更新于:2024-11-01
Trichosanthes Root Extract
栝蒌根提取物
更新于:2024-11-01
概要
基本信息
原研机构 |
在研机构- |
非在研机构 |
最高研发阶段无进展临床2/3期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
关联
1
项与 栝蒌根提取物 相关的临床试验The Effects of Trichosanthes Root on Blood Sugar in Patients With Diabetes Mellitus
Diabetes mellitus (DM) locates fifth of ten leading death etiology, the population of diabetic patients has a tendency of increase. Trichosanthes root is called as Tian Hua Fen, According to traditional Chinese writing record that Trichosanthes root has the action of downbear fire, moisten dryness, and slippery phlegm, and release thirst. The Trichosanthes root may lower blood sugar levels in the modern study. The investigators designed a double blind, randomized controlled study, a total of 60 diabetic patients with high blood sugar were divided into treatment and control groups, and each group was 30 patients. In the treatment group, oral administration of Trichosanthes root 1.5 g, bid/day for 84 days continuously; in the control group, oral administration of Trichosanthes root placebo 1.5 g, bid/day for 84 days continuously. The main outcome measure was the changes of plasma blood sugar and Glycated hemoglobin A1c (HbA1c) levels changed before and after Trichosanthes root treatment. The investigators predict the results of the present study indicated that Trichosanthes root can improve plasma blood sugar and glycated hemoglobin A1c levels.
开始日期2016-07-01 |
申办/合作机构 |
100 项与 栝蒌根提取物 相关的临床结果
登录后查看更多信息
100 项与 栝蒌根提取物 相关的转化医学
登录后查看更多信息
100 项与 栝蒌根提取物 相关的专利(医药)
登录后查看更多信息
4
项与 栝蒌根提取物 相关的文献(医药)1992-07-01·Journal of biochemistry4区 · 生物学
Chemical Synthesis, Molecular Cloning, and Expression of the Gene Coding for the Trichosanthes Trypsin Inhibitor—a Squash Family Inhibitor1
4区 · 生物学
Article
作者: Chang-Qing Chen ; Cheng-Wu Chi ; Ke-Da Gan ; Xiao-Ming Chen ; Man-Fang Zhang ; Yue-Wei Qian
The gene coding for a Trichosanthes trypsin inhibitor analog (Ala-6-TTI) in which methionine at position 6 was replaced by alanine was synthesized chemically. The synthetic gene was cloned into plasmid pWR590-1 and expressed in Escherichia coli as a fusion protein composed of beta-galactosidase fragment of 590 amino acid residues and (Ala-6)-TTI, with methionine as a connecting residue. After cyanogen bromide cleavage and reduction of the fusion protein, followed by refolding with trypsin-Sepharose 4B as a matrix and affinity chromatography on the immobilized enzyme, the fully active (Ala-6)-TTI was obtained. The trypsin inhibitory activity and amino acid composition of the recombinant (Ala-6)-TTI were consistent with those of the natural one. The (Ala-6)-TTI gene was also cloned into the secretion expression vector, pVT102U/alpha, in Saccharomyces cerevisiae. In order to make the reading frame of the gene compatible with the vector, a nucleotide was inserted into the (Ala-6)-TTI gene via site-directed mutagenesis. The secreted (Ala-6)-TTI was purified and found to be correctly processed at the junction between the alpha-factor leader peptide and (Ala-6)-TTI downstream. Of the two expression systems, the latter is more advantageous in the high yield (greater than 2 mg/liter), easy purification and needlessness of disulfide refolding.
1990-10-01·Science in China. Series B, Chemistry, life sciences & earth sciences
Total synthesis of Trichosanthes trypsin inhibitor and its analogue.
Article
作者: Wu, M L ; Qi, Z W ; Huang, Z F
Trichosanthes trypsin inhibitor (TTI) is a peptide consisting of 27 amino acid residues with three pairs of disulfide bonds. This paper reports the total synthesis and disulfide bond refolding of this inhibitor and its analogue. After purification, the amino acid sequence and stoichiometrical inhibitory activity against trypsin of the synthetic inhibitor were compatible with those of the natural inhibitor. The analogue of this inhibitor in which residue Met in position 6 was replaced by Ala was also synthesized. The antitrypsin activity of this synthetic analogue was also approximate to that of the natural inhibitor.
1990-05-01·Science in China. Series B, Chemistry, life sciences & earth sciences
Studies on natural and modified peptide Trichosanthes trypsin inhibitors.
Article
作者: Chi, Changwu ; Qi, Zhengwu ; Tan, Fulong ; Qian, Yuewei
A peptide trypsin inhibitor was isolated and purified from the roots of Trichosanthes kirilowii (a Chinese medical herb) by using immobilized anhydro-trypsin affinity chromatography and HPLC C18 column reverse chromatography. It contains two major components, both consisting of 27 amino acid residues with three pairs of disulfide bonds. The sequence determination indicated that the difference between them is only in the ninth position, being Gln and Lys, respectively. The peptide bond of the inhibitor reactive site Arg-Ile (3-4) is easy to cleave at low pH by trypsin, resulting in a modified inhibitor. It might be the smallest naturally occurring protein inhibitor so far known. The modification reaction of the Trichosanthes inhibitor with trypsin is similar to the catalytic enzyme-substrate reaction. The dissociation constant of the modified inhibitor with trypsin is around fourfold that of the natural inhibitor.
