预约演示
更新于:2025-04-15
AMG-595
更新于:2025-04-15
概要
基本信息
原研机构 |
在研机构- |
非在研机构 |
最高研发阶段无进展临床1期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
结构/序列
使用我们的ADC技术数据为新药研发加速。
登录
或

关联
1
项与 AMG-595 相关的临床试验NCT01475006
A Phase 1 First-in-Human Study Evaluating Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of AMG 595 in Subjects With Recurrent Malignant Glioma Expressing Mutant Epidermal Growth Factor Receptor Variant III (EGFRvIII)
This is an open-label, sequential dose exploration study of single agent AMG 595 administered in subjects with recurrent glioblastoma multiforme (GBM) and/or anaplastic astrocytomas (AA). The purpose of the study is to evaluate safety, tolerability, and pharmacokinetics (PK) of AMG 595, and also to evaluate the objective response rate in subjects receiving AMG 595. This study will be conducted in two parts. Part 1 will explore doses of AMG 595 in subjects with recurrent GBM and/or AA. Part 2 (dose expansion) will examine the MTD established in Part 1 in subjects with recurrent GBM.
开始日期2012-02-01 |
申办/合作机构 |
100 项与 AMG-595 相关的临床结果
登录后查看更多信息
100 项与 AMG-595 相关的转化医学
登录后查看更多信息
100 项与 AMG-595 相关的专利(医药)
登录后查看更多信息
2
项与 AMG-595 相关的文献(医药)2019-08-01·Cancer chemotherapy and pharmacology4区 · 医学
Safety, tolerability, and pharmacokinetics of anti-EGFRvIII antibody–drug conjugate AMG 595 in patients with recurrent malignant glioma expressing EGFRvIII
4区 · 医学
Article
作者: Reardon, David A ; Rosenthal, Mark ; Cloughesy, Timothy ; Henary, Haby A ; Upreti, Vijay V ; Curry, Richard ; Hill, John S ; Rasmussen, Erik ; Damore, Michael A
PURPOSE:
Epidermal growth factor receptor variant III (EGFRvIII) is expressed in a significant percentage of primary and recurrent glioblastoma (GBM), a common malignant primary brain tumor in adults. AMG 595 is an antibody-drug conjugate comprising a fully human, anti-EGFRvIII monoclonal antibody linked to DM1. The study goals were to assess safety, tolerability, and pharmacokinetics of AMG 595 in GBM.
METHODS:
In this phase 1, first-in-human, open-label, sequential-dose, exploration study, adults with recurrent GBM received AMG 595 once every 3 weeks (Q3W) according to incremental dosing cohorts (0.5-3.0 mg/kg). Primary endpoints were to assess safety, the incidence of dose-limiting toxicities (DLTs), objective response (per Macdonald criteria), evaluate pharmacokinetics, and estimate the maximum tolerated dose (MTD).
RESULTS:
Of 382 patients screened, 32 were enrolled and received ≥ 1 dose of AMG 595. Ten patients experienced 18 DLTs (all grade 4 thrombocytopenia), and the MTD was 2.0 mg/kg. Twenty-eight patients (88%) experienced ≥ 1 treatment-related adverse event (AE); the most common AEs were thrombocytopenia (50%) and fatigue (25%). Grade ≥ 3 treatment-related AEs occurred in 17 patients (53%); 11 (34%) had serious treatment-emergent AEs, and none were considered treatment related. Pharmacokinetic profiles indicated low levels of circulating unconjugated antibody and cytotoxin, dose-proportional increases in plasma exposures for the conjugated antibody over the studied range, and less than twofold accumulation following multiple Q3W dosing. Two patients (6%) had partial responses; 15 (47%) had stable disease.
CONCLUSIONS:
AMG 595 exhibited favorable pharmacokinetics and is a unique therapy with possible benefit for some patients with EGFRvIII-mutated GBM with limited therapeutic options.
2015-07-01·Molecular cancer therapeutics2区 · 医学
AMG 595, an Anti-EGFRvIII Antibody–Drug Conjugate, Induces Potent Antitumor Activity against EGFRvIII-Expressing Glioblastoma
2区 · 医学
Article
作者: Twomey, Brian ; Chen, Qing ; Hamblett, Kevin J. ; Meininger, David ; Arora, Taruna ; Trueblood, Esther S. ; Hill, John S. ; Liu, Hua ; Chang, Wesley S. ; Foltz, Ian N. ; Vonderfecht, Steven L. ; Fanslow, William C. ; Kozlosky, Carl J. ; Siu, Sophia
Abstract:
Epidermal growth factor receptor variant III (EGFRvIII) is a cancer-specific deletion mutant observed in approximately 25% to 50% of glioblastoma multiforme (GBM) patients. An antibody drug conjugate, AMG 595, composed of the maytansinoid DM1 attached to a highly selective anti-EGFRvIII antibody via a noncleavable linker, was developed to treat EGFRvIII-positive GBM patients. AMG 595 binds to the cell surface and internalizes into the endo-lysosomal pathway of EGFRvIII-expressing cells. Incubation of AMG 595 with U251 cells expressing EGFRvIII led to potent growth inhibition. AMG 595 treatment induced significant tumor mitotic arrest, as measured by phospho-histone H3, in GBM subcutaneous xenografts expressing EGFRvIII. A single intravenous injection of AMG 595 at 17 mg/kg (250 μg DM1/kg) generated complete tumor regression in the U251vIII subcutaneous xenograft model. AMG 595 mediated tumor regression in the D317 subcutaneous xenograft model that endogenously expresses EGFRvIII. Finally, AMG 595 treatment inhibited the growth of D317 xenografts orthotopically implanted into the brain as determined by magnetic resonance imaging. These results demonstrate that AMG 595 is a promising candidate to evaluate in EGFRvIII-expressing GBM patients. Mol Cancer Ther; 14(7); 1614–24. ©2015 AACR.
4
项与 AMG-595 相关的新闻(医药)2025-02-27
往期回顾(点击标题查看文章)
2023年抗体药物行业发展大盘点
单抗发展史,看这篇就够了!
浅谈抗体的作用机制和优缺点
单抗怎么起名,看这篇!
