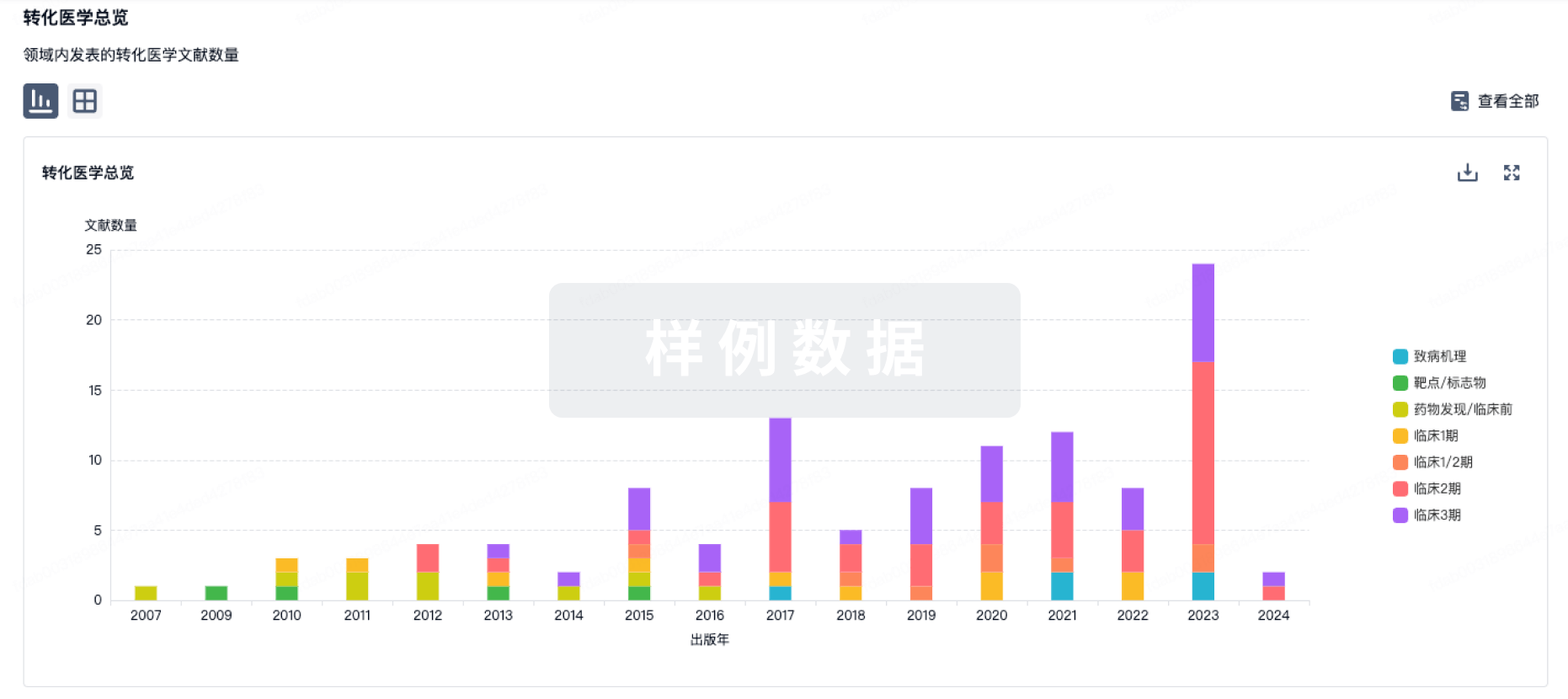
Objective: To perform subchronic toxicity study on Lactobacillus fermentum GM 090 in rat.Method: Groups of 10 male and 10 female weaning Sprague-Dawley rats were given 0, 1.25, 2.5 and 5.0 g/kg body weight Lactobacillus fermentum GM 090 by gavage, resp.Body weights and food consumption were measured weekly.Blood samples were obtained in the middle and at the end of the study, and finally selected organs were weighed and recorded.Results: There were no statistically significant differences in body weights between treatment groups and control group in each week.No significant differences were observed between the treatment groups and the control group in weekly food consumption of females and males.On day 46, for males at 2.5 g/kg body weight group, red blood cell counts were significantly higher than the control group.At the end of the study, urea nitrogen and creatinine at 2.5 g/kg body weight group of females were significantly decreased as compared with the control group.Conclusions: Lactobacillus fermentum GM 090 did not lead to any general organ or systematic toxicity when fed to rats at dietary concentrations as high as 5.0 g/kg body weightLactobacillus fermentum GM 090 is non-toxic up to a level of 5.0 g/kg body weight, when given orally.