品收集和处理的要求。这也为纵向样本的分析铺平了道路。使用随机引物而不是oligo-dT引物捕获转录本,不仅可以拯救部分降解转录本的档案临床样本,而且具有更高的灵敏度(一个转录本有多个可捕获的位点)和更高的覆盖率(转录本的3 '和5 '端,非聚腺苷化转录本和聚腺苷化转录本)。该预索引策略应用于基于液滴和基于平板的方法,提供至少一个数量级的吞吐量增益,并促进临床样品的多路分析。
研究进展
02
为了能够评估大量临床冷冻样本,减少解离的批次,研究人员建立了snHH-seq,一个基于液滴的高通量、高灵敏度的单核总RNA测序平台。snHH-seq是一种结合随机RT引物和预索引策略优势的强大方法。
snHH-seq具有高通量和高灵敏度,为肿瘤图谱绘制和其他基因组研究提供了新的可能性。
接下来,研究人员使用snHH-seq分析冷冻临床肿瘤样本。研究人员分析了32例患者的735 722个细胞核,涵盖了中国发病率和死亡率最高的肿瘤类型,包括肺腺癌(LUAD)、肝细胞癌(HCC)、肝内胆管癌(ICC)、胶质瘤、结肠腺癌(COAD)、直肠腺癌(READ)、乳腺浸润性癌(BRCA)、食管癌(ESCA)和胃腺癌(STAD)。在单细胞癌症研究中,识别恶性细胞是至关重要的一步,因为它们构成了癌症样本的主要组成部分。为了在研究人员的数据集中识别细胞类型(恶性细胞和非恶性细胞),研究人员使用了标记基因和推断拷贝数变异(CNV)的组合。考虑到所研究的癌症类型,研究人员使用带注释的非上皮细胞,如内皮细胞、基质细胞和巨噬细胞作为intercnv的参考,以推断每个患者的CNV特征。
为了进一步说明TME(肿瘤微环境)的异质性,研究人员收集了来自不同患者的内皮细胞、骨髓细胞和基质细胞,并进行了亚聚类分析。研究人员获得了15个肿瘤内皮细胞(TEC)亚簇,包括动脉TEC (C4、FBLN5和SULF1)、静脉TEC (C6、ACKR1)、淋巴TEC (C2、PROX1)、毛细血管TEC (C7、BTNL9和CD36)、外周血TEC (C9、PDGFRB)、尖端TEC (C3、COL4A1)、细胞周期TEC (C12、C2orf48和CIT)和免疫样TEC (C10、DOCK2和PTPRC)。Tip TEC (C3)和cell cycle TEC (C12)在多个肿瘤中富集。在泛癌髓系细胞中,研究人员观察到肿瘤和NAT之间共享和特异性的基因特征。此外,研究人员在乳腺肿瘤和NAT样本中观察到显著的异质性。C2和C5主要驻留在乳腺肿瘤中,表达抑癌基因,可能调控乳腺TME中硫酸肝素的表达;C13主要存在于乳腺NAT和高表达的LRP1B中,LRP1B是一种可能抑制肿瘤迁移和侵袭的肿瘤抑制因子。表达C8的TNXB主要存在于NAT中,对多种肿瘤有贡献,可能阻碍肿瘤细胞的侵袭和转移。