“魔法子弹”ADC之结构与作用
7大亮点回顾2024全球ADC产业图景
在ADC药物研发中,靶点的选择是决定药物疗效和安全性的关键因素。理想的靶点通常在肿瘤细胞中特异性高表达,而在正常细胞中的表达水平较低,以减少对正常组织的损害。此外,靶点的选择还应考虑其在肿瘤发展中的作用,以及是否具有内吞性,这决定了ADC药物能否被肿瘤细胞有效内化并释放细胞毒性载荷。
目前,当前ADC药物研发的“明星靶点”包括HER2、TROP2、组织因子(TF)、Nectin-4、FRα、EGFR、BCMA、CD家族(CD33、CD30、CD22、CD79B、CD19)等。
表 全球ADC药物上市汇总
实体瘤ADC热门靶点介绍
用于治疗实体瘤的ADC热门靶点包括HER2、TROP2、组织因子(TF)、Nectin-4、FRα和EGFR。从ADC产品的靶点分布来看,HER2是ADC研发最热门的靶点,其次是TROP2。
01
HER2
HER2是一种185kda的跨膜糖蛋白,属于表皮生长因子受体家族的一员。人表皮生长因子受体家族(human epidermal growth factor receptor,HER)承担着信号传导,调节正常细胞的生长、存活、转化及凋亡的作用,HER家族包括4个成员,分别是HER1(EGFR/ErbB1 )、HER2(ErbB2)、HER3(ErbB3)、HER4(ErbB4)。HER2/NEU基因的扩增是人类恶性肿瘤和转移的重要因素。
目前,市场上有三种靶向HER2的ADC药物,分别是T-DM1, DS-8201和RC48。
靶向HER2的ADC药物
1、T-DM1
T-DM1(kadcyla,Ado-trastuzumab emtansine,赫赛莱)是第一个获得FDA批准用于乳腺癌的ADCs药物,携带的单抗靶头是靶向HER2的单抗——曲妥珠单抗(赫赛汀),HER2广泛用于胃癌、乳腺癌细胞;化疗药物是微管蛋白抑制剂小分子抑制剂细胞毒素DM1(Mertansine,美坦新,美登素类),以不可水解的硫醚键为连接子进行连接。TDM1结合到HER2受体的第IV亚区,由受体介导的内化,随后在溶酶体降解,在细胞内释放含有DM1。DM1与微管蛋白的结合破坏细胞内的微管网络,导致细胞周期阻滞和细胞凋亡。
2、DS-8201
DS-8201(Enhertu,fam-trastuzumab deruxtecan-nxki,优赫得),由第一三共和阿斯利康推出。DS-8201携带靶头仍然是曲妥珠单抗(赫赛汀),而细胞毒性药物则是喜树碱类药物,即拓扑异构酶1抑制剂Exatecan(依喜替康)衍生物,并以可水解的四肽连接子,采用半胱氨酸残基的定点偶联,载药量大。目前DS-8201已被NCCN指南列入HER2阳性晚期乳腺癌推荐治疗方案,并在全球不同地区获批上市7种适应症。
3、RC48
在国内,NMPA于2021年6月9日,附条件批准荣昌生物注射用RC48(Disitamab- MMAE,爱地希)上市。RC48没有采用曲妥珠单抗,而是采用自研的人源化维迪西妥单抗(disitamab vedotin)靶向HER2的胞外区,以可裂解的val-cit为连接子,连接了抑制微管蛋白剂单甲基澳瑞他汀E(Monomethyl Auristatin E, MMAE,海兔毒素衍生物),用于局部晚期或转移性胃癌/胃食管交界(GEJ)腺癌患者的三线治疗。
02
TROP-2
TROP-2(滋养层细胞表面抗原2)是一种在多种肿瘤组织中高表达的跨膜糖蛋白,其在肿瘤的发生、发展及转移中发挥重要作用,从而被认为是癌症靶向治疗的一个新型且有前途的分子靶点。
目前,已有多种TROP-2 ADC药物在临床研究中显示出显著的抗肿瘤效果。例如,戈沙妥珠单抗(Sacituzumab Govitecan,SG)由人源化的抗 Trop-2 抗体(hRS7)与拓扑异构酶抑制剂(SN-38)结合而成,已获得美国FDA批准用于治疗三阴性乳腺癌(TNBC)的TROP-2 ADC药物。
03
HER3
相比于拥挤的HER2赛道,HER3由于受体本身结合力较低,不具备内在激酶活性,且没有合适的反映HER3激活与否的生物标志物,在研发中没有受到重视,但现在也有许多企业在跃跃欲试。
如第一三共开发的靶向HER3的ADC药物U3-1402,HER3-DXd(Patritumab Deruxtecan,U3-1402)是一种新型的HER3靶向ADC,其是将靶向HER3单克隆抗体patritumab与拓扑异构酶I抑制剂DXd结合构建。目前U3-1402已被美国FDA授予突破性疗法资格,用于接受过第三代EGFR靶向药和铂类化疗后疾病进展的转移性或局部晚期EGFR阳性非小细胞肺癌患者。
04
EGFR
在EGFR(HER1)的赛道上,Amgen公司的AMG595是人源化的抗EGFR-Ⅷ IgG1单克隆抗体与DM1偶联而成的ADCs,主要用于治疗恶性胶质瘤。但临床研究显示AMG595对一些EGFRvIII突变GBM患者有益,但作用有限。此外,艾伯维的ABT-414也在GBM三期临床中未达到主要终点。
国内EGFR的ADC药物如乐普生物的MRG003靶向EGFR,以MMAE为载荷,以可裂解连接子连接,用于治疗晚期实体肿如头颈癌、鼻咽癌和非小细胞肺癌的研究正在进行中。此外还有恒瑞医药(SHR-A1307)、荣昌生物(RC68)、百力司康(BB-1705 )等多家生物技术公司布局靶向EGFR的ADCs。
05
TF(组织因子)
TF(组织因子),也称为凝血活酶因子III 或CD142,是一种具有促凝血活性的跨膜糖蛋白,该抗原的内化特性非常适合开发TF靶向 ADC。首款用于宫颈癌治疗的ADC,由Seagen和Genmab公司合作开发的Tivdak® (tisotumab vedotin),于2021年取得加速批准在美国上市。它是首个批准用于宫颈癌的ADC,也是目前唯一一款靶向组织因子(TF)的上市ADC。
06
Nectin-4
Nectin-4是一种66kDa的 I 型跨膜蛋白,其主要作用是促进细胞间接触。它在多种肿瘤类型中过表达,而在正常成人组织中几乎不存在,因此也被视为ADC药物的理想靶点。目前,上市的Nectin-4 ADC药物是Padcev(Enfortumab Vedotin),由Seagen和安斯泰来联合开发。
07
FRα
