更新于:2024-09-27

Xiangtan University
更新于:2024-09-27
概览
关联
100 项与 湘潭大学 相关的临床结果
登录后查看更多信息
0 项与 湘潭大学 相关的专利(医药)
登录后查看更多信息
12,555
项与 湘潭大学 相关的文献(医药)2025-01-01·SEPARATION AND PURIFICATION TECHNOLOGY
Fe/Mn-MOFs with monocarboxylic acid-induced defects enhances the catalytic oxidation of calcium sulfite in desulfurization ash
作者: Yang, Liuchun ; Su, Mianheng
The application of catalysis is an effective way to enhance calcium sulfite oxidation reactions, which is essential to the calcium-based wet flue gas desulfurization and the handle and/or utilization of the solid byproduct from semi-dry desulfurization process, i.e., desulfurization ash.In this report, we present a method for constructing Fe/Mn(BDC)(DMF,OA) bimetallic catalysts with defect engineering by using monocarboxylic acids with varying chain lengths as regulators.After removing the monocarboxylic acid, the unsaturated coordinated iron and manganese active sites in Fe/Mn(BDC)(DMF,OA) become more abundant.This makes it easier to adsorb O2 and activate it into reactive oxygen species (·O-2), which in turn activates the radical chain reaction and promotes the production of sulfur-oxygen free radicals(·SO-5).D. functional theory (DFT) calculations show that the synergistic effect of Fe-Mn bimetallic catalysts reduces the energy barrier for SO2-4 formation, thereby accelerating the oxidation of sulfite.Upon addition of Fe/Mn(BDC)(DMF,OA), the oxidation rate of calcium sulfite within 3 h increased from 2.76 % to 96.09 %, compared to non-catalytic oxidationAfter five cycles, the 3 h oxidation rate of calcium sulfite decreased by 10.42 % compared to the first cycle, and the higher leaching rate of Mn element was the main reason for the decrease in catalytic performance.The catalyst is non-toxic and economical, showing significant potential for resource utilization of CaSO3-containing desulfurization ash.
2025-01-01·SEPARATION AND PURIFICATION TECHNOLOGY
Formation of Fe2(SO4)3-Fe2O3 interface induced by OCFGs electrostatic anchoring on AC surface: High efficiency NH3-SCR performance at low temperatures
作者: Li, Pan ; Huang, Yan ; Li, Simi ; Liu, Mouli
Low-temperature activity and SO2 poisoning were the key factors limiting the application of NH3-SCR catalysts.In this study, Fe2(SO4)3/AC and Fe2(SO4)3/OAC catalysts were prepared by oxidation function modification and incipient wetness impregnation.In the temperature range of 100-250°, the catalytic performance and SO2 resistance of Fe2(SO4)3/AC and Fe2(SO4)3/OAC catalysts were investigated, the denitrification efficiency increased from 28% (Fe2(SO4)3/AC) to 80% at 100° and reached 100% at 170°, and 100 ppm SO2 was introduced at 250° for 24 h, the denitrification efficiency remains 100% unchanged.The mechanism of activity enhancement was further explored by BET, x-ray diffraction, ICP, TG, XPS, FT-IR, H2-TPR and NH3-TPD.(NH4)2S2O8 modification enhanced the surface acidity, and the increase of surface oxygen-containing functional groups improved the redox performance.At the same time, due to electrostatic anchoring effects of oxygen-containing functional groups, Fe3+ and SO2-4 were adsorbed at different sites and promoted the binding of S to the carbon skeleton to form -C-S-C-.Thus, the interaction between OAC and Fe2(SO4)3 caused part of Fe2(SO4)3 to decompose into Fe2O3, and the formation of Fe2(SO4)3-Fe2O3 interface further enhanced the acidity and redox properties.More importantly, the decomposition temperature of NH4HSO4 was lower and the decomposition was more thorough on the Fe2(SO4)3/OAC than that of Fe2(SO4)3/AC.Finally, the possible mechanism of (NH4)2S2O8-modified Fe2(SO4)3/OAC catalyst to improve NH3-SCR performance was proposed, which is of great significance for the development of NH3-SCR catalyst with sulfur resistance at low temperatures
2024-12-01·SEPARATION AND PURIFICATION TECHNOLOGY
Promotion of toluene catalytic oxidation by M−La/CoOx catalysts directly prepared from Co-MOFs precursors
作者: You, Zhimin ; Li, Mengke ; He, Yujiao ; Jiang, Sicheng ; Tang, Ning
Volatile organic compounds (VOCs) are identified as volatility and toxicity, and have become major pollutants in the atm.The development of low-energy and high-efficiency environmentally friendly VOCs purification technologies is urgent.Therefore, in this paper, a series of M-La/CoOx (M = La addition) catalysts with different La additions were synthesized by a combination of impregnation and calcination using Co-MOFs as precursors.It was shown that the catalytic activity of the constructed 0.012-La/CoOx catalyst was unexpectedly improved (T90% = 227 °C) under optimal reaction conditions (catalyst mass-air velocity = 60000 mL/(g•h)), initial toluene concentration = 1000 ppm when employing gaseous toluene (C7H8) as typical target VOCs.Among the La-doping catalysts, the catalysts containing La on the surface have smaller grain sizes as well as more homogeneous grain distributions than the pure Co3O4-MOF catalysts. 0.012-La/CoOx displayed the highest activity, which is attributed to its smaller particle size, larger sp. surface area (SSA), richer content of Co3+ and surface Oads, which could enhance the oxygen mobility and the redox performance.The possible degradation mechanism of toluene on 0.012-La/CoOx was revealed using in-situ DRIFTS.This work might provide guidance for the development of efficient and environmentally friendly catalytic oxidation of toluene.
