PYC'S FOURTH DRUG CANDIDATE HAS DISEASE-MODIFYING POTENTIAL IN POLYCYSTIC KIDNEY DISEASE
2023-11-13
PYC has developed a new drug candidate for the >5 million people worldwide[1] with Polycystic Kidney Disease (PKD)
This drug candidate has
demonstrated efficacy in human models derived from the kidneys of patients with end-stage renal failure due to PKD
[2]
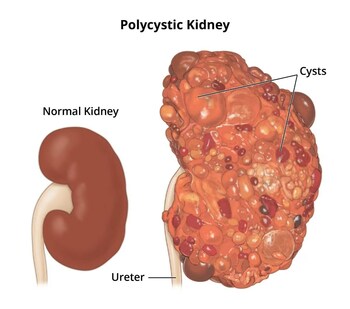
PKD is a life-changing disease affecting 1 in every 1,000 people[3]
Half of the patient population with PKD will require a kidney transplant by the age of 60 due to the absence of impactful treatment options in this disease[4]
PKD represents an addressable market of
>US$10 billion p.a. and is an area of major commercial interest for the drug development industry[5]
Pre-clinical models suggest that addressing the underlying cause of PKD (the way that PYC's drug candidate works) could
arrest the course of the disease in humans, enabling damaged kidneys to
regenerate and
restore function[6]
PYC plans to accelerate this drug candidate into human trials in ~12 months – pursuing a potential high-velocity path through clinical trials to market[7]
Investor Webinar Friday 17 November
PYC will host an investor call at 9am AWST/12pm AEDST on Friday 17 November to discuss these results – investors can register for the call here:
https://us02web.zoom.us/webinar/register/WN_p2QQKKZ-TxOms2MOaUjijg
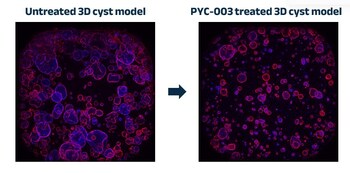
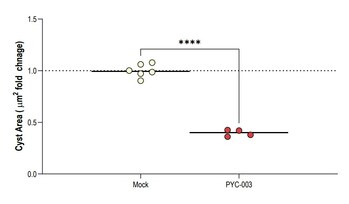
PERTH, Australia and SAN FRANCISCO, Nov. 13, 2023 /PRNewswire/ -- PYC Therapeutics today announces the results of a study conducted in human 3-dimensional models derived from patients with end-stage renal failure due to Autosomal Dominant Polycystic Kidney Disease (PKD). The results demonstrate that an investigational drug candidate designed by PYC (known as PYC-003) to address this disease at its root cause is effective. These 3D patient-derived cyst models represent the 'gold-standard' pre-clinical assay for evaluating drug candidates in this indication[8].
Continue Reading
Preview
来源: PRNewswire
Figure 1
Preview
来源: PRNewswire
Figure 2
Preview
来源: PRNewswire
Figure 3
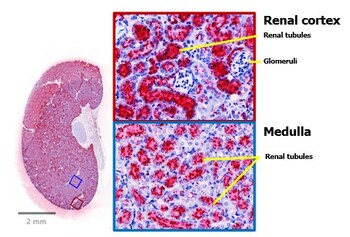
Preview
来源: PRNewswire
Figure 4
更多内容,请访问原始网站
文中所述内容并不反映新药情报库及其所属公司任何意见及观点,如有版权侵扰或错误之处,请及时联系我们,我们会在24小时内配合处理。
适应症
靶点
-药物
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。