FRα(叶酸受体α)是一种膜结合的代谢叶酸受体,参与叶酸的细胞内转运。一旦与叶酸结合,受体-配体复合物就会通过非经典的内吞机制内化。FRα在卵巢癌、乳腺癌、子宫内膜癌、间皮瘤和肺癌中高表达,但在正常细胞中几乎不表达,这使得该受体非常适合 ADC 靶向。
目前,全球首款且唯一获批的FRα的ADC药物是ImmunoGen公司开发的Elahere(Mirvetuximab Soravtansine),它由抗FRα的人源化单克隆抗体与细胞毒性药物分子DM4通过二硫键偶联,属于靶向FRα的First-in-class ADC。Elahere的作用机制是与肿瘤细胞表面的FRα结合,并通过内吞效应进入到肿瘤细胞中,然后连接键断裂,释放抗微管药物DM4,继而杀伤肿瘤细胞。
08
Claudin18.2
Claudin 18.2是一种紧密连接蛋白家族成员,在多种肿瘤中高表达,目前Claudin 18.2作为极具前景的肿瘤靶向治疗靶点。例如,恒瑞医药开发的SHR-A1904是一款拥有知识产权的Claudin18.2靶向ADC药物,其有效载荷是拓扑异构酶抑制剂(TOPOi),通过与肿瘤细胞表面的靶抗原结合,使得药物被内吞进入细胞后释放小分子毒素杀伤肿瘤细胞。
2025年1月6日,中山大学肿瘤防治中心徐瑞华教授团队在国际权威肿瘤学期刊《柳叶刀·肿瘤》(The Lancet Oncology)上公布了一项具有里程碑意义的临床研究:靶向Claudin 18.2的抗体偶联药物(CMG901)治疗晚期胃癌/胃食管结合部腺癌患者的I期临床试验结果。
作为全球首个针对Claudin 18.2靶点的抗体偶联药物临床研究报道,国内原研的CMG901在晚期胃癌/胃食管结合部腺癌患者中患者中展现的良好安全性与显著疗效,标志着中国在抗肿瘤新药研发领域取得了又一重大突破。
09
c-Met
c-MET,亦称为MET或肝细胞生长因子受体(HGFR),是一种属于MET家族的受体酪氨酸激酶,在多种肿瘤中高表达。
在研c-Met ADC药物
艾伯维ABBV-399和ABBV-400
2024年9月,艾伯维宣布ABBV-399在美申报上市,用于治疗既往接受过治疗的c-Met蛋白过表达、表皮生长因子受体(EGFR)野生型晚期/转移性非鳞状非小细胞肺癌(NSCLC)成人患者。
阿斯利康AZD9592
荣昌生物RC108
再生元REGN5093- M114
恒瑞SHR-A1403
血液瘤ADC靶点介绍
对于血液肿瘤,免疫谱系特异性生物标志物如CD19、CD20、CD22、CD33、CD79B和BCMA在恶性血细胞上广泛且均匀地高水平表达,因此已被广泛探索为ADC开发的候选靶点。此外,获批ADC的靶抗原在结合后容易内化,这是有助于ADC疗效的重要特征。
01
CD19
CD19被认为是一种泛B细胞标志物,也是成熟B细胞表面多分子复合物的主要信号转导成分。CD19的表达在大多数B细胞恶性肿瘤中高度保守,此外,CD19具有快速内化动力学,不会脱落到循环中,使其成为理想的ADC靶抗原。
02
CD22
CD22是一种140kDa的跨膜糖蛋白,与CD33一样,是Siglec家族的成员,与该家族具有多种结构特征。主要区别在于CD22比CD33大得多,因为它具有多个Ig结构域和ITIM/ITIM样基序。CD22的表达仅限于B细胞,并且CD22在包括ALL在内的各种B细胞恶性肿瘤的大多数母细胞中表达水平升高。
CD22被CME内吞。天然样配体通过CD22的组成型快速内吞作用在细胞内积累。这些配体被分选以在溶酶体中降解,而CD22则被回收回细胞表面。此外,CD22配体诱导的内吞作用激活细胞内池,补充或增加细胞表面CD22的表达水平。因此,CD22对ADC具有良好的内吞特性。
03
CD33
CD33是一种67kda的跨膜糖蛋白受体,是唾液酸结合免疫球蛋白样凝集素(SIGLEC)家族的成员,该家族通常在正常髓系细胞上表达,并且是Gemtuzumab ozogamicin的靶标,因为它优先在AML细胞上过表达。CD33的免疫受体酪氨酸抑制基序(ITIM)调节CD33内吞作用,可通过网格蛋白介导的内吞作用(CME)激活。在内吞作用方面,AML细胞中CD33的表达水平与其内吞速率之间没有相关性。CD33是一种缓慢内化的抗原,CD33交联不能改善内吞作用。对GO无反应的AML患者可能与CD33受体摄入功能不佳有关。
04
CD79b
CD79b仅在未成熟和成熟B细胞中表达,在恶性肿瘤中≥80%的B细胞中过表达。CD79a和CD79b是两种非共价结合的跨膜蛋白,介导信号传导和内吞作用。对于后者,CD79a-CD79b异二聚体是控制BCR内吞作用的支架。BCR的内吞作用主要由CME完成,并由AP-2介导。有趣的是,CD79a直接与AP-2的μ亚基相互作用,进而激活CD79b并导致整个BCR复合物的内吞作用。
此外,对于ADC,CD79a可以内化为单体,但CD79b不能。如果CD79b的近端膜酪氨酸(Y195)发生突变,则AP-2与CD79a的结合被阻断,内吞作用也被阻断。在18%的活化B细胞样DLBCL标本中,Y195发生突变。总之,有证据表明CD79b的内吞作用取决于整个BCR复合物的内化,而不是作为单体。
05
BCMA
BCMA或CD269,也称为TNFR超家族成员17,转导诱导B细胞存活和增殖的信号。BCMA的分子量仅为20.2kDa,其配体结合的细胞外区域具有“扶手椅”构象,由六个CRDS组成。除多发性骨髓瘤外,BCMA还在许多血液系统恶性肿瘤中表达,例如霍奇金淋巴瘤和非霍奇金淋巴瘤。
靶点研发的挑战与机遇
尽管上述靶点已成为ADC药物研发的热点,但仍面临诸多挑战,如靶点的同质化竞争、耐药性问题、以及对正常组织的潜在毒性。同时,这些挑战也带来了新的机遇,如开发新的靶点、探索联合用药策略、以及利用新技术提高ADC药物的特异性和疗效。
同质化竞争:随着多个药企聚焦于相同靶点,市场竞争日益激烈,这要求研发者探索新的靶点或开发差异化的产品。
耐药性问题:肿瘤可能对ADC药物产生耐药性,因此需要研究新的有效载荷和连接子技术,以克服耐药性。
对正常组织的潜在毒性:尽管ADC药物旨在减少对正常组织的损害,但某些靶点在正常组织中的表达可能导致不良反应,需要进一步优化药物设计以降低毒性。
小结
ADC药物研发的热门靶点不仅为癌症治疗开辟了新的方向,也推动了药物研发技术的革新与进步。尽管耐药性和市场竞争是挑战,但通过联合用药、适应症拓展等策略,ADC药物的潜力正在被不断挖掘!