2
项与 湘潭大学 相关的新闻(医药)2023-10-09
·药闻康策
2023全国高等教育硕士研究生报名须知教育部、国家发改委、财政部联合印发《关于加快新时代研究生教育改革发展的意见》,表示要将“硕士研究生招生规模稳步扩大。”并且,教育部明确提出要扩大研究生招生规模。为积极落实国家研究生扩招政策,保障基层人员考研需求,特开通一次硕士研究生专属报名通道,本次仅招50人,截止报名时间10月11日,本周三22:00。各省人员均可在规定时间内报名,具体报名工作安排如下:No.1 学历形式教育层次:高等教育研究生三种形式:专业硕士/同等学力申硕/国际研招学历查询:国家认可,中国高等教育学生信息网(学信网)可查学习形式:在职学习/全日制学习学习优势:方便灵活,工作学习两不误No.2 选报院校全国各所大学(部分院校有政策变动,具体以招生简章为准)北京大学、清华大学、中国人民大学、西北大学、宁夏大学、兰州大学、四川大学、西南财经大学、西南大学、四川外国语大学、云南大学、青海大学、安徽大学、河南大学、南京师范大学、福建师范大学、福州大学、华侨大学、东北师范大学、天津商业大学、中国传媒大学、河北大学、山东大学、山西大学、复旦大学、华东师范大学、浙江工商大学、浙江财经大学、南昌大学、湘潭大学、湖南大学、华南农业大学、暨南大学、广西民族大学、海南大学、华中师范大学、中南财经政法大学、西安交通大学等。No.3 报名条件及流程报名条件(以管理类联考为例,具体请加老师咨询)1.具有国家承认的大学本科毕业学历后,有三年或三年以上工作经历者。2.具有国家承认的大专毕业学历毕业学历后,有五年或五年以上工作经历者。3.已获硕士、博士学位,并有两年或两年以上工作经历者。↓在职研究生相关问题请扫码咨询↓ 院校详情·专业选择·课程学习扫码添加老师,申请助学金名额,限前3名报名流程1、报名时间>10月11日22:00提交本期最后一批次培训报名信息2、报名方式 >网上报名:填写报名登记表通过资格审核,由老师线上代办完善报名手续;报名材料(以下资料都由微信发送,仅作报名使用)A:身份证正反扫描件、最高学历毕业证或者学生证扫描件B:一寸蓝底电子版照片(384*512规格,jpg格式)C:填写新生报名登记表No.4 专业招生计划管理类联考(具有大专或本科学历后并具备一定工作年限后可报名)专业类别招生层次招生人数授予学位MBA 硕士研究生5工商管理专业硕士学位MPA硕士研究生7公共管理专业硕士学位MPAcc硕士研究生2会计专业硕士学位MEM硕士研究生4工程管理专业硕士学位图书情报硕士硕士研究生2图书情报硕士学位旅游管理硕士硕士研究生2旅游管理硕士学位审计硕士硕士研究生4审计硕士学位大硕士(具有大专或本科学历后并具备一定工作年限后可报名)专业类别招生层次招生人数授予学位应用心理学硕士研究生2应用心理学专业硕士学位护理学专业硕士研究生4护理学专业硕士学位教育学专业硕士研究生3教育学专业硕士学位农业硕士硕士研究生3农学硕士学位法律硕士硕士研究生3法学硕士学位免统考(具有大专或本科学历后并具备一定工作年限后可报名)专业类别招生层次招生人数授予学位同等学力申硕在职研究生3视院校专业而定国际研招在职研究生4视院校专业而定↓更多院校专业请扫码咨询↓ 院校详情·专业选择·课程学习扫码添加老师,申请助学金名额,限前3名No.5 研究生优势1、毕业双证:毕业后可获研究生学历证和硕士学位证,且在职研究生与全日制研究生证书具有同等法律效力,证书官网可查,社会认可度高。2、人脉扩张:读研期间积累人脉,结识众多顶尖、知名人物,获得更好的交流机会。为以后积累非常重要的资源路径;3、增加专业知识:读研的过程就是一种知识的扩充或实践,是一种上升的学习形式,通过导师授课,可以掌握其他学习理论或学习方法;4、薪资待遇:学历不一样,薪资待遇当然也不一样,提升核心竞争力,获得更大的职业发展空间。