总的来说,研究人员对细胞区室的泛癌症分析揭示了肿瘤环境的共享和癌症限制特征。
使用snHH-seq进行泛癌分析
研究结论
03
总之,研究人员证明了高分辨率单细胞全长转录组测序在鉴定新的肿瘤生物标志物方面的价值,并为全长单细胞测序提供了详细的分析。研究人员的综合分析方法包括检查基因表达、体细胞突变、剪接模式和克隆行为。通过利用这些不同的观点,研究人员可以确定与癌症相关基因和细胞类型相关的特定变异。这个项目代表了研究人员理解癌症潜在遗传机制的能力的重大进步。最终,这些发现加深了研究人员对肿瘤生物学的理解,并为更有效的诊断和治疗方法铺平了道路。
参考资料:
https://onlinelibrary-wiley-com.libproxy1.nus.edu.sg/doi/10.1002/advs.202304755
注:本文旨在介绍医学研究进展,不能作为治疗方案参考。如需获得健康指导,请至正规医院就诊。
热门·直播/活动
🕓 北京|12月19日-20日
▶第四届单细胞测序技术应用研讨会暨单细胞&空间组学研讨会(日程稍后公布)
点击对应文字 查看详情
基因疗法核酸药物
2023-01-11
·生物谷
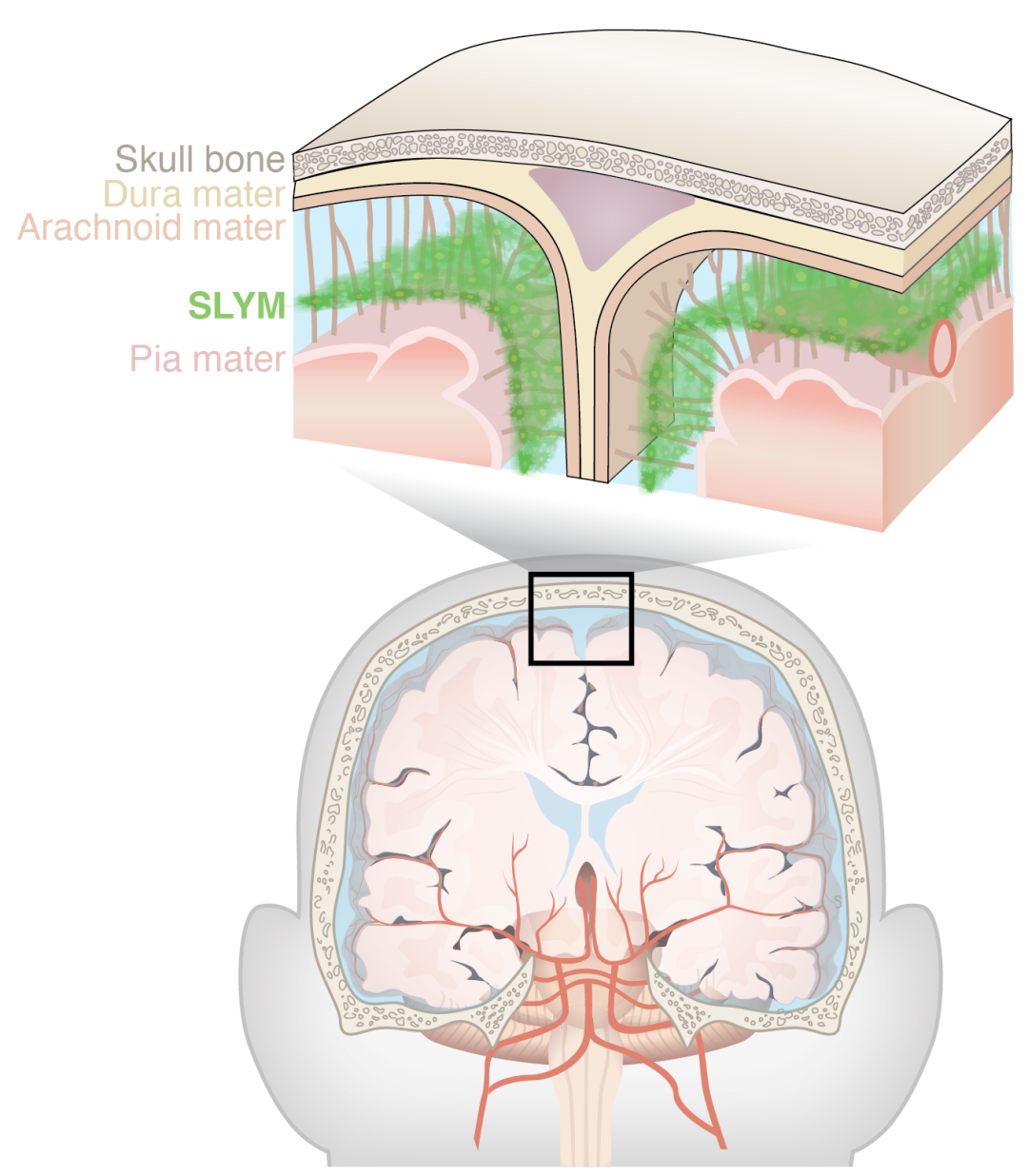
该研究首次发现了大脑表面存在不同于硬脑膜、蛛网膜和软脑膜的第4层膜结构——SLYM,这将更新教科书中的解剖学基础知识。
大脑表面自外向内具有3层脑膜结构——硬脑膜、蛛网膜和软脑膜,这是每个医学生学习大脑解剖的基础知识。
然而,这一基础知识或许到了该更新的时候了。
近日,来自罗切斯特大学医学中心的Maiken Nedergaard团队和哥本哈根大学的Kjeld Møllgård团队,在小鼠和人类大脑中发现了第4层脑膜结构,并将其命名为蛛网膜下淋巴管样膜(SLYM),相关研究成果发表于《科学》杂志[1]。
SLYM位于蛛网膜与软脑膜之间,将蛛网膜下腔分为浅层和深层,并包裹着蛛网膜下腔内的血管,其形态和免疫表型类似于外周器官表面的间皮层。在功能上,SLYM不仅可起到润滑作用,在运动过程中减少大脑和头骨之间的摩擦,还可分隔并控制脑脊液在大脑表面的流动和物质的交换。
SLYM的解剖位置
此外,SLYM对大脑的免疫功能也起到重要作用,完整的SLYM可阻止外周免疫细胞随意进入脑内。同时,SLYM上有着自己的免疫细胞群体,通过与脑脊液的接触,监控脑内是否有炎症或感染的迹象。
总的来说,SLYM的发现对于进一步理解脑内代谢物质的清除和免疫功能具有十分重要的意义。
论文首页截图
人类对大脑的探索始于对解剖的认识。
随着研究技术的进步,过去许多关于大脑的固有认知都被一一打破,如原本认为大脑内不存在淋巴系统,但Maiken Nedergaard团队在2012年发现的类淋巴系统[2](也叫做胶质淋巴通路),以及2015年脑膜淋巴管[3]的发现颠覆了这一认知。