11
项与 栝蒌根提取物 相关的新闻(医药)2024-06-15
转自:广东省药监局 编辑:水晶
2022年7月广东省药监局出台了《广东省中药材产地趁鲜切制工作指导意见(试行)》,试行期2年。
近日,广东省药监局就“指导意见”试行以来的情况,面向广东省中药相关行业公开征求施行意见:
1、梳理“指导意见”试行期间中药材产地趁鲜切制实践推动企业中药源头质量管理能力提升、取得的社会效益及经济成效;
2、反馈我省中药企业在“指导意见”试行期间中药材产地趁鲜切制工作实践中面临的问题、困难及其解决方案;
3、对“指导意见”及其附件(见广东省药品监督管理局官方网站http://mpa.gd.gov.cn/)的正式实施提出修订建议,并简要说明理由。
广东省各中药企业及相关单位:
为促进广东省中药产业高质量发展,提升我省中药生产质量,强化源头控制,我局根据国家药监局综合司《关于对中药饮片生产企业采购产地加工(趁鲜切制)中药材有关问题的复函》(药监综药管函〔2021〕367号)等有关文件要求,结合我省实际,于2022年7月出台了《广东省中药材产地趁鲜切制工作指导意见(试行)》(以下简称“指导意见”),试行期2年。现就“指导意见”试行以来的情况,面向我省中药相关行业公开征求施行意见:
1、梳理“指导意见”试行期间中药材产地趁鲜切制实践推动企业中药源头质量管理能力提升、取得的社会效益及经济成效;
2、反馈我省中药企业在“指导意见”试行期间中药材产地趁鲜切制工作实践中面临的问题、困难及其解决方案;
3、对“指导意见”及其附件(见广东省药品监督管理局官方网站http://mpa.gd.gov.cn/)的正式实施提出修订建议,并简要说明理由。
欢迎社会各界为指导意见试行版转正提供意见和建议,于6月30日前发送至邮箱gdmpa_ypyc@gd.gov.cn。
联系人:郭友立,电话:020-37886934
广东省药品监督管理局办公室
2024年6月3日
广东省中药材产地趁鲜切制工作指导意见
(试行)
为提升我省中药材、中药饮片质量,强化源头管控,促进广东省中药产业高质量发展,根据国家药监局综合司《关于对中药饮片生产企业采购产地加工(趁鲜切制)中药材有关问题的复函》(药监综药管函〔2021〕367号,以下简称“复函”)等有关文件要求,结合我省实际,现就规范我省中药生产企业自建和采购产地趁鲜切制中药材(以下简称“鲜切药材”)工作提出如下指导意见。
一、指导思想
以习近平总书记关于中医药工作的重要论述为指导,坚持传承精华、守正创新理念,全面落实《中共中央国务院关于促进中医药传承创新发展的意见》《国务院办公厅印发关于加快中医药特色发展若干政策措施的通知》《全国道地药材生产基地建设规划(2018~2025年)》《国家药监局关于促进中药传承创新发展的实施意见》,根据《药品管理法》《中医药法》《药品生产质量管理规范》《中药材生产质量管理规范》等有关规定,结合我省实际,依据风险管理原则,着力规范我省中药生产企业在全国各地自建和采购趁鲜切制中药材行为,促进资源要素向产业链源头集聚,提升中药材产地加工的规范化、规模化,保证我省中药生产质量,推进中医药产业高质量发展,确保公众用药安全。
二、工作目标
贯彻落实国家对中药材产地趁鲜切制工作的有关要求,加强中药材产地趁鲜切制管理,以解决中药材加工过程中因“二次浸润”、切制造成成分流失和损耗增大等难题为出发点,规范中药生产企业开展产地趁鲜切制加工中药材行为,按照风险管理原则确定中药材产地趁鲜切制风险管控品种目录,符合本指导意见要求的品种可有条件趁鲜切制加工,将中药材种植养殖和产地加工过程纳入中药生产企业质量管理体系,落实企业主体责任,促进道地药材产地趁鲜加工“品种基地化,加工规范化,质量可追溯,监管成体系”,提升中药全产业链质量,逐步实现中药材产地趁鲜加工与炮制一体化的目标。
三、工作任务
(一)加强趁鲜切制加工风险管理
1.规范中药生产企业开展产地趁鲜切制加工中药材行为,制定加工指导原则(附件1《广东省中药材产地加工(趁鲜切制)质量管理指南》,以下简称“质量管理指南”)。
建立《广东省中药材产地趁鲜切制风险管控品种目录》(附件2,以下简称“风险管控品种目录”),列入目录的中药材品种,中药生产企业应重点关注其趁鲜切制的质量风险。
中药生产企业或相关行业协会、科研单位、高校等可根据实际经验,结合有关研究验证数据,提出目录内相关品种的风险管控方案;广东省药品监督管理局(以下简称“省药品监管局”)组织专家评审通过,并发布相关品种的风险管控指南;企业经评估符合质量管理指南和风险管控指南要求的,可自建或采购相关品种鲜切药材。
2.明确纳入风险管控品种目录的原则。对不适宜趁鲜切制、有依据表明趁鲜切制对质量有不良影响或明显风险的中药材品种,省药品监管局组织专家评审,分批次纳入目录。纳入原则包含但不限于以下条款:
(1)基原较复杂、市场存在易混淆品或伪品,趁鲜切制后不利于辨别真伪或是否掺杂的;
(2)趁鲜切制对质量有不利影响的,如酸败度容易不合格、主要成分易挥发流失等;
(3)前处理工艺较为复杂,质量控制风险较高的;
(4)因技术原因,还无法实现较大规模或较规范种植、养殖的;
(5)有特殊管理要求的毒性药材;
(6)不利于储存、运输的;
(7)其他趁鲜切制风险较高的。
制成饮片全程无切制步骤的中药材如果实、种子类等,不纳入趁鲜切制管理。
3.建立目录动态调整机制。省药品监管局组织制定首批风险管控品种目录,通过省药品监管局门户网站等方式及时公开,并依据产品质量风险监测情况,按照风险收益比的管控原则,对目录进行动态调整。
(二)规范加工行为
1.总体要求
在满足国家有关要求和本指导意见的前提下,品种基原需符合《中国药典》等国家药品标准或者省、自治区、直辖市中药材标准或中药饮片炮制规范中的相应规定;中药生产企业需对中药材种植区域进行评估,在适宜种植区域自建趁鲜切制加工点或采购鲜切药材;种植、采收、加工等行为应当符合《中药材生产质量管理规范》(GAP)要求;中药生产企业应当有能力落实鲜切药材全过程追溯和质量管理要求并确保最终产品质量。
