更新于:2024-06-20
NMP
更新于:2024-06-20
概要
基本信息
非在研机构- |
最高研发阶段临床1期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
结构
分子式C5H9NO |
InChIKeySECXISVLQFMRJM-UHFFFAOYSA-N |
CAS号872-50-4 |
关联
1
项与 NMP 相关的临床试验A Phase I, Open Label Dose Escalation Trial of Orally Administered N-methyl-pyrrolidone (NMP) in Patients With Relapsed or Refractory Myeloma
The study will evaluate if the N-methyl-pyrrolidone (NMP) can be safely administered to humans at doses, which induce measurable immunological and anti-tumour effects in patients with myeloma who are resistant to or intolerant of lenalidomide and bortezomib.
开始日期2015-08-20 |
100 项与 NMP 相关的临床结果
登录后查看更多信息
100 项与 NMP 相关的转化医学
登录后查看更多信息
100 项与 NMP 相关的专利(医药)
登录后查看更多信息
885
项与 NMP 相关的文献(医药)2024-03-01·Food chemistry: X
Metabolomics investigation of the chemical variations in white teas with different producing areas and storage durations
Article
作者: Chen, Zewen ; Zhou, Hongjie ; Gao, Jianjian ; Chen, Dan ; Xiong, Mengfan ; Dai, Weidong ; Li, Yali
In this study, we employed nontargeted metabolomics and quantitative analysis to explore the variations in metabolites among white teas from different production areas and with varying storage durations. A total of 83 compounds exhibited differential levels between Zhenghe and Fuding white tea, 89 between Zhenghe and Jinggu, and 75 between Fuding and Jinggu white tea. Concerning the storage of white tea, the concentrations of flavanols, dimeric catechins, and amino acids decreased over time, while N-ethyl-2-pyrrolidone-substituted flavanols (EPSFs), caffeine, adenosine monophosphate (AMP), and adenosine increased. Galloylated flavanols showed a higher propensity to form EPSFs with theanine compared to nongalloylated flavanols during storage. Theanine and epigallocatechin gallate were more inclined to generate S-configuration EPSFs during storage in Fuding and Jinggu white tea samples, while R-configuration EPSFs were more readily formed in Zhenghe white tea samples. This study offers a comprehensive understanding of the changes in metabolites during the storage of white tea.
2024-03-01·Journal of pharmaceutical and biomedical analysis
Application of advanced high resolution mass spectrometric techniques for the analysis of losartan potassium regarding known and unknown impurities
Article
作者: Kinzig, Martina ; Backer, Laura ; Buschmann, Helmut ; Holzgrabe, Ulrike ; Scherf-Clavel, Oliver ; Sörgel, Fritz
Recalls of medicinal products can cause supply bottlenecks. This is often due to the findings of unexpected impurities that pose a health risk to patients. A recent example is losartan potassium which was contaminated with azido-impurities. The choice of the analytical method determines which substances can be detected and thus controlled. In this study a combination of an untargeted screening approach for impurities and a targeted evaluation of high-resolution mass spectrometry data was applied to search for impurities not described so far, leaving out a precise quantification. Six losartan potassium samples were studied regarding known and unknown impurities and hence highlight the applicability and capability of the approach. For separation a Zorbax RR StableBond C18 column (3.0 ×100 mm, particle size of 3.5 µm, pore size of 80 Å), a gradient elution and an electrospray ionization in positive and negative mode for mass spectrometric detection was used. An information-dependent acquisition method was applied for the measurement of losartan potassium samples. The untargeted data evaluation using general unknown comparative screening revealed the presence of N-methyl-2-pyrrolidone (NMP) and another impurity from synthesis. The identity of NMP was corroborated by a spiking experiment and the amount was estimated by means of standard addition. A targeted data evaluation by generating extracted ion chromatograms resulted in finding of four additional impurities. Combined approaches like this are needed to detect and respond to changes in the quality of drugs precociously.
2024-03-01·Advanced materials (Deerfield Beach, Fla.)
