A multicenter, open-label, balanced, randomized, two-treatment, single-period, parallel-group, single-dose, bioequivalence study of Paliperidone palmitate 100 mg prolonged-release suspension for injection of Qilu Pharmaceuticals Co., Ltd. with Xeplion (paliperidone) 100 mg prolonged-release suspension for injection of Janssen-Cilag International NV in subjects with schizophrenia. - NIL
A randomized, open label, multi-center, two-treatment, two-period, two-sequence, fully replicate, cross-over, multiple dose, steady-state, bioequivalence study of Olaparib Tablets 150 mg (2x150 mg tablets) of Alembic Pharmaceuticals Limited, India with Lynparza® 150mg Filmtabletten Olaparib (2x150 mg tablets) of AstraZeneca AB, SE-151 85 Södertälje, Schweden, in adult patients with carcinoma of the ovary, breast, prostate or adenocarcinoma of the pancreas under fasting condition. - NIL
A randomized, open label, multi-center, two-treatment, two-period, two-sequence, fully replicate, cross-over, multiple dose, steady-state, bioequivalence study of Olaparib Tablets 150 mg (2x150 mg tablets) of Alembic Pharmaceuticals Limited, India with Lynparza® 150mg Filmtabletten Olaparib (2x150 mg tablets) of AstraZeneca AB, SE-151 85 Södertälje, Schweden, in patients with ovarian cancer, breast cancer, prostate cancer or adenocarcinoma of the pancreas under fed condition. - NIL
100 项与 CBCC Global Research LLP 相关的临床结果
0 项与 CBCC Global Research LLP 相关的专利(医药)
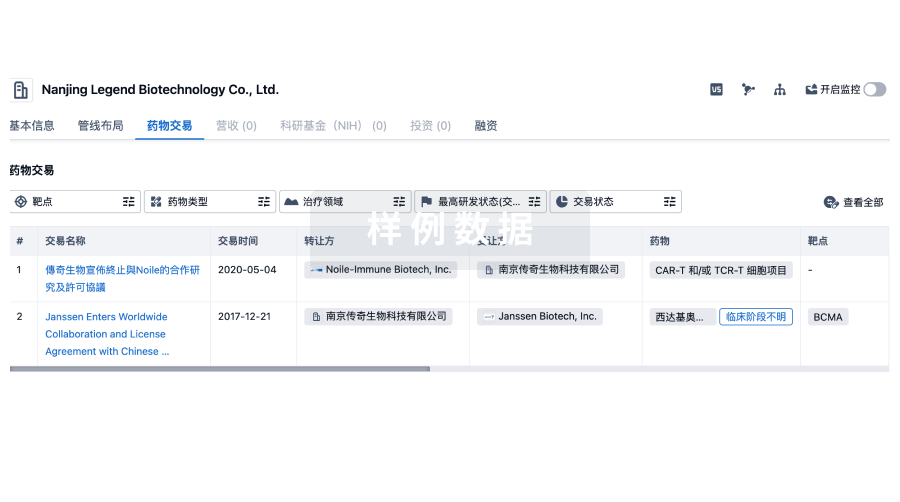
100 项与 CBCC Global Research LLP 相关的药物交易
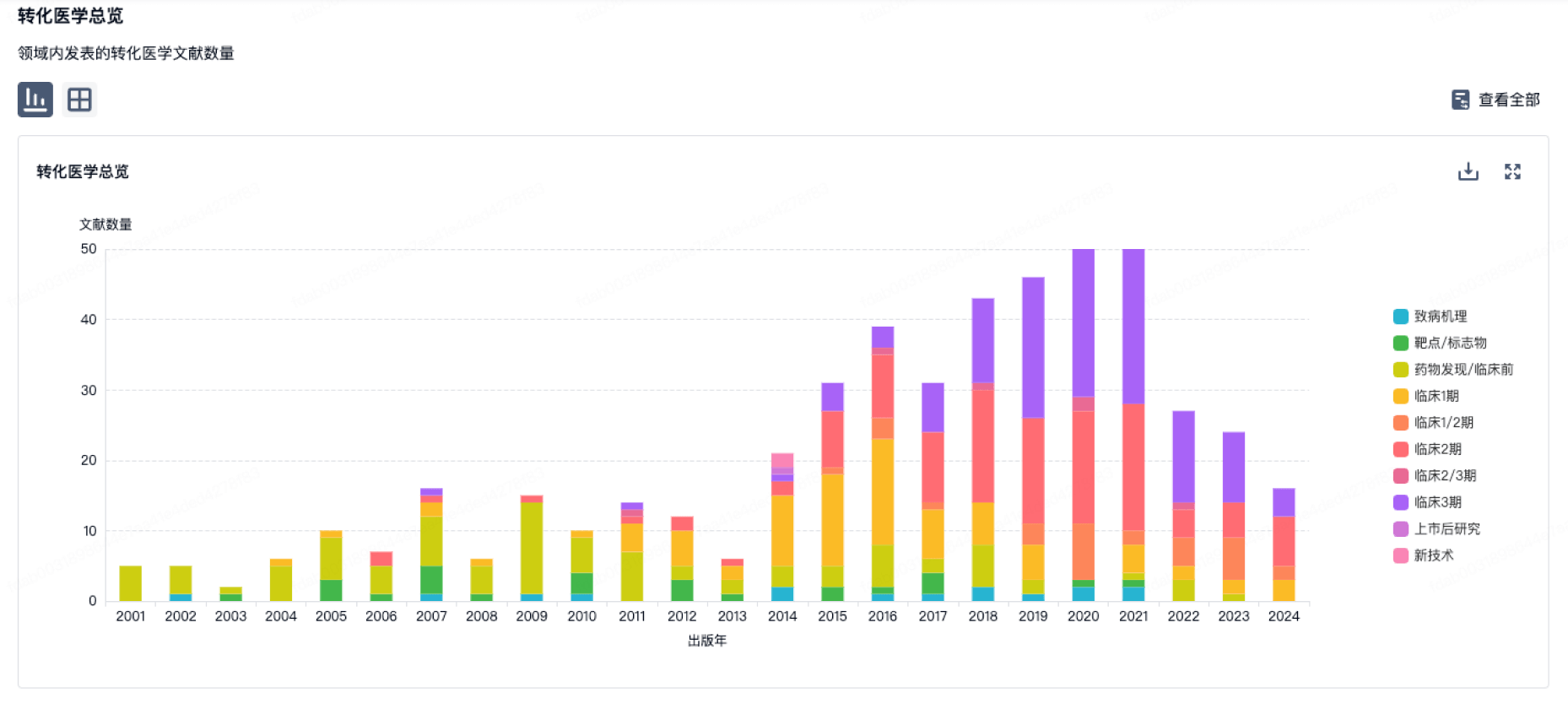
100 项与 CBCC Global Research LLP 相关的转化医学