No.6 助学金减免招生条件现在咨询报名还可申请助学补贴,满足以下任一条件即可申请!1.本科学历证书(包含成人学历)2.本省户籍3.获奖证明(工作或学校)4.公共事业单位↓助学金条件查询请扫码咨询↓ 院校详情·专业选择·课程学习扫码添加老师,申请助学金名额,限前3名特别提醒1.为避免报考时网络拥堵,现对本次报名学员优先注册,可根据考生意愿优先就近选择考试地点;2.报名时应仔细阅读报名相关事项,提交的报名材料信息须真实、准确、有效。对提供信息不实且影响报名审核的,一经发现,即取消报名资格;3.新生报名务必在规定时间内进行,超过截止时间,视为自动放弃。【免责声明】1.“药闻康策”部分文章信息来源于网络转载是出于传递更多信息之目的,并不意味着赞同其观点或证实其内容的真实性。如对内容有疑议,请及时与我司联系。2.“药闻康策”致力于提供合理、准确、完整的资讯信息,但不保证信息的合理性、准确性和完整性,且不对因信息的不合理、不准确或遗漏导致的任何损失或损害承担责任。3.“药闻康策”所有信息仅供参考,不做任何商业交易或医疗服务的根据,如自行使用“药闻康策”内容发生偏差,我司不承担任何责任,包括但不限于法律责任,赔偿责任。
2023-01-13
·生物谷
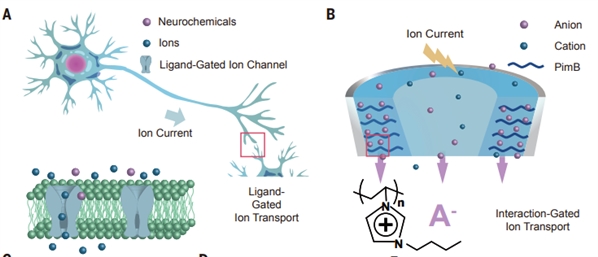
中科院化学研究所活体分析化学院重点实验室于萍和毛兰群团队发展了一种聚电解质限域的流体忆阻器,利用单个器件首次实现了神经化学信号与电信号转导的模拟。
大脑的功能与化学信号密切相关。然而,目前的仿突触器件只能实现对电信号的识别,很难直接感知化学信号。制备具有化学信号响应功能的人工突触成为神经智能传感与模拟等领域的科学难题之一。
在国家自然科学基金委、科技部和中国科学院的大力支持下,中科院化学研究所活体分析化学院重点实验室于萍和毛兰群团队发展了一种聚电解质限域的流体忆阻器,利用单个器件首次实现了神经化学信号与电信号转导的模拟。该研究为发展类化学突触功能器件、神经智能传感、神经形态计算以及神经假肢等提供了新的思路。
该研究团队在长期从事脑神经电分析化学和限域离子传输研究的基础上,提出了基于限域流体器件发展仿神经突触功能的构思。他们构建了聚电解质限域流体体系,发现此体系具有忆阻器的特征,进而利用溶液中对离子在聚电解质刷限域空间内的传输,实现了器件的记忆效应,成功模拟了多种神经电脉冲行为。相比于传统固体器件,他们所发展的流体器件具有可与生物体系相比拟的工作电压和功耗。更重要的是,基于流体体系的特征,此器件可以在生理溶液中模拟神经递质对记忆功能的调控,模拟了突触可塑性的化学调控行为。进一步,他们利用聚电解质对不同对离子的识别能力,实现了神经化学信号与电信号之间转导的模拟,在化学突触的模拟研究中迈出了关键的一步。相关研究结果发表在Science.(2023,379,156-161)上, 文章第一作者是博士生熊天逸,通讯作者为于萍研究员和毛兰群教授,完成单位为中国科学院化学研究所、中国科学院大学、湘潭大学和北京师范大学。
图. 神经元(A)和纳流体忆阻器(B)示意图
100 项与 湘潭大学 相关的药物交易
登录后查看更多信息
100 项与 湘潭大学 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2024年10月05日管线快照
无数据报导
登录后保持更新
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或

营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或

科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用