参考来源:1、收藏!实体瘤和血液肿瘤的热门ADC靶点总结https://mp.weixin.qq.com/s/TyWFhawME7rngqavLwhC4w2、ADC药物研发的热点靶点与未来趋势https://mp.weixin.qq.com/s/s4ychEG-SoJy3s2Ce5Vqcw3、靶向HER家族的ADCs药物概览https://mp.weixin.qq.com/s/FgWfzopNgYX3FTYqfNHM4Q
END
整理及编辑:芝麻核桃
声明:本文仅代表作者个人观点,不代表任何组织及本公众号立场,如有不当之处,敬请指正。如需转载,请注明作者及来源:蒲公英Biopharma。
皮尔法伯集团创立于1962年,总部位于法国西南部,拥有世界第二大护肤品及化妆品实验室,也是全法第二大私营制药集团。集团业务涵盖处方药、医学护肤品及公共健康三个领域。
皮尔法伯制药作为集团重点业务板块之一,聚焦肿瘤,罕见病和皮肤科三大领域的研发和对外合作。集团在肿瘤学领域拥有40年的创新、开发、制造和商业化经验。集团每年约80%的研发支出用于肿瘤学,目前的肿瘤学商业产品组合涵盖结直肠癌、乳腺癌、肺癌、黑色素瘤、血液学疾病和癌前皮肤病。
抗体药物偶联物临床1期申请上市临床2期CSCO会议
2024-08-26
摘要:抗体-药物偶联物(ADCs)代表了一类新型的癌症治疗药物。它们的设计包括一个针对肿瘤的特异性抗体、一个连接子和一个细胞毒性有效载荷。它们的设计目的是允许高度有效的细胞毒性药物特异性地靶向肿瘤细胞,同时避免对正常细胞的损害。已有报告称,ADC的任一组成部分都可能驱动常见的毒性。目前有50多个ADC正在进行积极的临床开发,另有约20个已被终止。在这篇综述中,分析了ADC的报告毒性,并详细探讨了它们的作用机制。讨论了减少毒性的方法,包括剂量策略和药物设计。对活跃和已终止药物报告的毒性很重要,以推动合理设计并提高未来ADC的治疗指数。
1.引言
抗体-药物偶联物(ADCs)是肿瘤治疗领域的新成员。目前,已有3种药物获得许可:吉妥单抗-奥佐加米辛(CMA-676, Mylotarg®),除了日本外,已在所有地区撤回;本妥昔单抗-维多汀(BV; SGN-35, Adcetris®)和阿多-曲妥珠单抗-埃姆坦辛(T-DM1, Kadcyla®)。然而,ADC 开发领域正在迅速扩展,约有55种活跃的临床化合物,约20种已终止临床试验,还有更多在临床前阶段显示出前景。ADC 的设计旨在利用抗体靶标的特异性与化疗药物的细胞毒性,因此应表现出高度特异性和低毒性。这一理想在实践中难以实现,因为ADC反复诱发限制这些化合物治疗窗口或阻碍进一步临床开发的毒性。有许多ADC在早期临床前阶段显示出前景并短暂进入临床试验,但未能取得进展或突然终止。这些终止的原因多种多样,包括财务和战略原因,但也有一些可能因不可预见或不可接受的毒性而失败。重要的是,这些失败化合物的结果需要公开,以便ADC研究和开发不再重复过去的错误。
在这篇综述中,确定了35种活跃和已终止化合物的报告毒性,并全面评估了ADC的每个组成部分对所展示毒性的作用。正如预期的那样,有效载荷通常驱动毒性效应;然而,靶标和连接子可能决定毒性的器官特异性,这将在详细讨论。了解每个组成部分在ADC中的作用将有助于通过提高疗效和降低毒性来开发具有改善治疗指数的ADC。设计ADC的新技术正在推动下一代进入临床。随着ADC设计领域的发展,评估过去ADC的毒性将变得越来越重要,以便设计未来的ADC。
2.ADC设计
ADC由三个明确定义的组成部分组成:抗体、连接子和有效载荷,这些已经在 Sievers 等人的综述中进行了全面回顾。一个理想的ADC要实现最大疗效和最小毒性,需要适当组合这三个组成部分。
2.1.抗体
在毒性的背景下,抗体需要准确地高效地结合到肿瘤细胞上的抗原,同时避免正常细胞。因此,非常重要的是抗体结合到一个合适的靶标抗原,该抗原优先或仅在肿瘤细胞上表达。抗体可能不适用的原因包括不适当或低亲和力结合、肿瘤细胞上抗原表达不足或结合后缺乏内化。然而,一些ADC可能不需要内化就能释放有毒有效载荷。正如CEACAM5被认为不内化,Immunomedics 抗CEACAM5的 labetuzumab govitecan (IMMU-130) 可能在抗体结合后释放有效载荷,无需内化。CEACAM5 也是 SAR408701 的靶标,该药正在进行1期研究。
不适当的结合可能是由于靶标抗原在正常细胞上的表达或对 Fc 受体(FcR)或凝集素受体,如甘露糖受体的非特异性结合。此外,具有特定生物学效应的抗体如果非特异性或不适当地结合到正常细胞上的靶标抗原,可能会引起毒性效应,而与有效载荷的传递无关。事实上,MEDI-547,一种针对 EphA2 的 ADC,与 MMAE 结合,在非常低的剂量下表现出毒性,这可能表明是抗体介导的生物学效应而不是细胞毒性有效载荷的传递。
2.2.连接子
连接子化学对于及时从抗体释放有效载荷非常重要。像抗体一样,有效的连接子将通过确保准确释放有效载荷来提高ADC的治疗指数。虽然连接子本身似乎不会引起毒性,但连接子的稳定性对随后由有效载荷施加的毒性有相当大的影响。更稳定的连接子将以目标特异性方式释放其有效载荷,引起更特定的毒性,而不太稳定的连接子更有可能经历非特异性裂解,导致更广泛的毒性概况。增加连接子的空间位阻(例如,SPDB-DM4 连接子有效载荷结构)可以提高 ADC 在循环中的稳定性,目标是减少非特异性毒性。可裂解连接子主要通过各种机制在内体或溶酶体室中从有效载荷裂解,包括酸性降解(腙);通过组织蛋白酶 B 裂解的蛋白酶裂解(二肽);巯基-二硫交换反应(二硫键,碳酸盐)。相反,不可裂解连接子(MC 和 SMCC)需要完全的溶酶体蛋白水解抗体,生成带有带电赖氨酸或半胱氨酸的有毒有效载荷(表1)。
2.3.有效载荷
ADC中使用的有效载荷是高度有效的细胞毒素,对生存所需的关键细胞过程产生影响(表1)。大多数当前临床测试中的化合物使用 either maytansine derivatives (DM1/DM4) or auristatins (MMAE/MMAF),它们都是微管抑制剂。这些通常通过在 G2/M 期引起细胞周期停滞,诱导正在有丝分裂的细胞凋亡。最近的工作表明,微管抑制剂也可能扰乱间期的非分裂细胞。这些发现为微管抑制剂如何对缓慢复制或非分裂的肿瘤细胞具有细胞毒性提供了解释。
ADC中使用的其他类别的细胞毒素包括 enediynes (calicheamicin), duocarmycin derivatives, pyrrolobenzo diazepines (PBDs) 和 indolinobenzodiazepines,所有这些针对 DNA 的小沟,以及 quinoline alkaloids (SN-38),它抑制拓扑异构酶 I。
ADC中使用的大多数有效载荷非常有效,通常在皮摩尔范围内具有细胞毒性,这是必要的,因为只有非常少的量(<1%)的注射剂量的抗体定位到肿瘤。然而,正是这种效力驱动了 ADC 的毒性,导致大多数毒性被有效载荷的类别所表征。
2.4.考虑其他组成部分