该研究则来源于Kjeld Møllgård团队的一个猜想,即在外周脏器表面存在的间皮层(包裹并保护脏器,同时含有免疫细胞)在大脑中是否存在[4]?
为了更深入地分析脑组织外的膜结构,Møllgård等使用双光子显微镜观测了Prox1-EGFP+(Prox1:一种决定淋巴管发育的转录因子)小鼠体感皮层的脑膜。同时,研究人员使用二次谐波对未标记的胶原纤维进行可视化,并分别使用Cascade Blue偶联葡聚糖和磺基罗丹明101来标记血管和星形胶质细胞,以区分脑组织外的各层结构。
在硬脑膜纤维束下方,他们发现存在一层与松散排列的胶原纤维混合在一起的单层连续扁平Prox1-EGFP+细胞。这层细胞将蛛网膜下腔分为浅层和深层,并覆盖蛛网膜下腔血管,其厚度为14.2±0.5μm,比硬脑膜要薄(21.8±1.3μm)。研究人员将这层膜命名为SLYM。
小鼠体内双光子成像显示脑表面存在第4层膜
紧接着问题来了,SLYM对物质的通透性如何呢?这关系到膜两侧物质的交换。
为了测试这一点,Møllgård和他的同事将与红色荧光基团结合的直径1μm微球注射到Prox1-EGFP+小鼠蛛网膜下腔的浅层,同时将与蓝色荧光基团结合的直径1μm微球注射到蛛网膜下腔内深层,然后使用双光子显微镜进行观察。结果显示,红色微球被限制在浅层,而蓝色微球则被限制在深层,表明SLYM对直径1μm的微球不具有通透性。
但脑脊液中的许多溶质,如细胞因子和生长因子,大小都远远小于1μm。因此,研究人员将更小的示踪剂TMR-葡聚糖(3 kDa)注射到Prox1-EGFP+小鼠蛛网膜下腔的深层,结果显示TMR-葡聚糖仍然无法透过SLYM。然而,在硬脑膜损伤和脑脊液漏的小鼠中,SLYM的两侧均可观察到注射的示踪剂。
基于以上研究结果,Møllgård团队认为SLYM将蛛网膜下腔从物理上分成了浅层和深层,并可限制了大多数多肽和蛋白质在蛛网膜下腔浅层和深层之间的交换。
SLYM可限制了大多数多肽和蛋白质在蛛网膜下腔浅层和深层之间的交换
接下来,Møllgård团队对SLYM的免疫表型进行了表征,发现SLYM中另一个淋巴管标志PDPN表达呈阳性,而淋巴管内皮细胞受体1(LYVE1)表达呈阴性。此外,SLYM中细胞维甲酸结合蛋白2(CRABP2)表达呈阳性。与SLYM不同,硬脑膜上的淋巴管表达所有的经典淋巴标志物(Prox1-EGFP+、PDPN+、LYVE1+和VEGFR3+,但CRABP2-)。
值得注意的是,对成人大脑皮质的分析表明,在软脑膜上方的蛛网膜下腔同样存在CRABP2+/PDPN+的膜结构。因此,SLYM在人类的大脑中同样存在。
SLYM在人类的大脑中同样存在