Terahertz Liquid Biosensor Based on A Graphene Metasurface for Ultrasensitive Detection with A Quasi‐Bound State in the Continuum
Article
作者: Xie, Yiyang ; Chang, Pengying ; Yao, Haiyun ; Liang, Lanju ; Yan, Xin ; Huang, Chengcheng ; Zhang, Yonggang
Abstract:
The concept of a quasi‐bound state in a continuum (QBIC) has garnered significant attention in various fields such as sensing, communication, and optical switching. Within metasurfaces, QBICs offer a reliable platform that enables sensing capabilities through potent interactions between local electric fields and matter. Herein, a novel terahertz (THz) biosensor based on the integration of QBIC with graphene is reported, which enables multidimensional detection. The proposed biosensor is distinctive because of its ability to discern concentrations of ethanol and N‐methylpyrrolidone in a wide range from 100% to 0%, by monitoring the changes in the resonance intensity and maximum wavelet coefficient. This approach demonstrates an excellent linear fit, which ensures robust quantitative analysis. The remarkable sensitivity of our biosensor enables it to detect minute changes in low‐concentration solutions, with the lowest detection concentration value (LDCV) of 0.21 pg mL−1. 2D wavelet coefficient intensity cards are effectively constructed through continuous wavelet transforms, which presents a more accurate approach for determining the concentration of the solution. Ultimately, the novel sensing platform offers a host of advantages, including heightened sensitivity and reusability. This pioneering approach establishes a new avenue for liquid‐based terahertz biosensing.
1
项与 NMP 相关的新闻(医药)2022-12-09
·ema
CVMP opinions on veterinary medicinal products
Under Regulation (EU) 2019/6
The Committee adopted by consensus a positive opinion for a
marketing authorisation application
for
Brucellin Aquilon
(
Brucella abortus
), from Aquilon Cyl S.L., a new product for
in vivo
diagnosis of
Brucella
infected pigs (skin test) to discriminate false positive results by
Brucella
serological tests.
The Committee adopted by consensus a positive opinion for a
variation
requiring assessment for
Porcilis PCV ID
(subject to
worksharing
procedure), concerning the update of the
product information
of Porcilis PCV ID to include new associated use combinations of Porcilis PCV ID, Porcilis Lawsonia ID, Porcilis M Hyo ID ONCE and Porcilis PRRS.
The Committee adopted by consensus a positive opinion for a grouped
variation
requiring assessment for
Simparica Trio
, concerning the addition of three new therapeutic
indications
: for the treatment of sarcoptic mange (caused by
Sarcoptes scabiei
var.
canis
), for the treatment of demodicosis (caused by
Demodex canis
), and for the prevention of establishment of thelaziosis (adult
Thelazia callipaeda
eyeworm infection).
The Committee adopted by consensus a positive opinion for a
variation
requiring assessment for
Credelio Plus
, concerning the addition of a new therapeutic
indication
for the treatment of demodicosis (caused by
Demodex canis
).
The Committee adopted by consensus a positive opinion for a grouped
variation
requiring assessment for
NexGard
and
Nexgard Spectra
, subject to
worksharing
procedure, concerning the addition of two new therapeutic
indications
for the treatment of tick infestations with
Hyalomma marginatum
and for the treatment of ear mite infestations (caused by
Otodectes cynotis
), and to amend the
product information
to allow the use of the products in breeding, pregnant and lactating female dogs.
The Committee adopted by consensus a positive opinion for a
variation
requiring assessment for
Prevomax
to align the
product information
with version 9.0 of the QRD template.
The Committee adopted by consensus positive opinions for
variations
requiring assessment concerning quality-related changes for :
Locatim
Solensia
Purevax RC, Purevax RCP FeLV, Purevax RCPCh FeLV, BTVPUR, Eurican Herpes 205, Purevax RCPCh
and
Purevax RCP
(subject to
worksharing
procedure)
Suvaxyn CSF Marker, Fevaxyn Pentofel
and
Suvaxyn PRRS MLV
(subject to
worksharing
procedure)
Mirataz
Imoxat
Panacur Aquasol
(subject to
worksharing
procedure)
Procox
(2 procedures)
Bluevac BTV
Union referrals and related procedures
The Committee concluded the
referral
procedure for
veterinary
medicinal products
containing
N-methyl pyrrolidone as an
excipient
.