ADCs 的结合过程导致具有多个药物-抗体比率的异质性构建物群体。事实上,第一个获得 FDA 批准的 ADC,吉妥单抗-奥佐加米辛,平均 DAR 为 2-3,但几乎一半的抗体没有药物附加。每个抗体结合的细胞毒素分子数量的变化可能对 ADC 的处置产生显著影响,对毒性有影响。
结合位点是目前正在进行的密集审查领域,随着对这种方式作为控制特定药代动力学(PK)概况的增加,人们越来越重视。将 ADC 工程化以在已知位点上包含每个抗体的特定数量的有效载荷分子,将产生一个同质的 ADC 群体,这可能导致更好的治疗窗口(增加疗效 vs. 降低毒性)。目前正在研究的 ADC 特异性结合技术包括使用工程化半胱氨酸、非天然氨基酸和添加硒代半胱氨酸、谷氨酰胺或醛标签。添加一个工程化半胱氨酸位点以产生工程化硫(THIOMAB)抗体结合物结果是每个抗体大约有 2 个药物的 ADC。有几种使用这种技术的 ADC 正在进行临床前测试。
唯一在临床中披露使用特异性结合技术的 ADC 是 SGN-CD33A 和 SGN-CD70A,它们使用工程化半胱氨酸将每个抗体特异性结合 2 个 PBD 二聚体。
随着 ADC 的发展,越来越明显的是,没有一个 ADC 的组成部分将驱动疗效或确实毒性。相反,每个都有一定的作用,肿瘤和靶标抗原的生物学也可能影响 ADC 的活性。提高 ADC 的治疗指数需要优化每个组成部分与其他部分的结合,以产生针对特定疾病适应症的最佳 ADC。
3.毒性机制
ADCs 的毒性可能由药物的任何组成部分介导。靶标抗原在正常细胞上的低水平表达可能导致特定的毒性,而连接子的早期裂解释放游离药物可能产生更广泛的毒性。ADC 可能通过 Fc 和甘露糖受体结合诱导毒性的其他方式。大多数 ADC 毒性被认为源自有效载荷。正常的快速分裂细胞因微管抑制剂而面临毒性风险,因为它们对快速增殖的细胞产生细胞毒性作用。通常受标准化疗和 ADCs 影响的正常细胞包括消化道内衬细胞,导致胃肠道症状;毛囊中的细胞,导致脱发;以及骨髓细胞,导致骨髓抑制。不同有效载荷发现了一些关键毒性(表2)。特别是,MMAE 诱导周围神经病变和中性粒细胞减少症;MMAF 与血小板减少症和眼部毒性相关;DM1 根据连接子和随后的代谢产物,导致胃肠道效应以及血小板减少症和中性粒细胞减少症;DM4 偶联的 ADCs 最常见的不良事件是眼部毒性;卡利霉素导致血小板减少症和肝功能障碍;来自 SN-38 偶联药物的早期迹象表明中性粒细胞减少症是一种常见的毒性。
ADCs 如何以及为什么会对不同细胞类型产生这些毒性,在试图最小化未来药物开发的毒性时具有相当大的兴趣,一些已知因素在下面进行了讨论。
3.1.血小板减少症
血小板减少症可能是由于血小板的增强破坏或减少产生引起的。它表现为增加的瘀伤和出血(牙龈和鼻出血),在严重情况下可能导致粘膜出血。ADCs 诱导的血小板减少症被认为是由于抑制巨核细胞分化和巨核细胞前体的凋亡。它是利用卡利霉素的 ADCs 的关键毒性,以及使用不可裂解连接子的强大微管作用剂。在 MMAF 和 DM1 结合的 ADCs 的情况下,毒性是通过毒性代谢产物 cys-mc-MMAF 或 lys-SMCC-DM1 分别介导的。在接受 MMAF ADCs AGS-16M8F,SGN-75,和 ABT-414,治疗的患者中,血小板减少症(所有等级)的报告分别为32%、26%和11%。在 SGN-75 试验中,没有累积效应随着重复周期。在 SGN-CD19A 的情况下,3级血小板减少症的发病率约为10%。血小板减少症对于含有卡利霉素的 ADCs 比含有 MMAF 的更为普遍。在非霍奇金淋巴瘤(NHL)患者中进行的 inotuzumab ozogamicin 的2期试验中,77%的患者经历了血小板减少症,其中53%为3级,并导致22%的患者停止治疗。恢复是可变的,但在大多数患者中,3个月后治疗后降低到1级或得到解决。同时,几乎所有(99%)接受吉妥单抗-奥佐加米辛治疗的急性髓性白血病(AML)患者都经历了3级血小板减少症,15%的患者经历了3级出血。来自 PF06647263 的首次人体研究的早期结果表明,33%的患者经历了血小板减少症。
此外,血小板减少症已报告为 DM1 结合物 ado-trastuzumab emtansine 和 lorvotuzumab mertansine。有趣的是,接受 ado-trastuzumab emtansine 治疗后血小板减少症的发病率在亚洲人中高于高加索人(分别为3级45%和12%)。Trastuzumab 和 ado-trastuzumab emtansine 已被证明可以结合巨核细胞前体上的 FcgRIIa,但只有 ado-trastuzumab emtansine 影响了血小板的产生,表明这种 ADC 诱导的血小板减少症是由于 DM1 或其代谢产物 lys-SMCC-DM1。相反,使用可裂解连接子的微管抑制剂,如 vc-PAB-MMAE 或二硫键连接的 DM4 结合物,血小板减少症在临床上并不特别重要。
3.2.中性粒细胞减少症
骨髓中微管功能在有丝分裂期间的干扰导致中性粒细胞减少症。降低的中性粒细胞计数增加了感染的发生率,包括发热性中性粒细胞减少症和败血症。这是利用 MMAE 的 ADCs 一致的毒性。人们认为这是由于血浆中缬氨酸瓜氨酸可裂解连接子的不稳定性或由于这些结合物中存在的高药物负载物种的快速清除,导致游离药物的系统释放,从而产生更广泛的毒性谱。积极分裂的造血细胞受到 MMAE 的影响,导致中性粒细胞减少症,这在很大程度上是可逆的,随着治疗周期之间中性粒细胞数量的改善而改善。中性粒细胞减少症已报告用于 BV、pinatuzumab vedotin、polatuzumab vedotin、glembatumumab vedotin 和 PSMA-ADC(表2)。
在靶标依赖性毒性中,CD33 特异性 ADCs 吉妥单抗-奥佐加米辛和 SGN-33A 也已知通过与髓系前体细胞表面的 CD33 结合诱导中性粒细胞减少症,而 AVE9633 未观察到骨髓抑制。
3.3.眼部毒性
已报告 ADCs 和各种靶标存在眼部毒性(表2)。最近对 ADCs 报告的眼部毒性的全面回顾强调了与 ADCs 相关的各种眼部事件,其中视力模糊、角膜炎、干眼和微囊性上皮损伤是最常报告的。存在明确的有效载荷关联,通常由包括 DM4 和 MMAF 的 ADCs 诱导眼部毒性。两者倾向于使用稳定的连接子:在 SPDB-DM4 的情况下是稳定的可裂解二硫键连接,在 mc-MMAF 的情况下是不可裂解的连接。SPDB-DM4 最终释放中性可扩散的代谢产物(DM4 和 S-甲基-DM4),这些代谢产物具有旁观者杀死邻近细胞的潜力,而 mc-MMAF 在细胞内产生半胱氨酸-mc-MMAF 作为带电活性代谢产物,没有旁观者效应。目前尚不清楚为什么眼睛对这些有效载荷的毒性特别敏感,但对于 MMAF 结合的 ADC,毒性可能与药物在细胞内的积累有关。相反,使用 MMAE 的 ADCs 很少描述眼部事件。