2.趁鲜切制加工品种要求。以下几种情况中药生产企业可自建或采购鲜切药材:
(1)符合现行版《中国药典》收载的允许趁鲜切制中药材品种(附件3);
(2)其他省、自治区、直辖市发布的允许趁鲜切制的中药材品种;
(3)其他经中药生产企业研究评估适合产地趁鲜切制加工,能够确保最终产品质量的中药材品种;
(4)已列入《广东省中药材产地趁鲜切制风险管控品种目录》的,包括前三款内的品种,我省中药生产企业应重点关注其趁鲜切制的质量风险。经过研究验证,自评符合质量管理指南和省药品监管局发布的相关品种风险管控指南要求,能够确保最终产品质量的,可自建或采购该鲜切药材品种。
3.质量标准及工艺制定实施。中药生产企业使用产地趁鲜切制中药材,应按照“确有需要,质量提升”的原则,制定与中药饮片质量标准相适应的中间产品质量标准(具体要求见附件4),即中药材产地趁鲜切制品标准,作为企业标准试行,以保证最终产品符合预期用途和法规要求。切制加工规程应当有传统经验或者研究验证数据支持,其工艺规程和技术要求应按品种制定,并严格控制。工艺流程包括净选、洗润(软化)、切片(段、块、瓣等)、干燥、包装等工序。应当参照《药品生产质量管理规范》及其中药饮片附录(以下简称“中药饮片GMP”)、《中药材生产质量管理规范》(GAP)相关规定实施,应有完整准确的批生产记录。
4.加工点条件。中药材产地趁鲜加工是中药企业生产的前端延伸链,中药生产企业对鲜切药材的质量安全承担主体责任。中药生产企业可自建加工点或向适当的加工点(产地加工企业)协议采购鲜切药材,并将质量管理体系延伸到该药材的种植、采收、加工等各环节。中药生产企业应与加工点(产地加工企业)签订购买合同和质量协议,严格审核加工点的质量管理体系,条件至少应包含以下内容:
(1)经当地政府相关部门确认的中药材产地趁鲜加工点,或成规模的传统种植基地设置的中药材产地趁鲜加工点。
(2)加工点应具备相应的质量管理体系,配备与其加工规模相适应的专业技术人员及加工、干燥、包装、仓储等设施设备,并具备配合中药生产企业落实药品质量管理要求的能力。
(3)中药生产企业通过对其质量保证能力和风险管理能力进行评估后,按照中药生产企业+加工点(合作社、加工厂)+种植基地的模式,签订采购协议,约定质量责任、操作规程等内容,协议应体现中药生产企业对加工点的鲜切药材质量安全承担完全主体责任。
(4)中药生产企业应确立可实现产品“来源可知、去向可追、质量可查、责任可究”的质量追溯体系建设目标。鼓励中药生产企业利用信息化手段,逐步实现种植、采收、加工、贮存、运输等全产业链质量可追溯。
(5)中药生产企业采购的鲜切药材应有规范的包装和标签,附质量合格标识。其直接接触鲜切药材的包装材料至少应符合食品包装材料标准。标签内容应与企业内控标准表述一致,至少应包括:品名、规格、数量或重量、产地(道地区域明显的药材可标注到市辖区、县)、采收时间、产品批号、贮藏、加工点(企业)名称等。可根据鲜切药材的产品特性、包装形式、质量检验等因素,确定复验期。超过复验期未复验合格的鲜切药材不得用于药品生产。品名项下应括号备注“鲜切药材”,如“品名:黄连(鲜切药材)”。
5.规范采购要求。中药生产企业采购鲜切药材,应符合“必要、可行、可控、可追溯”的原则,对照本指导原则及附件《中药材产地加工(趁鲜切制)质量管理指南》要求,从具备健全质量管理体系的产地加工点采购。不得从各类中药材市场或个人等处购进鲜切药材用于中药饮片生产;也不得从质量管理体系不健全或者不具备质量管理体系的产地加工企业购进鲜切药材用于中药饮片生产;不得将采购的鲜切药材直接包装后作为中药饮片销售。
(三)加强趁鲜切制加工质量管理
1.加强自收自制质量管理。中药生产企业自行开展中药材产地趁鲜切制加工,应当按照广东省药品监督管理局《中药材产地加工(趁鲜切制)质量管理指南》和当地监管部门对趁鲜切制加工的有关要求建设加工点,并对其质量管理体系和风险管理能力进行评估,认为符合要求的,由中药生产企业在药品年度报告中列明相关情况。报告内容应包括:趁鲜切制加工品种的工艺规程和质量标准及其起草说明、《中药材产地趁鲜加工点质量管理自查表》(附件5)等。
2.加强外购鲜切药材质量管理。对于外购鲜切药材的,应当与供应方签订质量保证协议(参考样式见附件6),供应方应当为成规模的中药材种植企业或者农民专业合作社设置的鲜切药材加工点,中药生产企业对外购鲜切药材应当承担全部质量责任,应当对供应方的加工条件、技术水平和质量管理情况进行审查,确认其具备趁鲜加工的条件和能力,所采购品种的技术和质量文件符合要求,能按订单标准组织生产,并应定期开展供应商审计,确保其持续符合要求。外购鲜切药材的中药生产企业经审计评估认为供应方符合《中药材产地加工(趁鲜切制)质量管理指南》要求的,在药品年度报告中列明相关情况。
3.加强中药材源头质量管理。中药生产企业应当将鲜切药材种植养殖过程纳入质量管理体系。外购鲜切药材时,中药生产企业应当与供应方签订质量保证协议,规范中药材种植养殖管理。
(四)压实采购鲜切药材的中药生产企业主体责任
1.中药生产企业采购鲜切药材,应按本节第(三)项第2点要求加强质量管理,并严格审核供应方的质量管理体系,将本企业质量管理体系延伸到所采购鲜切药材的种植、采收、加工等环节,切实做好鲜切药材质量评估、检验和生产过程监测。
2.中药生产企业采购鲜切药材,应当在所采购鲜切药材的产地供应方质量追溯基础上进一步完善信息化追溯体系,逐步实现所采购的鲜切药材在种植、采收、加工、仓储、运输及中药饮片的炮制、生产、销售等全过程可追溯。
3.中药生产企业采购鲜切药材,应当按照中药饮片GMP和法定标准(规范)进行净制、炮炙等生产加工,并经检验合格后,方可放行和上市销售。
四、工作要求
(一)加强领导,强化监管