The matter was referred to the Committee by Germany under Article 82 of Regulation (EU) 2019/6 due to concerns relating to the appropriateness of user and target animal safety warnings in view of the teratogenic potential of N-methyl pyrrolidone. The Committee concluded that no changes need to be made in the
product information
for some of the veterinary
medicinal products
concerned by this
referral
. Furthermore, the Committee agreed that user and/or target animal safety warnings should be added to the
product information
of the remaining products concerned. The Committee adopted by consensus an opinion concluding that the benefit-risk balance of the concerned products remains favourable and that the
marketing authorisations
should be maintained or varied, as applicable.
Scientific advice
The Committee adopted two
scientific advice
reports further to requests for initial advice concerning one immunological product for piglets and a substance intended for inclusion in the list of substances considered as not falling within the scope of Regulation No 470/2009.
The Committee endorsed one clarification of a
scientific advice
concerning a pharmaceutical product for mice, rats, hamsters, gerbils, guinea pigs, chinchillas and rabbits.
Pharmacovigilance
Under Regulation (EU) 2019/6
The Committee adopted recommendations for changes to the
summary of product characteristics
for
Bravecto
,
Proteq West Nile
,
Procox
,
Zuprevo
,
Felpreva
and
Improvac
as outcome of signal detection activities.
Concept papers, guidelines and SOPs
Environmental Risk Assessment
The Committee adopted a draft
reflection paper
on the
environmental risk assessment
of ectoparasiticidal veterinary
medicinal products
used in
cats
and dogs (EMA/
CVMP
/ERA/31905/2022) for a 3-month period of public consultation. This
reflection paper
has been developed to communicate the
CVMP
's view on the scientific discussion regarding the potential environmental impact(s) of ectoparasiticidal veterinary
medicinal products
used in
cats
and dogs, and provides an opportunity for stakeholders to contribute their views.
Efficacy
The Committee adopted a Questions and answers document on the ‘
Guideline
on the
summary of product characteristics
for antiparasitic veterinary
medicinal products
' (EMA/
CVMP
/EWP/799840/2022).
Quality
The Committee adopted a draft
concept paper
on a
guideline
on risk management requirements for elemental impurities in veterinary
medicinal products
, including immunological veterinary
medicinal products
(EMA/
CVMP
/637041/2022) for a 3-month period of public consultation.
The Committee adopted a revised
reflection paper
on the definition of new
active substance
status - Veterinary (EMA/
CVMP
/QWP/3629/2016). This
reflection paper
has been revised to align to Regulation (EU) 2019/6.
The Committee adopted the following revised
guidelines
(GL) to align to Regulation (EU) 2019/6:
GL on development pharmaceutics for veterinary
medicinal products
(EMA/
CVMP
/QWP/684556/2022)
GL on chemistry of
active substance
veterinary (EMA/
CVMP
/QWP/707366/2017)
GL on control of impurities of pharmacopoeial substances (EMA/
CVMP
/QWP/907965/2022)
GL on parametric release (EMA/
CVMP
/QWP/339588/2005)
GL on declaration of storage conditions (EMA/
CVMP
/QWP/857608/2022)
GL on stability of existing
active substance
and related finished product (EMA/
CVMP
/QWP/709423/2022)
GL on additional quality requirements for products intended for incorporation into animal feed (EMA/
CVMP
/QWP/711629/2022)
GL on quality of single-dose spot-on products (EMA/
CVMP
/QWP/544461/2007)
GL on quality of modified release oral dosage forms - veterinary (EMA/
CVMP
/QWP/908160/2022).
Working parties
The Committee adopted the work plans for 2023 for the
CVMP
Working Parties on Antimicrobials (EMA/
CVMP
/AWP/715896/2022),
Environmental Risk Assessment
(EMA/
CVMP
/ERA/828487/2022),
Efficacy
(EMA/
CVMP
/EWP/817611/2022), Novel Therapies and Technologies (EMA/
CVMP
/NTWP/701391/2022),
Pharmacovigilance
(EMA/
CVMP
/PhVWP/593990/2022),
Scientific Advice
(EMA/
CVMP
/SAWP/827784/2022), and Joint
CHMP
/
CVMP
Quality
Working Party
, which will be published on the Agency’s website.
