Phase 1 Clinical Trial to Evaluate the Safety of FURESTEM-CD Inj. in Patients With Moderate to Severe Plaque-type Psoriasis.
Phase I clinical trial to evaluate safety of FURESTEM-CD Inj. in patients with moderate to severe in plaque-type psoriasis injection for 4weeks.
Extension Study, An Open-labelled, Multi Center, Comparative Phase I/IIa Clinical Trials to Evaluate the Efficacy and Safety of FURESTEM-CD Inj. in Patients With Moderately Active Crohn's Disease
The purpose of this clinical trial is to evaluate long-term safety and efficacy in patients who has history of FURESTEM-CD injection at least once.
An Open-labelled, Multi Center, Comparative Phase I/IIa Clinical Trial to Evaluate the Efficacy and Safety of FURESTEM-CD Inj. in Patients With Moderately Active Crohn's Disease
The purpose of phase I clinical trial is to evaluate safety and efficacy in subjects with moderately active Crohn's disease after injection for 28days.
The purpose of phase IIa clinical trial is to determine therapeutic safety and efficacy of FURESTEM-CD Inj. which is based on Crohn's disease activity index(CDAI)<150 after injection.
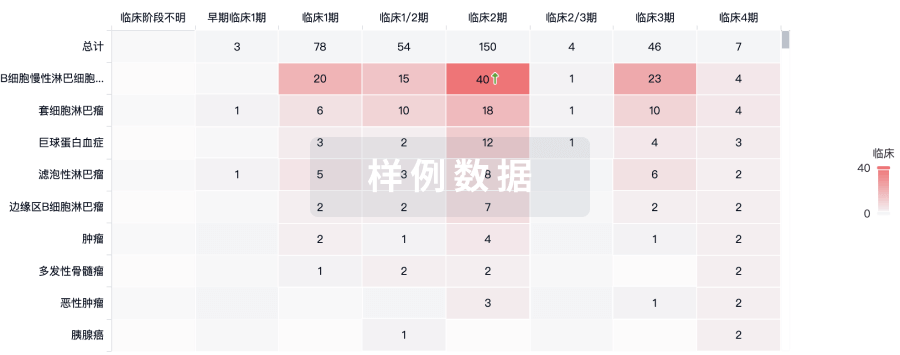
100 项与 Furestem-CD 相关的临床结果
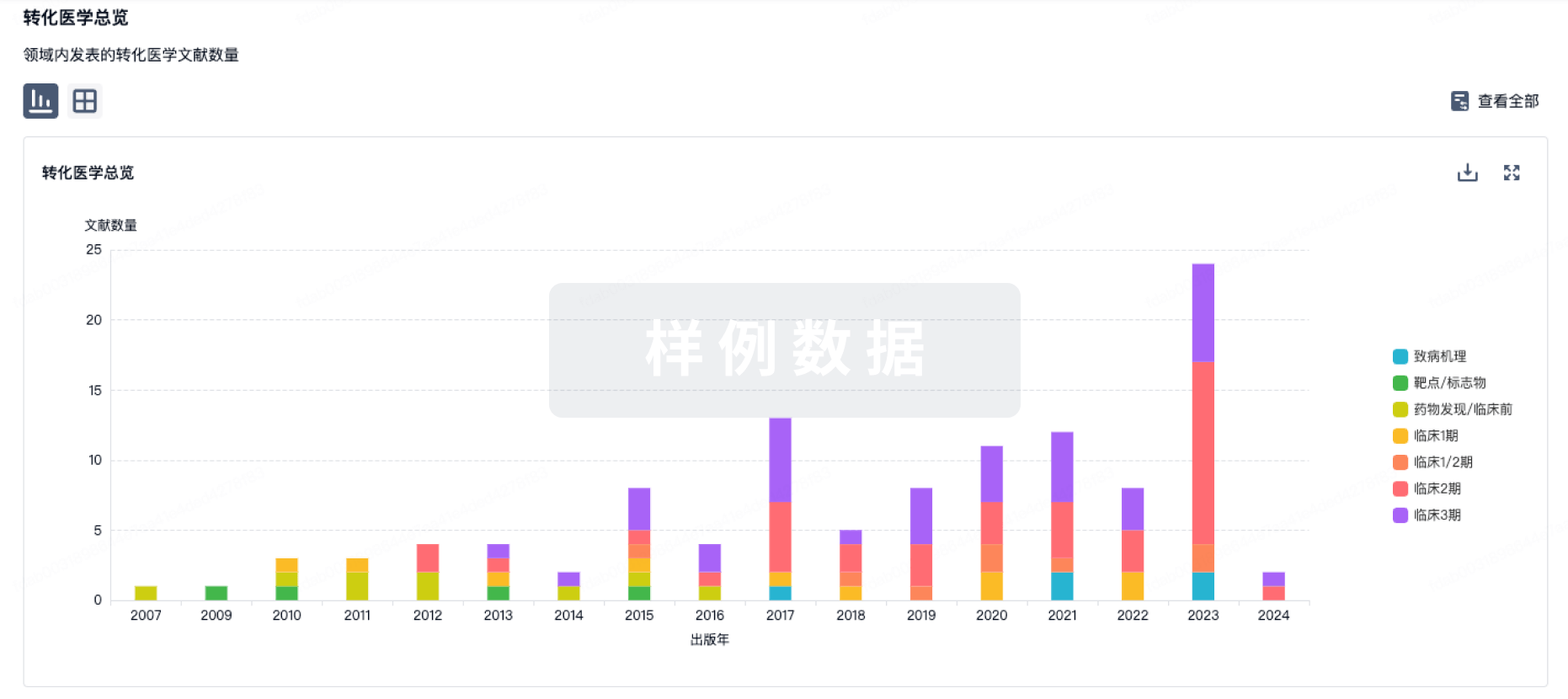
100 项与 Furestem-CD 相关的转化医学
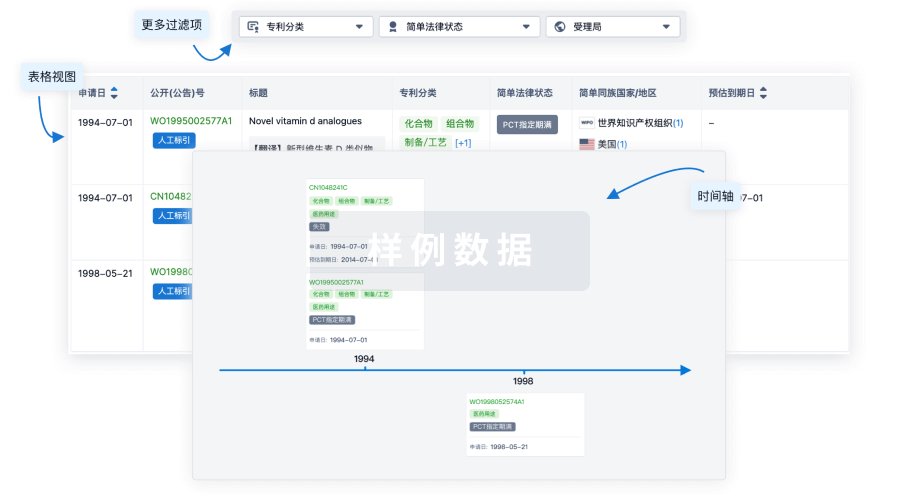
100 项与 Furestem-CD 相关的专利(医药)
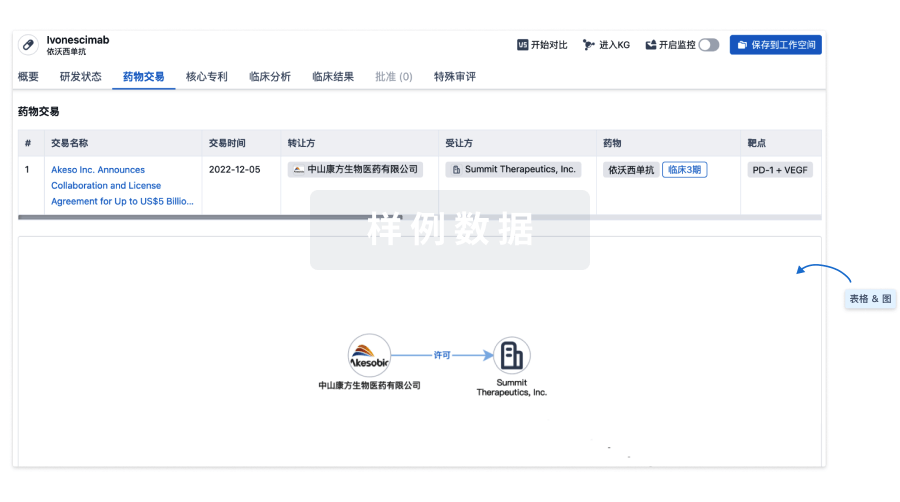
100 项与 Furestem-CD 相关的药物交易