眼部毒性已被报告为 ABT-414(角膜沉积物)的剂量限制毒性(DLT);AGS-16C3F;cantuzumab ravtansine(视力下降、角膜沉积物和角膜炎);mirvetuximab soravtansine(点状角膜炎和视力模糊);和 coltuximab ravtansine。SGN-75 的眼部毒性的中位发病时间为 44 天,随后多次给予 SGN-75。对于这次试验,眼部事件的发病率为 57%(3级,21%),导致 15% 的患者停止治疗。毒性包括干眼(30%)、角膜上皮病变(15%)、视力模糊(11%)和角膜炎(9%),并且是可逆的,中位解决时间为 79 天。预防性类固醇眼药水已成功用于减少 ABT-414 和 SGN-CD19A 的眼部事件的发生率,而 DM4 结合药物 SAR3419 和 IMGN853 的剂量调整已成功用于减少眼部毒性的发生率和严重程度(见下面关于剂量调整以最小化毒性的部分),并且向 SPDB 连接子添加硫酸基已在 IMGN853 中使用,作为减少眼部毒性的手段(专利号 US20120282282A1)。
许多驱动癌症生长的关键信号分子在眼部组织中表达,特别是表皮生长因子受体(EGFR)在角膜基底上皮细胞中表达,通过该受体的信号传导在许多眼部功能中很重要(例如,皮脂腺、毛囊、结膜、毛细血管)。针对 EGFR 的 ADCs 可能特别容易受到眼部毒性的影响;然而,两种处于临床开发中的 ADCs(AMG-595 和 ABT-414)旨在限制与野生型 EGFR 的结合,从而减少眼部毒性。AMG-595 特别结合 EGFR 的一种突变形式 EGFRvIII,这种形式由一些胶质母细胞瘤患者表达,并且不应该结合野生型 EGFR,这可能防止眼部事件。ABT-806,ADC ABT-414 中使用的抗体,结合到 EGFR 的一个表位,该表位只在激活的和肿瘤特异性的 EGFR 上暴露,不结合正常组织上的野生型 EGFR。36 ABT-414 通过 mc 连接子与 MMAF 结合,而 AMG-595 使用带有 SMCC 连接子的 DM1。ABT-414 的初步结果表明,眼部干扰确实是这种化合物的重要毒性,有 89% 的患者报告眼部事件,这似乎比已报告的其他 MMAF 结合的 ADCs 更多(见上文),但由于报告的差异和小数量的结果,很难得出明确的结论。
AMG-595 的毒性尚未报告。这两种化合物在临床测试中的头对头比较将确定是否针对这种突变会减少毒性,特别是与 EGFR 野生型 ADCs 相关的眼部毒性。
3.4.周围神经病变
肢体麻木和刺痛,蔓延引起刺痛和肌无力,是周围神经病变的特征。这主要见于微管抑制剂药物,如 auristatins 和 maytansinoid 衍生物(表2),以及 taxanes 和 vinca alkaloids。虽然这些药物通常对高度增殖的细胞有效,但成年神经元不积极分裂,但微管在神经元的存活和功能中起着重要作用,通过介导从细胞体到远端突触的蛋白质的主动运输。周围神经病变被认为是由于间期微管功能的破坏而发生的。它特别与具有蛋白酶可裂解连接子(例如,缬氨酸瓜氨酸)的 MMAE 结合物相关。可裂解连接子比其他连接子不稳定,允许游离药物系统释放。
在用 BV 治疗的患者中广泛报告周围神经病变,影响高达 50% 的患者,但 polatuzumab vedotin 和 glembatumumab vedotin 的发生率较低,分别影响 9% 和 4% 的患者。对于 BV,周围神经病变的发生率是累积的,任何级别周围神经病变的中位发病时间超过 12 周。2 和 3 级神经病变的发病时间分别为 27 周和 38 周。周围神经病变导致 10% 的患者减少剂量,13% 延迟剂量,9% 的患者停药。大多数患者的周围神经病变有解决或改善;然而,只有 50% 的患者注意到完全解决。
3.5.皮肤毒性
EGFR 信号通路和受体酪氨酸激酶在表皮细胞的正常功能中很重要。因此,针对这些途径的 ADCs 可能会表现出更高的皮肤毒性发生率,例如 bivatuzumab mertansine,由于正常角质形成细胞上 CD44v6 的表达,它引起了非常严重的皮肤毒性。这种 ADC 针对 CD44v6,它在各种实体瘤中表达,并正在进行头颈鳞状细胞癌和转移性乳腺癌的临床测试。在猕猴的临床前测试中,由于正常角质形成细胞上 CD44v6 的表达,显示了可逆的皮肤毒性。根据临床前数据,预计在临床上会有轻度至中度的皮肤毒性,并且在大多数患者中都有体验;然而,在剂量递增中,一名患者在 140 mg/m2(低于平行研究中确定的 MTD)发展为 4 级表皮坏死,并随后死亡。在这种情况下,人类角质形成细胞上 CD44v6 的表达导致 bivatuzumab mertansine 非常有效的靶向细胞毒性,但目标是不恰当的,导致不可接受的毒性。这突出了在引入临床之前需要彻底评估靶标抗原在正常表达的要求。
3.6.对内皮细胞的毒性
MEDI-547,一种潜在的实体瘤治疗候选药物,针对 EphA2,即受体酪氨酸激酶家族的成员。该抗体在体外与包括大鼠、小鼠和猕猴在内的各种物种的 EphA2 发生交叉反应。第一个人体研究从 0.08 mg/kg 的剂量开始,这比大鼠和猕猴预测的最高非严重毒性剂量低十倍。然而,在人类中,这个剂量超过了最大耐受剂量,接受治疗的患者出现了过度出血、出血和鼻出血。这种意外的毒性被认为是由 ADC 的抗体部分引起的,而不是 auristatin 有效载荷。这表明有时从动物研究预测的毒性水平与人类患者观察到的水平不同,并强调了继续开发合适的动物研究的必要性,这可能更准确地预测人类反应。
3.7.胃肠道
大多数患者在化疗后会经历胃肠道毒性。恶心和呕吐可能影响超过 90% 的患者,特别是那些接受顺铂、环磷酰胺或达卡巴嗪治疗的患者,即使在预防性抗呕吐药物存在的情况下,仍有 20% 的患者出现症状。大多数 ADCs 也会诱发胃肠道毒性,但它们大多是 1 或 2 级的严重程度。胃肠道毒性,包括恶心、呕吐、腹泻和便秘,最常报告用于 MMAE、卡利霉素和 DM1 结合的 ADCs(表2)。ADCs 诱发胃肠道毒性的机制尚未探讨,但可能源于微管抑制剂对胃肠道内快速增殖细胞的非特异性效应。有各种机制被认为会诱发化疗毒性,但 ADCs 诱发这种毒性的机制尚未确定。
值得注意的是,大约 30% 的接受伊立替康单药治疗的患者会出现腹泻,而来自 SN-38 结合的 ADCs 的早期迹象表明,它们的腹泻发生率比伊立替康显著降低(表3),可能表明 ADC 在胃肠道的分布比裸小分子细胞毒素低。
3.8.肝脏异常,包括肝窦阻塞症
当对肝脏窦状内皮细胞的有毒损伤导致内皮细胞脱落时,就会发生肝窦阻塞症。这些细胞随后栓塞至肝静脉,并导致小静脉纤维化,导致肝充血和无法清除毒素或其他废物。它已被报告为 inotuzumab ozogamicin 和 gemtuzumab ozogamicin,的毒性。其他报告肝转氨酶升高的化合物包括 cantuzumab mertansine、AVE963330 和 bivatuzumab mertansine.已提出这种效应是通过表达在肝脏窦状细胞上的甘露糖受体的 ADCs 摄取介导的。
4.临床前毒性