1.各级药品监管部门要加强对中药材产地趁鲜加工工作的管理,积极探索中药生产企业开展中药材产地趁鲜加工的监管方式,加强日常监管,必要时可延伸检查加工点质量保证体系的执行情况,依法监督中药生产企业履行质量主体责任,严格按照相关法规和本指导意见的要求开展中药材产地加工(趁鲜切制)。
2.加强相关中药生产企业监督管理,严防不符合要求、假冒伪劣的产品流入药用渠道。发现存在药品质量安全风险隐患的,如采购鲜切药材行为不符合本指导意见要求,制成饮片出现国抽、省抽不合格等情况,应当依法依规采取暂停生产销售,责令停止采购使用鲜切药材等风险控制措施;发现生产销售假劣药品等违法违规行为的,要依法依规严厉查处;发现中药生产企业采购鲜切药材工作存在重大问题的或者有重大完善建议的,应及时报告省药品监管局。
(二)示范先行,稳步推进
1.鼓励省内具备产地加工(趁鲜切制)生产条件和质量管理能力的中药生产企业采取多种方式建设鲜切药材加工点,将资金、人员、技术、设备等资源要素向产地集聚,开展产地加工(趁鲜切制)。并选择种植规模大、管理水平高、有一定基础的加工点作为示范单位,树立标杆,典型带路,不断总结建设经验,规范引导加工点遵循中药材产地趁鲜加工条件,推动加工点质量水平显著提升,逐步实现中药材产地趁鲜加工与饮片生产一体化。
2.鼓励大宗地产中药材绿色标准化种植养殖基地建设产地加工点,发挥技术、质量管理、设施设备、产能等优势,健全完善加工点质量管理和追溯体系,形成风险管理、全程控制的全链条管理体系,提升地产中药材质量。采取定点加工或共建加工点等方式,为中药生产企业加工鲜切药材。
(三)主动服务,积极帮促
各级药品监管部门要营造良好营商环境,站在厚植新时期广东中药产业发展新优势,促进形成高质量融合发展的产业生态高度,主动服务,积极作为,全方位为中药生产企业提供政策咨询服务和技术指导,协调解决中药生产企业在中药材产地趁鲜加工工作中遇到的困难和问题。
(四)加强沟通,共治共享
1.各级药品监管部门要积极探索中药材产地趁鲜切制加工的管理模式,加强与加工点所在地政府及相关职能部门的沟通协调,共享地产中药材趁鲜加工点信息,实现中药材趁鲜切制加工信息共享,推动中药生产企业建立完善药品质量追溯体系,促进中药材种植养殖及加工的科学化、规范化,进一步保障中药质量安全。
2.支持行业协会、产业联盟、科研院校等整合资源、搭建平台,在制定鲜切药材的加工技术规范,建立信息化追溯平台以及中药生产企业质量管理体系向种植养殖、加工环节延伸等方面发挥积极作用。
广东省中药生产企业自建和采购产地趁鲜切制中药材的管理以本指导意见为准,由广东省药品监督管理局负责解释,国家出台鲜切药材管理新规定的,按照新规定执行。
附件:1.广东省中药材产地加工(趁鲜切制)质量管理指南
2.广东省中药材产地趁鲜切制风险管控品种目录
3.《中国药典》(2020年版)收载产地趁鲜切制中药材品种目录
4.中药材产地趁鲜切制品质量标准编制要求
5.中药材产地趁鲜加工点质量管理自查表
6.中药材产地趁鲜切制加工质量保证协议(参考样式)
附件1
广东省中药材产地加工(趁鲜切制)质量管理指南
一、适用范围
本指南适用于中药材产地趁鲜加工点(企业、车间)的加工管理和质量控制全过程。
二、原则
中药材产地趁鲜切制品(以下简称“鲜切药材”)属于药用原料,可依法用于生产中药饮片、中药制剂、中药提取物、中药配方颗粒等药品。中药材产地趁鲜加工点应当严格控制鲜切药材加工质量和工艺流程。鲜切药材的基原和质量(形态除外)应当符合《中国药典》等国家药品标准或者省、自治区、直辖市中药材标准或中药饮片炮制规范中的相应规定,种植、采收、加工等应当符合《中药材生产质量管理规范》(GAP)要求;净选、切制和干燥应按照确定的工艺流程加工;在切制、干燥、贮藏、运输过程中,应当采取措施控制污染,防止变质,避免交叉污染、混淆、差错;鲜切药材应当是符合《广东省中药材产地趁鲜切制工作指导意见》相关要求的品种,中药材的趁鲜切制加工不得对药材质量产生不利影响。
三、人员要求
(一)中药生产企业应指定专业人员对加工点的中药材质量负责;加工点应当配备足够数量并具有和岗位职责相对应资质的生产和质量管理人员;生产、质量的管理负责人应当有中药学、药学或者农学等相关专业大专及以上学历并有中药材生产、质量管理实践经验,或者有中药材生产、质量管理五年以上的实践经验,且均须经过中药饮片GMP或《中药材生产质量管理规范》和《广东省中药材产地趁鲜切制工作指导意见》等与生产要求相适应的培训。生产、加工技术人员应具有3年以上中药材加工经验,具备鉴别中药材真伪优劣的能力。
(二)应由专人负责培训管理工作,培训的内容应包括中药专业知识、岗位技能和相关法规知识等。对直接从事中药材生产活动的人员应当培训至基本掌握中药材的生长发育习性、对环境条件的要求,以及田间管理或者饲养管理(如肥料和农药或者饲料和兽药使用),和采收、产地加工、贮存养护等的基本要求。
(三)加工点应当对管理和生产人员的健康进行管理。直接接触药材的工作人员应身体健康,患有可能污染药材疾病的人员不得直接从事养殖、产地加工、包装等工作;无关人员不得进入中药材养殖控制区域,如确需进入,应当确认个人健康状况无污染风险。
四、选址要求
(一)应设置在中药材种植规模较大且相对集中、交通便利的区域。
(二)应远离有害废弃物以及粉尘、有害气体、放射性物质和其他扩散性污染源等,整洁卫生,应符合环保、消防要求。基地选址范围内,加工点至少完成一个生产周期中药材种植或者养殖,并有收获期中药材质量检测数据且符合采购方中药生产企业内控质量标准。
(三)不宜选择易发生洪涝灾害的地区,难以避开时应设计必要的防范措施;厂区周围不宜有虫害大量孳生的潜在场所,难以避开时应设计必要的防范措施。
(四)内地面、路面应铺设混凝土、沥青或者其他硬质材料,厂区应有适当的排水系统,防止扬尘和积水等现象的发生。