Cooperation with other EU agencies
The Committee adopted a report, prepared in collaboration with EFSA, on the development of a harmonised approach on exposure assessment methodologies for residues from VMPs, feed additives and pesticides in food of animal origin following close of public consultation.
Organisational matters
The Committee adopted the
CVMP
work plan for 2023 (EMA/
CVMP
/617330/2022).
More information about the above-mentioned medicines (including their full
indications
),
guidelines
,
reflection papers
, questions and answers and other documents, such as overviews on comments received during consultation, can be found below in “Related content”.
100 项与 NMP 相关的药物交易
登录后查看更多信息
外链
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| - | - | - |
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 血液肿瘤 | 临床1期 | - | - |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
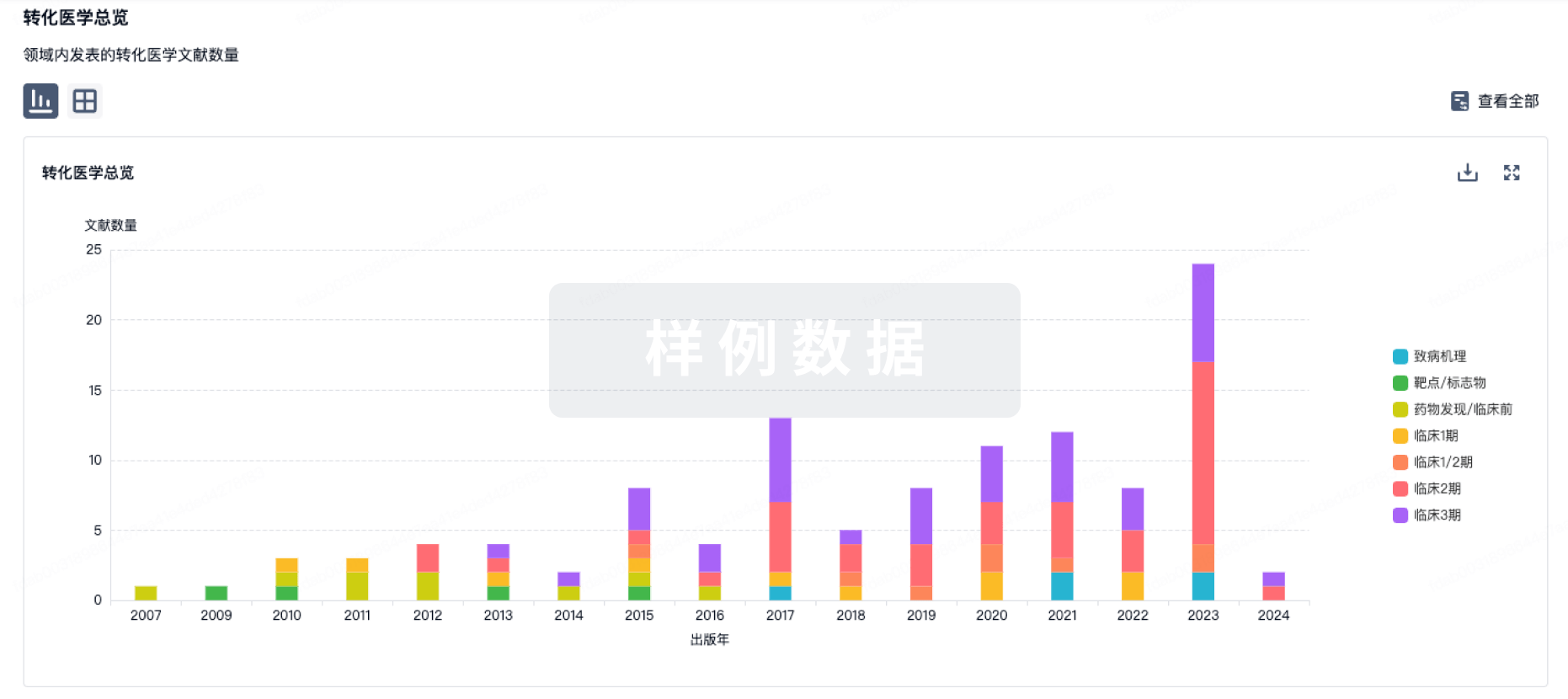
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