许多处于临床和临床前阶段的 ADCs 的抗体并不与小鼠或大鼠发生交叉反应,这阻碍了对“靶向”毒性的评估,并且只允许在这些物种中观察到非特异性、抗原无关的效应。对小鼠进行毒性评估可能对不太稳定的 ADCs 有用,但随着更具体、更稳定的 ADCs 开始进入临床,将需要更好地了解动物毒性,并且可能需要更好的临床前模型。
关于耐受性/毒性研究的最佳信息可能来自非人类灵长类动物模型,特别是猕猴,尽管这些模型在预测人类的毒性方面并不准确,例如 MEDI-547、bivatuzumab mertansine(如上所述)。阿多-曲妥珠单抗-埃姆坦辛的第一个1期临床研究的起始剂量是通过猕猴研究确定的。这些毒理学研究发现了坐骨神经的微观轴突变性,这表明周围神经病变将是剂量限制毒性(DLT)。预计人类和猕猴胶质细胞以及周围神经纺锤细胞上低水平的 HER2 表达将驱动这种毒性。然而,在患者中观察到的 DLT 是血小板减少症,在动物模型中很少见。这被认为是一种抗原无关效应,因为阿多-曲妥珠单抗-埃姆坦辛和使用相同连接子-有效载荷(SMCC-DM1)的 ADC 随后在体外被证明会干扰巨核细胞成熟。
5.ADCs 与标准治疗的毒性比较
标准化疗方案的疗效和耐受性都是剂量依赖性的,即,可能通过更高剂量实现更好的疗效,但毒性风险也会增加。由于 ADCs 的靶向特性,它们的耐受性可能与临床结果无关。因此,重要的是确保 ADCs 的毒性比标准治疗更不致残。目前有许多试验正在进行中,将目前批准的药物方案与 ADCs 进行比较,但这些试验的大多数尚未报告结果。随着这些研究的成熟,将 ADCs 的毒性与标准治疗进行比较将变得重要。目前,可以将阿多-曲妥珠单抗-埃姆坦辛、ino-阿霉素-奥佐加米辛和 glembatumumab vedotin 的毒性与对照组进行比较。对照组可能包含与 ADCs 相同或不同的机制的药物。像许多 ADCs 一样,紫杉醇类药物(例如多西他赛)和长春花生物碱(例如长春新碱)是微管抑制剂,因此具有类似的类别效应毒性。其他类别的化疗药物,如酪氨酸激酶抑制剂(例如拉帕替尼)、核苷类似物(例如吉西他滨)和烷化剂(例如苯达莫司汀)每种都有自己的关键毒性,可能比研究中的 ADC 更或更少致残。阿多-曲妥珠单抗-埃姆坦辛诱导了更多的血小板减少症和 AST 升高,但比其他方案包括曲妥珠单抗加多西他赛或拉帕替尼加卡培他滨的中性粒细胞减少症和腹泻更低(表4)。
Inotuzumab ozogamicin(抗 CD22-卡利霉素 ADC)在 INO-VATE ALL 研究中报告的急性淋巴细胞性白血病(ALL)患者的肝窦阻塞症发生率高于强化化疗(分别为 15% 和 1%)。这项试验显示出比对照组显著更好的反应。在一项针对复发/难治性 NHL 患者的3期试验中,ino-阿霉素-奥佐加米辛与更高发病率的血小板减少症相关,但与苯达莫司汀或吉西他滨的对照组相比,中性粒细胞减少症的发病率较低。这项研究因在这些主要难治性 NHL 患者中的无效性而终止。
在 Emerge 试验中,严重预处理的乳腺癌患者接受了 glembatumumab vedotin 或研究者选择的治疗,后者包括 eribulin(nD15)、ixabepilone(nD7)、gemcitabine(nD5)、vinorelbine(nD5)、doxorubicin(nD3)或与白蛋白结合的紫杉醇(nD2)。接受 glembatumumab vedotin 治疗的患者经历的血小板减少症(4%)、中性粒细胞减少症(29%)和白细胞减少症(10%)低于研究者选择的(分别为 15%、44% 和 27%)。相反,glembatumumab vedotin 治疗的皮疹发生率为 47%,而对照组为 2%,周围神经病变的发生率几乎是研究者选择的两倍(23% 对比 12%)。
在一项针对霍奇金淋巴瘤前线治疗的 BV 结合研究中,患者接受了包含多柔比星、博来霉素、长春新碱和氮烯咪胺(ABVD)加上 BV 或修改方案的多柔比星、长春新碱和氮烯咪胺(AVD)加上 BV 的标准方案。接受 BV 加 ABVD 的患者经历了不可接受的肺毒性效应(44% 的患者),这比单独使用 ABVD 通常看到的比率高。博来霉素以其对肺部的不良影响而闻名,将 BV 添加到含有博来霉素的 ABVD 方案中增强了这种效应。从 BV 组中去除博来霉素消除了肺部效应,并没有减少完全反应的数量。目前有一项新试验正在进行,比较 BV 加 AVD 与单独 ABVD。
SN-38 是伊立替康的活性代谢产物,可以对使用 SN-38 有效载荷的 ADCs(labetuzumab govitecan 和 sacituzumab govitecan)与伊立替康单药治疗的毒性进行直接比较。最显著的区别是,与单药治疗相比,接受 ADCs 的患者经历的腹泻减少(表3)。
尽管这些研究规模较小,但这一研究组表明 ADCs 的毒性表现出一些限制性分布,而不是与对照组看到的总体细胞毒性。然而,仍然清楚的是,在迄今为止开发的 ADCs 中,主要毒性是由有效载荷代谢产物驱动的,表明 ADCs 的非特异性。下一代 ADCs,具有更好的靶向抗体、更稳定的连接子,并对最终有毒代谢产物的化学性质给予关注,希望能够显示出更少的有效载荷特异性毒性。相反,毒性可能更具有器官或细胞类型特异性,可以预测,并且在必要时,可以通过预防性护理进行管理。
6.调整剂量以尽量减少毒性
药代动力学(PK)的评估对于在最小化毒性的同时优化药物的疗效非常重要。IMGN853 和 SAR3419 都有较窄的治疗窗口,但有效的剂量调整降低了高峰血浆浓度并最小化了毒性。IMGN853(mirvetuximab soravtansine)是一种靶向叶酸受体α(FRα)的 ADC,通过磺酸-SPDB 连接子与 DM4 结合,目前正在进行针对 FRαC 铂耐药卵巢癌患者的1期临床研究。使用全体重来确定剂量的早期测试显示,与 mirvetuximab soravtansine 的高早期暴露水平相关的高度可逆的眼部毒性。在较低剂量下观察到临床效益,眼部毒性的发生率较低。基于调整后的理想体重计算的剂量调整减少了血浆浓度的变异性,并将它们保持在眼部毒性的阈值以下。由于这种剂量调整,1期剂量扩展队列继续进行,有早期迹象表明在严重预处理的 FRαC 铂耐药卵巢癌中具有临床效益,总响应率(ORR)为 40%。
另一种为限制毒性而进行剂量调整以最大化抗肿瘤效果的药物是 SAR3419(coltuximab ravtansine)。这是一种靶向 CD19 的 ADC,使用 DM4/SPDB 有效载荷/连接子组合。最初的1期研究每3周(q3w)剂量一次,实现了 22% 的 ORR,并通过增加剂量频率同时减少剂量来尝试提高这一比例。每周一次的 coltuximab ravtansine 给药导致药物累积,由于半衰期长 7 天和清除缓慢,这反过来又导致在第 7 或第 8 周晚发性(3级)周围神经毒性的发展。通过在最初每周一次给药 4 周后,将剂量减少到每2周一次,维持了血浆浓度在稳定水平。这种最佳剂量计划与每周给药相比,减少了神经系统和眼部毒性的发生率,并将 ORR 提高到 33%,与 q3w 给药相比。