(五)布局合理,加工用房、仓库面积应与加工规模相适应;生活区应与生产区保持适当距离或分隔。
(六)厂区地面、路面及运输等不应当对药材的加工造成污染。
五、加工车间与设施要求
(一)车间与设施应按加工工艺流程合理布局,并设置与其加工规模相适应的净制、切制、干燥等操作间。
(二)车间地面、墙壁、天棚等内表面应平整,易于清洁,不易产生脱落物,不易滋生霉菌;应有适当的排水措施防止积水;应有防止昆虫或其他动物等进入的设施,灭鼠药、杀虫剂、烟熏剂等不得对设备、物料、产品造成污染或影响安全生产。
(三)具备与加工规模相适应的硬化晾晒场(或与加工品种相适应的干燥设备或者烘房),应有防止昆虫、鸟类或啮齿类动物等进入的设施。
(四)仓库应当设置有足够的存放区域避免混淆,便于通风换气,并配备适当的设施,采取有效措施,对温、湿度进行监控,防止虫蛀、鲜药材堆积发霉、发酵变质。
六、设备要求
应根据中药材的不同特性需要,选用能满足加工工艺要求的设备。与中药材产地加工品直接接触的设备、工具、容器应易清洁消毒,不易产生脱落物,不对中药材质量产生不良影响。
七、包装、放行与储运要求
(一)加工点应当制定包装、放行和储运技术规程,主要包括以下环节:
1.包装材料及包装方法要求:包括采收、加工、贮存各阶段的包装材料要求及包装方法;
2.标签要求:标签的样式、标识的内容等;
3.放行制度:放行检查内容、放行程序、放行人等。
4.贮存场所要求:包括采收后临时存放、加工过程中存放、成品存放等对环境条件的要求;
5.运输及装卸要求:车辆、工具、覆盖等的要求及操作要求;
6.发运要求。
(二)产地加工药材应当按照制定的包装技术规程选用包装材料,有规范的包装和标签,并附质量合格标识。选用能保证其贮存和运输期间质量的包装材料或容器。
(三)应当分垛存放中药材,不同品种、不同批的中药材不得放在同一个货位上;根据中药材对贮存温度、湿度、光照、通风等条件的要求,确定仓储设施条件;鼓励采用有利于中药材质量稳定的冷藏、气调等现代贮存保管新技术、新设备。有特殊贮存要求的中药材贮存,应当符合国家相关规定。
(四)包装必须印有或者贴有清晰标签,不易脱落或者损坏;标签内容应当包括:品名、基原、规格、产地、数量或重量、采收日期、包装日期、产品批号、贮藏、复验期(如有)、追溯标志(或产地片标识)、加工点名称等信息,应能防止混淆和差错。直接接触药材的包装材料至少应当符合食用标准要求,不得对产品质量和后续的生产、使用产生不良影响。
(五)应当执行中药材放行制度,对每批药材进行质量评价,审核生产、检验等相关记录;由质量管理负责人签名批准放行,确保每批中药材生产、检验符合标准和技术规程要求;不合格药材应当单独处理,并有记录。
(六)应当按照技术规程装卸、运输,运输过程应采取有效可靠的措施,防止中药材发生变质,保证其质量稳定,防止发生混淆、污染、异物混入、包装破损、雨雪淋湿等。
八、文件管理要求
(一)应具有相应的产地加工产品质量标准和工艺文件以及包括人员管理、原料管理、加工过程管理、仓储管理等制度文件。生产规程应包括净选、切制、干燥、包装等工序,且在规定时限内完成趁鲜切制加工。切制加工规程应当有传统经验或者研究验证数据支持。
(二)应对中药材产地加工和包装全过程的管理和质量控制情况进行批生产记录,批生产记录至少应包括以下内容:中药材的名称、批号、投料量及投料记录;净制、切制、干燥工艺的设备编号;加工前的检查和核对的记录;各工序的加工操作记录、清场记录、生产人员记录等。
(三)应建立人员档案,包括人员资质、人员培训和健康记录等内容。培训记录至少应包括培训内容、培训人、参加培训人员、培训时间、培训地点、规模、培训效果评价等内容。
(四)中药材的质量追溯体系应能够保证中药材种植、采收、加工、干燥、包装、仓储及销售等全过程可追溯。追溯信息可包括:产地地理坐标、种植面积、亩产数量、种子种苗来源、农药化肥的使用记录、种植时间、采收时间、加工日期、成品数量、成品去向等,保证中药材来源质量可溯可控。
(五)质量标准、工艺文件以及管理制度等应长期保存,批生产记录应至少保存至产品复验期后一年,未制定复验期的至少保存至该批中药材销售后三年。
(六)加工点应当根据影响中药材质量的关键环节,结合管理实际,明确生产记录要求:
1.按生产单元进行记录,覆盖生产过程的主要环节,附必要照片或者图像,保证可追溯;
2.药用植物种植主要记录:种子种苗来源及鉴定,种子处理,播种或移栽、定植时间及面积;肥料种类、施用时间、施用量、施用方法;重大病虫草害等的发生时间、为害程度,施用农药名称、来源、施用量、施用时间、方法和施用人等;灌溉时间、方法及灌水量;重大气候灾害发生时间、危害情况;主要物侯期。
3.药用动物养殖主要记录:繁殖材料及鉴定;饲养起始时间;疾病预防措施,疾病发生时间、程度及治疗方法;饲料种类及饲喂量。
4.采收加工主要记录:采收时间及方法;临时存放措施及时间;拣选及去除非药用部位方式;清洗时间;干燥方法和温度;特殊加工手段等关键因素。
5.包装及储运记录:包装时间;入库时间;库温度、湿度;除虫除霉时间及方法;出库时间及去向;运输条件等。
九、加工管理要求
(一)进入加工区的人员应进行更衣、洗手,着装应便于安全操作,保持整洁。不得携带或存放与药材加工无关的个人用品。
(二)清洗后的中药材应当及时进行晾晒,并不得直接接触地面。晾晒过程应有有效的防虫(动物)、防雨等防污染措施,控制环境尘土等污染。选择适合中药材的干燥方式,应当阴干的药材不得暴晒。采用设施、设备干燥中药材,应当控制好干燥温度、湿度和干燥时间。
(三)应当使用流动的生活饮用水清洗中药材,及时、迅速完成中药材清洗,防止长时间浸泡。用过的水不得用于清洗其他中药材。不同的中药材不得同时在同一容器中清洗、浸润。
(四)应当及时清洁加工场地、容器、设备。保证清洗、晾晒和干燥环境、场地、设施和工具不对药材产生污染;注意防冻、防雨、防潮、防鼠、防虫及防禽畜。
