更新于:2024-09-19
Prodencel
普乐登赛
更新于:2024-09-19
概要
基本信息
药物类型 治疗性疫苗 |
别名 Autologous dendritic cell tumor vaccine (Shangai Humantech Biotechnology)、Puledengsai、前列腺癌治疗性疫苗(惠盾生物) + [1] |
靶点- |
作用机制- |
在研适应症 |
非在研适应症- |
原研机构 |
在研机构 |
非在研机构- |
最高研发阶段临床1期 |
首次获批日期- |
最高研发阶段(中国)临床1期 |
特殊审评- |
关联
2
项与 普乐登赛 相关的临床试验在标准治疗失败的转移性去势抵抗性前列腺癌( mCRPC)患者中评价普乐登赛安全性的多中心、非随机、开放和剂量递增的 I 期临床研究
主要目的:评价接受不同剂量的普乐登赛免疫治疗期的安全性、耐受性和剂量限制性毒性( DLT)。
次要目的:评价接受普乐登赛加强免疫期的安全性、耐受性。
探索性目的:评价接受普乐登赛治疗后的特异性免疫效应;评价接受普乐登赛治疗后有效性。
开始日期2022-09-20 |
申办/合作机构 |
A multicenter, non-randomized, open-label, and dose-escalation phase I study to evaluate the safety of Prodencel treatment in patients with metastatic castration-resistant prostate cancer (mCRPC) who failed standard therapy
开始日期2022-07-30 |
申办/合作机构- |
100 项与 普乐登赛 相关的临床结果
登录后查看更多信息
100 项与 普乐登赛 相关的转化医学
登录后查看更多信息
100 项与 普乐登赛 相关的专利(医药)
登录后查看更多信息
19
项与 普乐登赛 相关的文献(医药)2023-01-02·Human Vaccines & Immunotherapeutics
Autologous dendritic cells loaded with antigens from self-renewing autologous tumor cells as patient-specific therapeutic cancer vaccines
Article
作者: Nistor, Gabriel I. ; Keirstead, Hans S. ; Dillman, Robert O.