为了进一步证明SLYM与蛛网膜是不同的膜结构,研究人员进行了CLDN-11免疫染色,CLDN-11是形成蛛网膜屏障细胞层(ABCL)紧密连接的主要成分。染色结果显示,SLYM中CLDN-11表达阴性,而CLDN-11在ABCL和脉络丛基质细胞中均有密集表达,表明SLYM与蛛网膜是不同的两层。同时,SLYM与软脑膜的免疫标记特征同样存在差异。
由此研究人员得出结论,SLYM为环绕小鼠和人类大脑的第4层脑膜结构,在免疫表型上有别于硬脑膜、蛛网膜和软脑膜,并显示出淋巴样特征(Prox1-EGFP+,PDPN+,LYVE1-,CRABP2+,VEGFR3-,CLDN-11-和E-Cad-)。有意思的是,SLYM与外周器官表面的间皮层具有相同的特征。因此,SLYM可能就是大脑的间皮层,起到减少运动时脑组织与颅骨之间摩擦的作用。
SLYM在免疫表型上有别于硬脑膜、蛛网膜和软脑膜
那SLYM还有其他功能吗?答案是肯定的。
研究人员在Prox1-EGFP+小鼠的大脑切片中发现,Prox1-EGFP+SLYM细胞经常与静脉窦内皮细胞直接接触并形成紧密连接,且这种紧密结合是可渗透的,允许小分子在血液和脑脊液之间进行交换。
此外,SLYM还起到免疫屏障的作用,可阻止外周的免疫细胞直接进入深层的脑脊液中,同时SLYM中常驻着免疫细胞。而在脑部出现炎症或脑部退行性改变后,SLYM中的免疫细胞明显增多,LYVE1+、CD206+和CD68+巨噬细胞和CD11c+树突状细胞会在SLYM上聚集,表明SLYM具有免疫监视的作用。
SLYM具有监测脑脊液是否有感染和炎症迹象的功能
总的来说,该研究首次发现了大脑表面存在不同于硬脑膜、蛛网膜和软脑膜的第4层膜结构——SLYM,这将更新教科书中的解剖学基础知识。
SLYM控制着脑脊液和血液物质的交换,同时具有免疫屏障和免疫监视功能,与中枢神经系统感染和阿尔茨海默病等各种疾病均具有密切关联,值得进一步深入研究。
参考文献
1.Mollgard K, Beinlich FRM, Kusk P, Miyakoshi LM, Delle C, Pla V, Hauglund NL, Esmail T, Rasmussen MK, Gomolka RS et al: A mesothelium divides the subarachnoid space into functional compartments. Science 2023, 379(6627):84-88.
2.Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, Benveniste H, Vates GE, Deane R, Goldman SA et al: A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Sci Transl Med 2012, 4(147):147ra111.