(五)以中药材投料日期作为加工日期。可追溯且品质均一的一定数量成品确定为同一个批次。
(六)在同一操作间内同时进行不同品种、规格的中药饮片加工操作应有防止交叉污染的隔离措施;合格品和不合格品及异物应有效区分。
(七)药材加工过程中禁止使用有毒、有害物质用于防霉、防腐、防蛀虫;禁止一切染色、增重、漂白、掺杂使假等行为。有特殊加工要求的中药材,应当严格按照制定的技术规程进行加工,如及时去皮、去心,控制好蒸、煮时间等。
(八)对于协议采购,采购方中药生产企业应当对供应方加工点的中药材加工过程进行监督。
(九)应当在规定时间内加工完毕,加工过程中的临时存放不得影响中药材品质。
十、质量控制管理要求
(一)中药生产企业应对产地加工的中药材质量和来源进行监督和控制。
(二)中药生产企业应对鲜切药材加工工艺流程、技术要求、工艺验证等进行审核,确认其符合要求。工艺流程技术包括净制、切制、干燥、整理和包装等。对于协议采购,供应方加工点应根据采购方中药生产企业的工艺流程与技术要求,制定相应的技术标准;采购方应对供应方的管理文件进行审核,确认其符合要求。
(三)中药生产企业应制定产地加工产品的内控检验质量标准,该标准原则上应不低于同品种中药材、中药饮片的法定检验标准(除形态外),能保证生产的中药成品在其有效期内持续符合规定。
(四)中药生产企业应对其采购的每批产地加工产品进行留样。留样量至少应为两倍全检量。留样时间应当保存至使用该批鲜切药材生产的最后一批中药饮片或制剂产品放行后一年。
(五)中药生产企业应对其使用的产地加工产品进行年度质量回顾分析,对影响最终产品质量的变更应当按法规要求进行变更管理,应当保存所有变更的文件和记录。
(六)对于协议采购的,采购方中药生产企业应当对供应方(加工点)的加工条件、技术水平和质量管理情况进行现场审核,确保供应方的加工条件和质量管理能力持续符合要求。
(七)应根据双方质量协议,通过自行检验或委托具备相应资质的检验机构等方式对产品进行检验,检验合格后方可销售和采购。
十一、中药生产企业购进鲜切药材用于中药生产,应当符合以下要求
(一)购进的鲜切药材应当是符合《广东省中药材产地趁鲜切制工作指导意见》相关要求的品种。
(二)质量管理体系延伸到鲜切药材的种植、采收、加工等环节,审核种植、采收等环节是否符合《中药材生产质量管理规范》的基本要求,审核切制加工环节是否符合中药饮片GMP要求,审核产地加工企业的质量管理体系是否有效运行。
(三)与产地加工点签订购买合同和质量协议并妥善保存。
(四)鲜切药材应当入库验收。
(五)对采购的鲜切药材,应当按照中药饮片GMP要求以及《中国药典》等国家药品标准或者省、自治区、直辖市中药材标准或中药饮片炮制规范进行净制、炮炙等生产加工,并经检验合格后,方可销售或进入后续生产环节;仅净制的,经验证评估后可引用鲜切药材的检验数据,并在中药饮片检验报告中注明。
(六)对采购的鲜切药材进行质量评估,承担质量管理主体责任。
(七)建立中药信息化追溯系统,逐步实现使用鲜切药材生产、销售的中药全过程可追溯。可与产地加工点(企业)及其所在地政府建立的鲜切药材信息化追溯平台对接,共享有关信息和数据;或在所采购鲜切药材产地加工点(企业)质量追溯基础上进一步完善信息化追溯体系,逐步实现所采购的鲜切药材在种植、采收、加工、干燥、包装、仓储及生产的中药(饮片)炮制、生产、销售等全过程可追溯。
附件2
附件3
《中国药典》(2020年版)收载产地
趁鲜切制中药材品种目录
一、药材切片(共29个品种):干姜、土茯苓、山柰、山楂、山药、川木通、三棵针、片姜黄、乌药、功劳木、附子、地榆、皂角刺、鸡血藤、佛手、苦参、狗脊、粉萆薢、浙贝母、桑枝、菝葜、绵萆薢、葛根、紫苏梗、黄山药、竹茹、桂枝、滇鸡血藤、狼毒。
二、药材切段(共18个品种):大血藤、小通草、肉苁蓉、青风藤、钩藤、高良姜、益母草、通草、桑寄生、黄藤、锁阳、槲寄生、颠茄草、野木瓜、广东紫珠、首乌藤、桃枝、铁皮石斛。
三、药材切块(共3个品种):何首乌、茯苓、商陆。
四、药材切瓣(共4个品种):木瓜、化橘红、枳壳、枳实。
五、药材切瓣或片、段(共11个品种):丁公藤、大黄、天花粉、木香、白蔹、防己、两面针、虎杖、香橼、粉葛、大腹皮。
六、去心(共3个):远志、莲子、牡丹皮。
七、去粗皮(共2个品种):苦楝皮、椿皮。
附件4
中药材产地趁鲜切制品质量标准编制要求
一、原则
中药材产地趁鲜切制品(以下简称“鲜切药材”)属于药用中药材原料,可依法用于生产中药饮片、中药制剂、中药提取物、中药配方颗粒等药品。鲜切药材应当是符合《广东省中药材产地趁鲜切制工作指导意见》相关要求的品种,品种选择应当遵循:市场需要、品质提升、依据充分、数据支持、能够确保最终产品质量。标准的制定应当在总结传统经验的基础上,根据中药材自身的生物学与理化特性,通过科学的研究评价,并在实践应用中证实其切实可行。
二、一般体例要求
标准所用术语、符号、计量单位、检验方法及相关要求等,均执行《中国药典》等法定标准的有关规定。
标准应包括:封面、目次、前言、引言、品名、范围、规范性引用文件、术语和定义、鲜切药材质量要求、包装、规范性附录、资料性附录、参考文献等。
三、鲜切药材标准内容要求
(一)标准名称
鲜切药材名称应参照《中国药品通用名称命名原则》有关规定命名,应与《中国药典》、《部颁标准》等收载的名称一致,并表明鲜制。示例:“xx企业标准大黄(鲜切药材)”。
(二)前言、范围、规范性引用文件、术语和定义
前言明确标准的提出、归口单位及主要起草单位和起草人等。
范围对标准规定的具体内容及其适用范围界定的原则和方法等进行具体解释说明,也应根据标准内容对于不适用方面做出具体解释说明。
规范性引用文件应列出标准中规范性引用文件的清单,其排列顺序为:国家标准、行业标准、国际标准或文件、其他国际标准或文件。法规不应作为规范性引用文件。
术语和定义写出标准中出现的需要说明的术语和定义。