2020-10-01·Xi bao yu fen zi mian yi xue za zhi = Chinese journal of cellular and molecular immunology
[Dendritic cell tumor vaccine assisted by Tiaohengfang polysaccharides enhances immune function of S180 tumor-bearing mice and inhibits tumor growth].
Article
作者: Liu, Yang ; Xu, Yangyang ; Wang, Yuanyuan ; Zhang, Hongfang ; Li, Wenxia ; Tan, Zhifen ; Zhang, Yimin
2018-12-01·Journal of Translational Medicine2区 · 医学
First results on survival from a large Phase 3 clinical trial of an autologous dendritic cell vaccine in newly diagnosed glioblastoma
2区 · 医学
ArticleOA
作者: Fink, Karen L ; Goldlust, Samuel A ; Salacz, Michael ; Lindhorst, Scott ; Prins, Robert M ; New, Pamela Z ; Heth, Jason A ; Cobbs, Charles S ; Wu, Julian ; May, Sven-Axel ; Maida, Anthony E ; Kim, Lyndon J ; Sloan, Andrew E ; Thompson, Reid C ; Pillainayagam, Clement P ; Moshel, Yaron A ; Abram, Steven R ; Khagi, Simon ; Walter, Kevin A ; Krex, Dietmar ; Dropcho, Edward J ; Piccioni, David ; Trusheim, John E ; Brem, Steven ; Duma, Christopher ; Pearlman, Michael ; Ashkan, Keyoumars ; Pluard, Timothy J ; Lillehei, Kevin O ; Iwamoto, Fabio M ; Bota, Daniela A ; Cloughesy, Timothy F ; Loudon, William G ; Mulholland, Paul ; Etame, Arnold B ; Bosch, Marnix L ; Mathieu, David ; Taylor, Sarah ; Portnow, Jana ; Taylor, Lynne P ; Villano, John L ; Westphal, Manfred ; Baskin, David S ; Elinzano, Heinrich ; Aiken, Robert ; Liau, Linda M ; Mikkelsen, Tom ; Geoffroy, Francois J ; Tse, Victor ; Lutzky, Jose ; Schackert, Gabriele ; Kesari, Santosh ; Avigan, David E ; Petrecca, Kevin ; Toms, Steven A ; Green, Richard M ; Grewal, Jai ; Meisel, Hans-Jorg ; Ewend, Matthew G ; Schulder, Michael ; Tran, David D ; Duic, Paul ; Chaudhary, Rekha ; Brenner, Andrew J ; Lacroix, Michel ; Walbert, Tobias ; Davis, Raphael P ; Campian, Jian L ; D'Andre, Stacy D
100 项与 普乐登赛 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 去势抵抗性前列腺癌 | 临床2期 | 中国 | 2021-12-02 | |
| 转移性去势抵抗性前列腺癌 | 临床1期 | 中国 | 2022-08-08 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
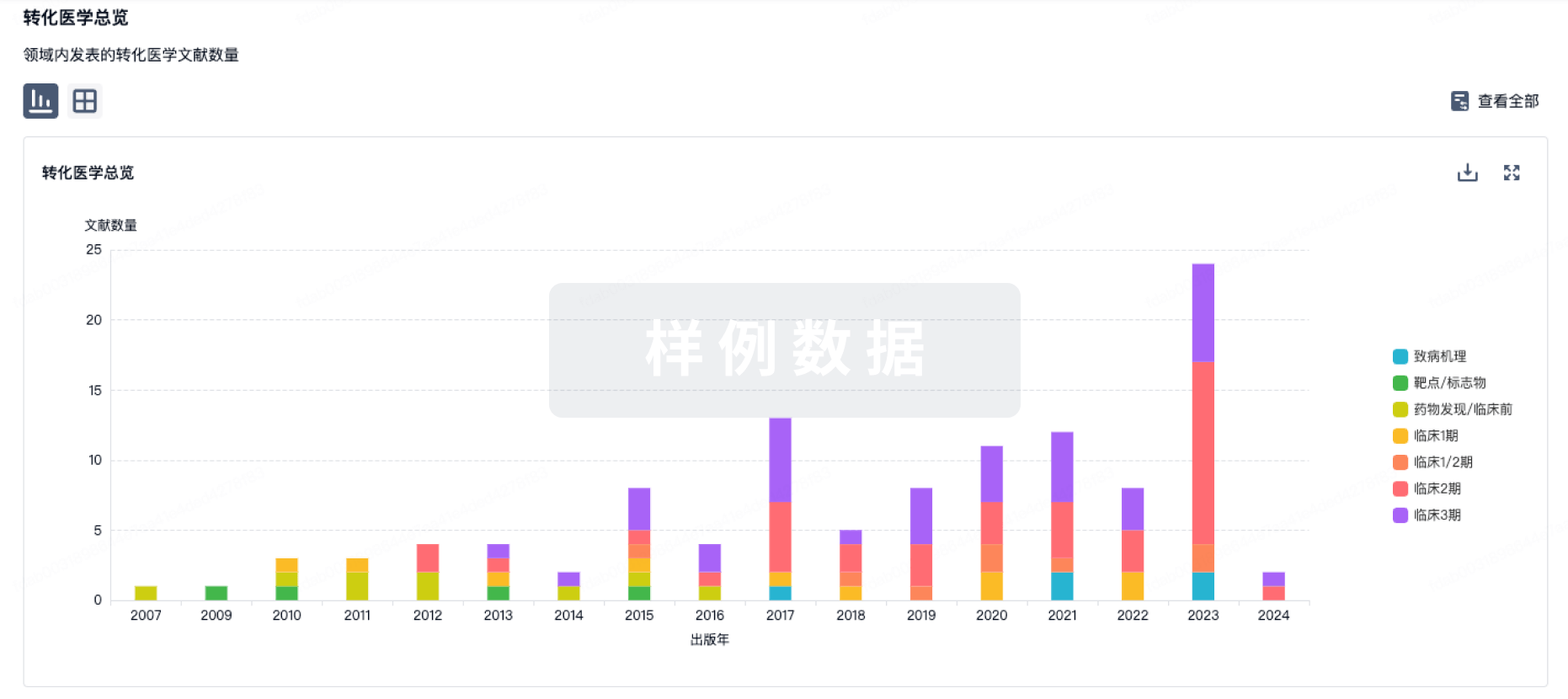
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
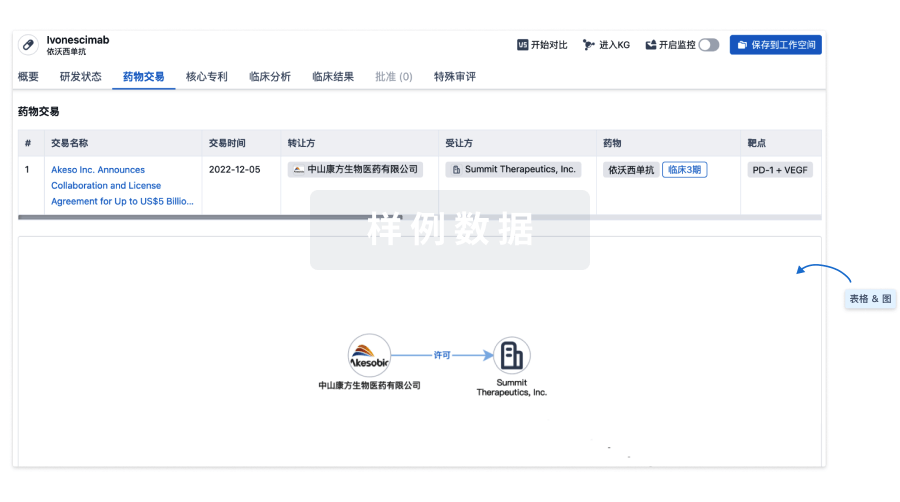
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
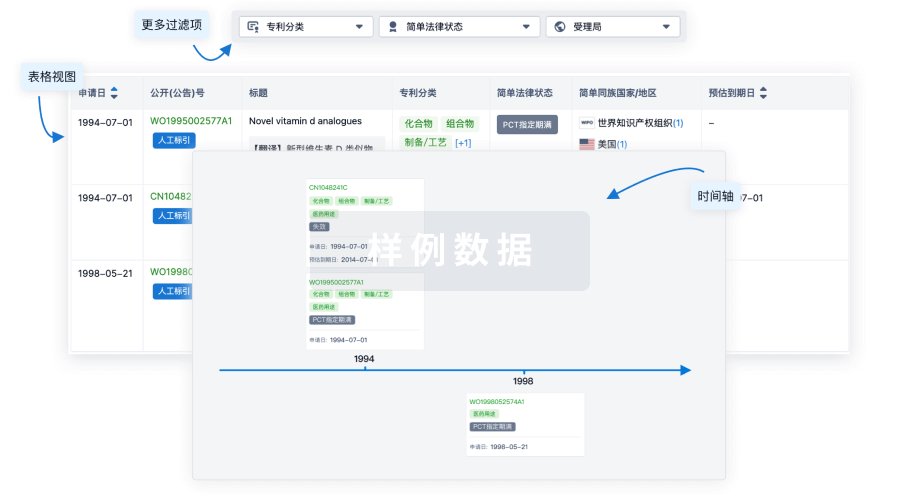
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
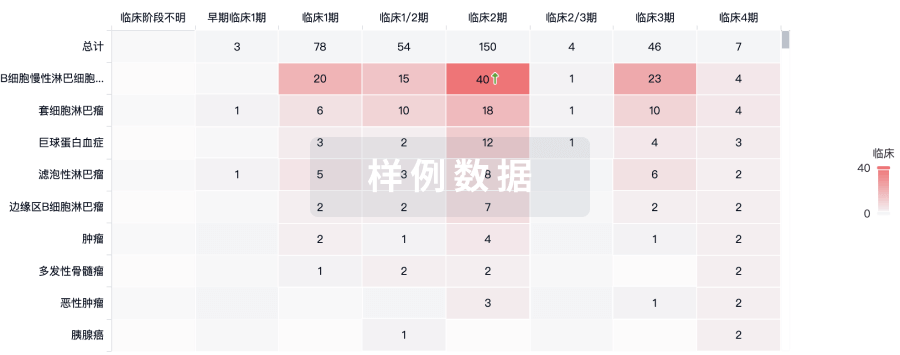
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