3.Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, Derecki NC, Castle D, Mandell JW, Lee KS et al: Structural and functional features of central nervous system lymphatic vessels. Nature 2015, 523(7560):337-341.
4.https://www.urmc.rochester.edu/news/story/newly-discovered-anatomy-shields-and-monitors-brain
2022-09-23
·生物谷
该研究通过机器学习的方法,在单细胞分辨率下构建了首个人海马区imGCs在整个生命周期内的动态分子特征图谱,证明了成年后海马中仍然存在imGCs,具有发展为成熟的新颗粒细胞的能力。
关于大脑神经元能否再生,一直是神经科学领域十分关注的问题。
传统的观点认为大脑中的神经元在成年后难以再生,但也有研究表明在大脑的某些区域,如海马,即使在成年后,仍然具有一定的产生新神经元的功能[1]。
目前的理论认为,在成年后海马的神经发生过程中,神经干细胞首先产生中间神经前体细胞和神经母细胞,然后分裂为未成熟的齿状回颗粒细胞(imGCs,接受大脑皮层信息输入的神经元),并逐渐变为新的成熟神经元[2]。
然而由于海马未成熟神经元的标志物有限,且研究方法较为复杂,因此科学家们对其知之甚少。
近期,来自宾夕法尼亚大学佩雷尔曼医学院的宋洪军和明国莉的研究团队,通过单核RNA测序(snRNA-seq)和基于机器学习的方法,对人一生中不同阶段的海马imGCs进行分析,构建了首个人全生命周期imGCs的分子特征,相关研究发表于《自然》杂志[3]。
他们发现,在人的整个生命周期中,海马中均存在imGCs,婴儿期imGCs占到颗粒细胞总数量的9.4%,4岁以后则为3.1%~7.5%,并随着年龄的增长这一比例逐渐下降。与成熟颗粒细胞(mGCs)相比,imGCs的基因表达特征与神经发生及可塑性相关。同时,手术获得的人成年海马组织在体外培养后能够获得大量的imGCs,这些结果都支持成年大脑具有神经再生的功能。
此外,研究人员还注意到,在阿尔茨海默病(AD)患者中,imGCs数量减少,且基因表达改变,表明了imGCs在AD中具有一定的研究价值。
论文首页截图
研究人员首先对4个婴儿(这一时期imGCs丰度较高[4])的海马样本进行了snRNA-seq分析,并根据未成熟神经元标记DCX、颗粒细胞标记PROX1和CALB1(CALB1在某些imGCs表达,但在mGCs中表达更高)对细胞进行分类。
他们发现传统的非监督聚类方法无法在snRNA-seq数据集中识别imGCs群体(在以PROX1为标志的颗粒细胞中,DCX+imGCs与其他细胞混合在一起,无法进一步分离)。
通过统的非监督聚类方法无法识别imGCs
为了识别imGCs,研究人员尝试使用一种有监督机器学习方法来对细胞进行分类[5]。该方法通过对原型细胞的分子特征进行提取,进而对每个待分类细胞与原型细胞进行相似性分析,从而进行细胞分类。
进行为了验证该方法的准确性,研究人员首先将其应用于不同年龄小鼠海马的单细胞测序(scRNA-seq)数据集(可准确区分神经母细胞、imGCs和mGCs)。紧接着,研究人员将小鼠海马数据集中颗粒细胞集群中的Dcx+Prox1+Calb1-imGCs细胞作为原型,同时还有主要的非神经元细胞类型的原型(星形胶质细胞、少突胶质细胞前体细胞和小胶质细胞),对模型进行训练。
模型训练完成后,将其用于不同年龄阶段小鼠海马数据集的每个细胞进行分类,并将模型的分类与集群注释相比较,发现模型识别的未成熟神经元主要为imGCs和神经母细胞,且imGCs占到绝大多数。重要的是,该模型可区分imGCs与mGCs。因此,这种基于机器学习的方法可以有效地区分未成熟神经元和成熟神经元。