基原写出适用于标准的基原名称和种质类型。基原名称应包括正确的中文名、拉丁学名。植物名称(正名、拉丁名)原则上参考《中国药典》、《中国植物志》等相关参考书。必要时,基原名称和种质类型可分条描述。
术语使用规范的自然科学名词术语,以全国自然科学名词审定委员会公布的规范名词(科学出版社)为准。
(三)质量要求
1.来源鲜切药材来源包括基原(单基原或多基原)即原植(动)物的中文名、拉丁学名、药用部位、生长年限(如有)、采收季节等。多基元鲜切药材制定质量标准时应考虑到其差异。
2.产地加工工艺流程与技术要求
(1)鲜切药材的工艺流程描述经试验验证的鲜切药材的工艺流程图。
(2)鲜切药材的工艺技术要求主要包括但不限于以下环节:
①净制要求。净制的主要目的是去除鲜药材中含有的泥沙等杂质,分离并去除非药用部位,以达到符合相关标准对药材的净度要求。净制应根据鲜药材具体情况,分别选用清洗、挑选、风选、水选、筛选、剪、切、压片、刮削、剔除、刷、擦、碾串、火燎及泡洗等方法,制定适宜的净制技术,鼓励运用科学、现代、稳定的净制设备代替人工进行。
②切制要求。切制的主要目的是根据相关标准对于饮片片型的要求,将净制合格的药材趁鲜或蒸、烫后干燥、烘干,到一定程度后直接或经压片等操作后切制成为片、段、块、丝等。并应充分考虑并研究鲜药材其道地或传统的产地加工方法,对其切制加工投料的原料状态、加工技术及标准作出明确要求;对切制并干燥后的片型、长度、厚度等变化,制定趁鲜切制的片、段、块、丝鲜品规格参数。切制一般选择以机械为主,辅以手工切制。
③干燥要求。干燥的主要目的是对净制或切制合格的药材进行干燥处理,以达到符合标准对水分含量的要求。干燥处理应以不影响饮片质量为原则,根据鲜药材特点,研究确定适宜的干燥设备,并制定干燥温度、干燥时间等工艺参数。
④筛选与整理要求。筛选与整理的主要目的是去碎屑、去掉异形片、分等级等。应根据鲜药材具体情况,选用挑选、筛选、风选、色选等方法进行筛选与整理。应制订产地趁鲜切制药材的等级要求。
⑤包装要求。干燥完成并经检验合格的趁鲜加工饮片,及时进行包装,应明确包装场所及选用的包装材料的安全卫生等级,最小包装规格(如小包装、大包装),包装方式(如手工包装、半自动包装和全自动包装)。
3.性状按实际形态描述主要特征,尤其注意鲜切药材因趁鲜加工所引起的部分性状改变,包括形状、大小(长度/厚度、直径)、颜色、表面特征、质地、断面、气、味等。因客户要求改变的规格也应列出,并予以描述。
4.鉴别包括显微鉴别、理化鉴别、薄层鉴别、特征图谱/指纹图谱等。鉴别试验应具有专属性并说明选择依据。
(1)显微鉴别应选择容易观察、具有鉴别意义的专属特征列入质量标准;应按照《中国药典》显微鉴别的收录原则、书写顺序和方法进行规范描述。因趁鲜切制引起的特征改变也应列出描述。
(2)理化鉴别包括一般理化鉴别、荧光鉴别及光谱鉴别等方法。中药成分复杂,应根据所含成分的化学性质选择适宜的专属性方法,并说明选择依据。
(3)薄层色谱应能反映该药材的整体特性并尽可能区分正品不同基原及混伪品;应明确对照品的选择及其溶液的制备、供试品溶液的制备、点样量、薄层板、展开剂、展开条件(温度、相对湿度、饱和平衡时间等)、检视方法等,说明鉴别方法、鉴别指标成分或专属性成分的选择依据。
5.检查一般包括杂质、水分、灰分、酸不溶性灰分、内源性有毒有害物质、外源性有毒有害物质等的检查。要注重中药安全性检测方法和指标的建立和完善,加强对重金属及有害元素、残留农药、二氧化硫、真菌毒素、生长调节剂等外源性有害物质的检查。
6.浸出物应参照《中国药典》相关要求建立浸出物的检测项,并对溶剂、浸出方法等作必要的考察,根据品种具体研究数据拟定限度,限度可严于原标准该品种项下的规定。
7.含量测定应建立具有代表性的、与活性相关联的多成分含量测定标准,含量限度的制定应有充分的依据和数据积累。加强对毒性药材相关成分的限量研究,保证安全用药。
8.包装应明确外包装箱标识内容、包件重量等。
9.贮藏确定鲜切药材的贮藏条件。
(四)起草说明
应充分反映研究的全过程,主要内容包括:标准编制概况、标准编制过程(样品收集、研究方法、研究结果以及必要的数据、彩色图片、参考文献)、对比鲜切药材与一般品质量指标提升情况、三批次生产工艺验证与试验数据、鲜切药材企业检验报告、其他相关资料等信息。
附件5、6(略)
欢迎留言,更多观
推荐阅读:
蒲公英Ouryao视频号
投稿、广告、商务合作:
Qinrenlvcha
带量采购核酸药物
2023-10-12
转自:四川省药监局 编辑:水晶10月11日,四川省药监局发布了《四川省中药材产地加工(趁鲜切制)品种目录(第一批)》和《四川省中药材产地加工(趁鲜切制)质量管理指南(试行)》四川省中药材产地加工(趁鲜切制)品种目录(第一批)11个: 天麻、枳壳、白芷、白芍、川芎、黄精、丹参、杜仲、厚朴、黄柏、姜黄。《公告》指出,《中国药典》(2020年版)收载产地趁鲜切制中药材品种可直接开展趁鲜切制,不再在我局公布的单独列入产地加工(趁鲜切制)品种目录中重复收载。 21个地方鲜切药材品种目录(391个)1、重庆20个:川牛膝、党参、独活、杜仲、黄连、黄柏、木香、前胡、天麻、枳壳;白芷、百部、陈皮、大黄、佛手、金荞麦、黄精、牡丹皮、桑白皮、枳实。2、湖北32个:川牛膝、天麻、木瓜、白及、白茅根、陈皮、黄连;百部、大黄、独活、杜仲、骨碎补、合欢皮、厚朴、黄柏、黄精、金樱子肉、桔梗、木香、青风藤、桑白皮、五加皮、玄参、重楼、灵芝。3、湖南15个:玉竹、黄精、茯苓、白术、厚朴、杜仲、枳壳(实)、栀子、白莲子、石菖蒲、陈皮、黄柏、荆芥、蕲蛇、蜈蚣。4、辽宁9个:人参、西洋参、细辛、龙胆、鹿茸、泽兰、黄芪、黄精、玉竹5、吉林13个:人参、西洋参、鹿茸、天麻、苍术、淫羊藿、甘草、返魂草、虎眼万年青、桑黄;灵芝、防风、板蓝根。6、黑龙江42个:药材切片(共27个品种):刺五加、人参、西洋参、赤芍、白芍、黄精、黄芪、黄芩、板蓝根、防风、白鲜皮、草乌、地榆、苦参、柴胡、桔梗、党参、鹿茸、甘草、苍术、天麻、关黄柏、知母、北豆根、蒿本、升麻、穿山龙。药材切段(共8个品种):益母草、槲寄生、返魂草、紫苏梗、暴马丁香、蒲公英、车前草、威灵仙。药材去芯(共2个品种):远志、莲子。药材切丝(共1个品种):关黄柏其他(共4个品种):五味子、金银花、车前子、紫苏子7、天津51个:①药材切片(共34个品种):知母、桔梗、白芍、白术、白芷、牡丹皮、苏木、当归、党参、黄芪、甘草、延胡索、苎麻根、丹参、三棱、柴胡、拳参、生地黄、西洋参、赤芍、黄芩、天花粉、郁金、莪术、槟榔、川牛膝、天麻、泽泻、前胡、川芎、苍术、人参、鹿角、山药。 ②药材切段(共11个品种):徐长卿、北沙参、荆芥、泽兰、忍冬藤、蒲公英、水蛭、牛膝、细辛、石斛、远志。 ③药材切丝(共1个品种):桑白皮。 ④药材切瓣(共2个品种):金樱子(除去毛、核)、川楝子。 ⑤药材切丝或片、段、块(共2个品种):茯神(块)、樟木(片、块)。 ⑥去心(共1个品种):巴戟天。 8、山东 31个: ①药材切片:丹参、柴胡、生地黄、西洋参、拳参、赤芍、桔梗、白芷、黄芩、山楂、天花粉、山药、白芍、牡丹皮;②药材切段:北沙参、荆芥、泽兰、忍冬藤、徐长卿、水蛭、蒲公英、远志。药材切片:木瓜、百部、防风、香附、虎杖;药材切片或段:玉竹;药材切丝或块:瓜蒌;药材切丝:荷叶;药材切段:益母草。9、安徽26个:白芍、白术、桔梗、知母、丹参、板蓝根、桑白皮、紫菀、射干、何首乌、天麻、灵芝、蒲公英、墨旱莲、马齿苋、半枝莲、白花蛇舌草、穿心莲、大蓟、藿香、马鞭草、佩兰、仙鹤草、紫苏、桑枝、杜仲。 10、福建16个:铁皮石斛、巴戟天、黄精、灵芝、显齿蛇葡萄、荷叶、盐肤木、穿心莲、福建胡颓子叶、养心草、满山白、肿节风、福建山药、三叶青、绞股蓝、泽泻。 11、甘肃7个:当归、党参、黄芪、红芪、大黄、甘草、板蓝根。 12、云南12个:三七、天麻、白及、重楼;桔梗、黄精、秦艽;黄柏、石斛(金钗石斛)、石斛(鼓槌石斛)、石斛(流苏石斛)干姜 13、内蒙古5个:黄芪、防风、苍术、桔梗、甘草5个品种。14、陕西26个:大黄、天麻、白及、丹参、西洋参、玄参、甘草、远志、茜草、苦参、苍术、延胡索、秦皮、秦艽、葛根、柴胡、黄连黄柏、黄芩、黄芪、黄精、猪苓、淫羊藿、杜仲、厚朴、牡丹皮。15、江西1个:枳壳16、河南20个:丹参、柴胡、生地黄、山药、桔梗、白芷、黄芩、山楂、黄精、牛膝、何首乌、茯苓、商陆;半枝莲、白花蛇舌草、冬凌草、益母草、首乌藤、忍冬藤、板蓝根17、浙江14个:莪术、金荞麦、白花蛇舌草、楤木、杜仲、芦根、三叶青、蛇六谷、无花果、玄参、温郁金、泽泻、天冬、香茶菜。18、广西25个:郁金、莪术、广山药、牛大力、天冬、肉桂、广金钱草、千斤拔、穿心莲、泽泻、巴戟天、百部、广藿香、白及、青蒿、肿节风、黄柏、金樱子肉、玉竹、黄花倒水莲、杜仲、姜黄、厚朴、灵芝、三叉苦。19、新疆11个:板蓝根、甘草、肉苁蓉、新疆赤芍、锁阳、黄芪、丹参、黄芩、牛膝、防风、党参。20、宁夏4个:黄芪、党参、甘草、板蓝根21、四川7个:天麻、枳壳、白芷、白芍、川芎、黄精、丹参、杜仲、厚朴、黄柏、姜黄。中药材和饮片标准查询:
上市批准
2023-06-08
·米内网
精彩内容6月7日,NMPA官网发布最新中药品种保护受理公示,步长制药的人知降糖胶囊申请初保。该药是一款治疗2型糖尿病的独家产品,米内网数据显示,2022年中国三大终端六大市场(统计范围详见本文末)消化系统疾病中成药市场规模超过400亿元,同比增长7.83%。来源:NMPA官网人知降糖胶囊是由知母、人参、黄柏、天花粉等18味中药组成,具有益气养阴,清热生津的功效,常用于2型糖尿病属气阴两虚兼燥热伤津证的辅助治疗,缓解以下症状:倦怠乏力、气短懒言、口干口渴、五心烦热、自汗盗汗、多食易饥、便秘溲赤、心悸失眠、腰酸不适等。近年来中国三大终端六大市场消化系统疾病中成药销售趋势(单位:万元)来源:米内网格局数据库米内网数据显示,2022年中国三大终端六大市场消化系统疾病中成药市场规模超过400亿元,其中零售药店(实体药店+网上药店)终端为主力销售市场,销售规模超过200亿元,同比增长13.27%。步长制药获批上市的消化系统疾病中成药来源:米内网中国上市药品(MID)数据库作为一家以专利中成药为核心,致力于推动中药现代化的医药企业,步长制药目前已有近200款中成药获批上市,包括脑心通胶囊、稳心颗粒、丹红注射液等明星大单品;其中,40多款为消化系统疾病中成药,通脉降糖胶囊、木丹颗粒、胆石利通胶囊等28个品种均已纳入国家医保目录。资料来源:米内网数据库、NMPA官网注:米内网《中国三大终端六大市场药品竞争格局》,统计范围是:城市公立医院和县级公立医院、城市社区中心和乡镇卫生院、城市实体药店和网上药店,不含民营医院、私人诊所、村卫生室,不含县乡村药店;上述销售额以产品在终端的平均零售价计算。数据统计截至6月8日,如有疏漏,欢迎指正!本文为原创稿件,转载请注明来源和作者,否则将追究侵权责任。投稿及报料请发邮件到872470254@qq.com稿件要求详询米内微信首页菜单栏商务及内容合作可联系QQ:412539092【分享、点赞、在看】点一点不失联哦
上市批准专利侵权
100 项与 栝蒌根提取物 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 糖尿病 | 临床3期 | 中国台湾 | 2016-07-01 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
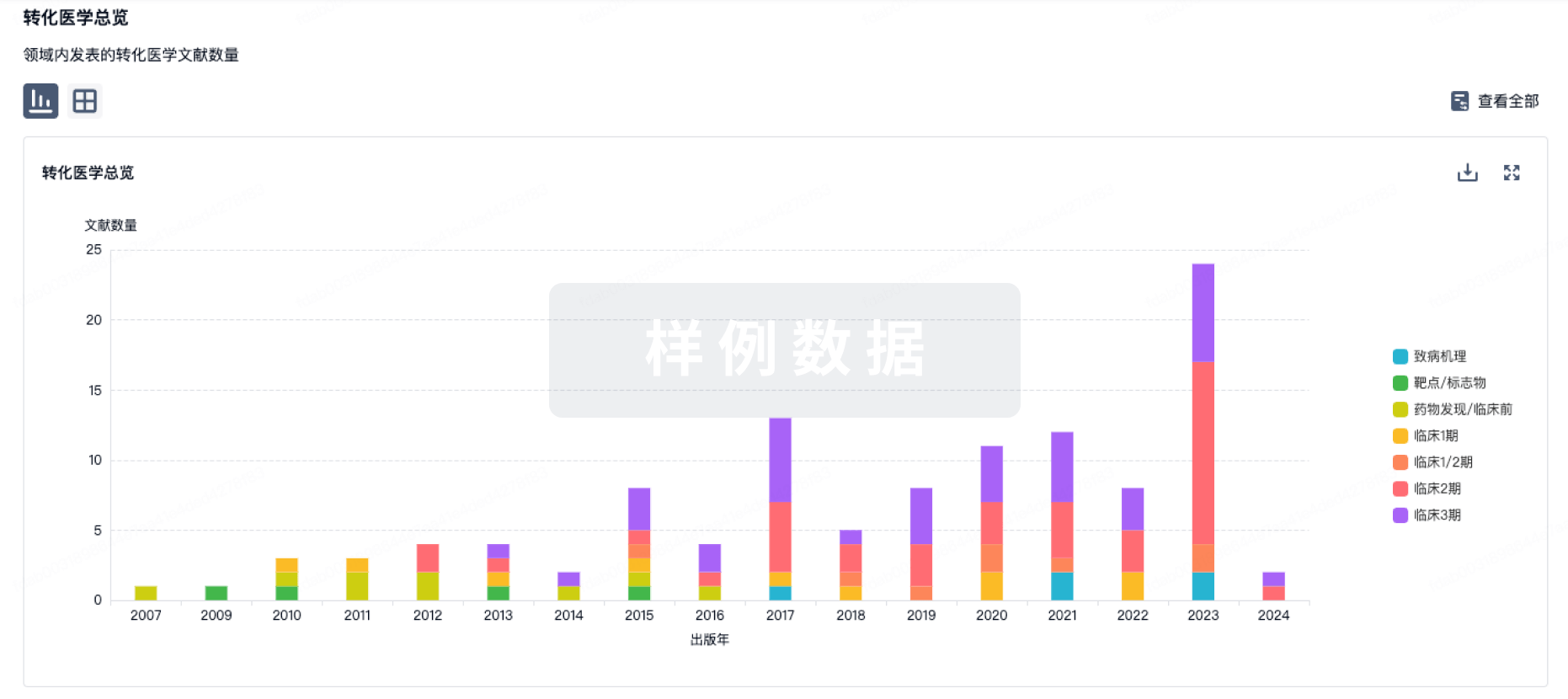
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用