紧接着,研究人员将该模型应用于婴儿海马数据集,以婴儿imGCs(Dcx+Prox1+Calb1-)和其他非神经元细胞(星形胶质细胞、少突胶质细胞前体细胞、成熟少突胶质细胞和小胶质细胞)为原型细胞进行分类,识别出样本中的imGCs。
使用机器学习的方法识别人婴儿时期海马中imGCs
与mGCs相比,imGCs的基因表达特征与神经系统发育、神经发生和突触发生相关。
此外,研究人员还发现,在婴儿齿状回标本中,93.8%的Dcx+Prox1+Calb1-imGCs表达STMN1(微管解聚蛋白),而91.4%的Stmn1+Prox1+细胞Dcx阳性(imGCs),20.7%的Stmn1+Prox1+细胞Calb1阳性(更大概率为mGCs),这提示了STMN1是imGCs潜在的生物标记。
STMN1是imGCs潜在的生物标记
完善了方法学后,研究人员将此模型应用于不同生命阶段(出生前、儿童期、青春期、成年期和老年期)和区域的人脑样本。
研究结果显示,在出生前,人海马区和前额叶皮质都含有丰富的未成熟神经元,其中大部分为imGCs。在出生后,在所有年龄段的每个海马区标本中,也都发现了与人类imGCs原型细胞高度相似的细胞。每个年龄组颗粒细胞中imGCs的平均百分比为:产前51.8%,婴儿期9.4%,4岁及以后的3.1-7.5%,并呈逐年下降趋势。
不同生命阶段imGCs丰度
为了找到不同生命阶段的人imGCs的共同分子特征,研究人员将imGCs与mGCs进行了比较,发现所有生命阶段的imGCs的基因表达与神经系统发育(如NEUROD1和BHLHE22)、离子转运(如FXYD7和KCNQ5)和神经元投射发育(如SEMA6D和NR2F1)更相关。
与mGCs相比,imGCs的基因表达与神经再生与发育相关
同时,研究人员还发现,imGCs的基因表达存在一定的时间规律,即随着人类发育而产生一定差异,包括谷氨酸受体信号通路的持续上调和神经元迁移和突触投射相关基因的下调。
imGCs的基因表达随年龄增长而发生变化
此外,研究人员还探索了imGCs在不同大脑疾病中的潜在作用,对AD、自闭症谱系疾病、癫痫、抑郁症和精神分裂症疾病风险基因进行了富集分析,发现这些基因在特定年龄的imGCs中的选择性表达,且表达时间与这些疾病发病关键时期相吻合,如AD患者的风险基因在老年时期的imGCs表达更高,自闭症谱系疾病的风险基因在早期发育阶段的imGCs表达更高。
多种疾病的风险基因在疾病发病关键时期相吻合
而在AD患者样本中,海马中imGCs占颗粒细胞的比例远低于正常对照组,且与突触可塑性和信号转导相关的基因表达显著下调。
细胞-细胞相互作用分析显示,AD患者的imGC与星形胶质细胞、少突胶质细胞前体细胞、GABA能中间神经元等的相互作用显著减少。这些结果都表明imGCs在AD中很可能起到一定的作用,具有潜在的研究价值。
AD患者海马中imGCs数量减少且功能失调
最后,为了验证成年后海马产生新神经元的能力,研究人员对年龄从2-61岁的10个癫痫患者的海马样本进行体外培养,其中有8个在体外培养1-2周后可观察到EDU+Prox1+Calb1-新生颗粒细胞,且超过80%的细胞DCX或STMN1阳性。这些结果表明了成年海马后齿状回中仍可产生新的颗粒细胞。
总的来说,该研究通过机器学习的方法,在单细胞分辨率下构建了首个人海马区imGCs在整个生命周期内的动态分子特征图谱,证明了成年后海马中仍然存在imGCs,具有发展为成熟的新颗粒细胞的能力。
同时,研究人员还发现人不同生命阶段的imGCs存在差异表达的基因,且一些与疾病相关的风险基因可在特定阶段在imGCs中选择性表达,具有作为干预靶点的潜力。

分析
对领域进行一次全面的分析。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用