吉妥单抗-奥佐加米辛被批准用于首次复发的 CD33 阳性 AML 患者,他们不是其他治疗的候选者。推荐剂量是两次输注,每次 9 mg/m2 吉妥单抗-奥佐加米辛,间隔至少 2 周。接受此剂量药物的患者经历的关键毒性包括血小板减少症(99%)和中性粒细胞减少症(97%),通常持续时间较长。肝脏事件很常见,有 23% 的患者出现 3 级或 4 级高胆红素血症,17% 的患者有 3 级或 4 级丙氨酸转氨酶或天门冬氨酸转氨酶水平升高。26 还有很高的肝窦阻塞症发生率,特别是在接受吉妥单抗-奥佐加米辛治疗后接受造血干细胞移植的患者中。26 最近,对吉妥单抗-奥佐加米辛的分次剂量进行了研究,以便患者接受 9 mg/m2 分为 3 次剂量的 3 mg/m2,分别在第 1、4 和 7 天。65 这种剂量调整导致血小板减少症的发生率降低,肝功能异常指标减少。对吉妥单抗-奥佐加米辛和 inotuzumab ozogamicin 的 PK 分析表明,毒性是由峰值血浆浓度驱动的,而疗效是由 AUC 驱动的。因此,减少剂量将降低峰值血浆浓度并减少毒性,而更频繁的给药将允许更长时间的药物暴露,以维持疗效。
总体而言,研究结果表明,通过剂量调整来优化血浆浓度,从而增加 ADCs 的治疗窗口,并在限制毒性的同时增强临床效益。在1期之后,继续监测患者的 PK 概况将对管理在试验扩展期间观察到的毒性非常重要。
7.降低毒性的药物修改
Agensys, Inc.,Astellas Pharma Inc. 的子公司,已开发 AGS-16M8F 和 AGS-16C3F 用于治疗肾细胞癌。这些是完全人 IgG2k 抗 ENPP3 抗体,通过 mc 连接子与 MMAF 结合。AGS-16C3F 在中国仓鼠卵巢细胞中产生,与杂交瘤衍生的 AGS-16M8F 相比,允许增加生产。在临床前测试中,它们显示出类似的 PK 和毒理学概况,但1期研究确实显示出一些差异。在最初的1期研究中,使用 AGS-16M8F,4.8 mg/kg 未达到 MTD,但有显著的眼部毒性,导致 3/8 的受试者停止治疗。然而,4.8 mg/kg 超过了 AGS-16C3F 的 MTD,导致降低剂量以找到 1.8 mg/kg 的 MTD。AGS-16C3F 报告的副作用是疲劳和血小板减少症。由于观察到疾病控制,AGS-16C3F 正在继续进行临床试验。给予任一种药物的患者血小板减少症的发生率相似,这可能表明诱导眼部毒性和血小板减少症的机制不同。这两种化合物的毒性差异令人惊讶,因为临床前结果发现 AGS-16M8F 和 AGS-16C3F 的抗体结合、细胞毒性和肿瘤大小减少相似。尚不清楚为什么这些化合物有不同的 MTDs,但可能是由于蛋白质糖基化的差异。差异表明制造过程以及 ADC 的每个组成部分在驱动毒性方面的作用。这是一个需要进一步调查的领域。
Cantuzumab mertansine(Can M)和 cantuzumab ravtansine(Can R)是两种靶向 CanAg 的 ADC,CanAg 在结肠和胰腺肿瘤上过度表达。它们在有效载荷和连接子组合上有所不同。虽然 Can M 有一个相对不稳定的二硫键连接在 SPP 连接子和 DM1 之间,但 Can R 有一个更受阻碍的、相对稳定的二硫键,通过将 DM4 连接到 SPDB 形成。这两种药物有不同的毒性概况;在 Can M 的情况下是转氨酶升高,而在 Can R 的情况下是眼部毒性。转氨酶升高的发生率在有肝转移的患者中最高,表明对正常肝细胞的旁观者效应。两种化合物都没有超过1期,可能是由于它们的抗肿瘤效果最小。
8.在不同疾病类型中的不同毒性
在不同疾病类型中使用相同药物可能出现不同的毒性。在 polatuzumab vedotin 的剂量递增过程中,CLL(慢性淋巴细胞性白血病)患者的剂量限制毒性(DLTs)出现在比 NHL(非霍奇金淋巴瘤)低得多的剂量下。NHL 的最大耐受剂量(MTD)定义为 2.4 mg/kg,但在 5 位接受 1.8 mg/kg 治疗的 CLL 患者中,有 2 位经历了 4 级中性粒细胞减少症和 4 级真菌感染的 DLTs。这导致 CLL 的 MTD 为 1.0 mg/kg,低于所需的临床效益水平。对 PK 概况的分析显示,与 NHL 相比,CLL 患者的暴露量更低,清除速度更快,这与由于 CLL 中循环 B 细胞数量更多而导致的靶标介导清除一致。
在一项针对 43 名黑色素瘤患者使用 glembatumumab vedotin(CDX-011)的研究中,皮疹作为不良事件的发生率是 74%,在 30% 的患者中这是 3 级或更高严重程度,影响了超过 50% 的体表面积,而乳腺癌患者的发生率为 47%,只有 4% 为 3 级。研究发现,皮疹的发生率与黑色素瘤患者的改善 ORR(客观响应率)和无进展生存期以及乳腺癌患者总体生存期的改善相关。黑色素瘤患者还经历了更高水平的瘙痒(63%)和脱发(65%),而乳腺癌患者分别为(21% 和 25%)。两组患者的血液学不良事件的发生率相似,大约 30% 的患者经历了中性粒细胞减少症(其中 20% 为 3 级)和 5% 的患者经历了血小板减少症。
来自 sacituzumab govitecan(IMMU-132)的早期报告表明,不同的患者群体中中性粒细胞减少症的发生率可能存在差异,三阴性乳腺癌患者中有 30%,胃肠道癌症患者中有 24%,肺癌患者中有 18% 报告了中性粒细胞减少症。由于患者数量较少,现在下结论这些是真正的差异性毒性可能为时过早,随着每个疾病群体中更多患者的加入,需要进一步监测。
9.ADCs 的未来,临床前证据
减少 ADC 的 DAR(药物-抗体比率)异质性可能会导致更好的临床概况,无论是在疗效还是毒性方面。目前大多数正在进行临床测试的 ADC 是不同 DAR 的化合物的异质混合物,通常范围是每个抗体 0-8 个药物。多种 ADC 变体可能导致广泛的 PK 值,这可能限制治疗窗口。
设计 ADC 的新技术,能够生产每个抗体具有特定数量药物的化合物,可能会产生具有更好治疗指数的 ADC。例如,对一系列传统结合的 CD30-MMAE ADCs 的研究表明,DAR 为 2、4 或 8,DAR 8 在小鼠研究中比 DAR 4 或 2 更快地被清除,治疗指数更低。通过位点特异性结合或改进连接子设计,可能通过减少高 DAR ADCs 的疏水性,从而增加药物在体内的暴露量,提高 ADCs 的治疗指数。
有几种潜在的方法可以位点特异性结合 ADC,包括用半胱氨酸(THIOMAB)或非天然氨基酸替换氨基酸,以及使用细菌转谷氨酰胺酶进行酶促结合。使用 THIOMAB 技术,通过将重链丙氨酸 114 替换为半胱氨酸,产生了一个平均 DAR 为 1.6 的 THIOMAB 药物结合物(TDC),相比之下,原始抗 MUC16 ADC 的 DAR 为 3.5。在小鼠异种移植模型中,TDC 显示出优于原始 MUC16 ADC 的体内效果和耐受性,肝脏和骨髓毒性较低。使用细菌转谷氨酰胺酶方法将 cAC10(与 BV 中的抗体相同)与 MMAE 结合,产生一个 DAR 为 4 的均一 ADC。在大鼠中,这种新的 ADC 显示出比 BV 更好的肿瘤摄取,并且在肝脏和脾脏中的非靶向摄取更低,这允许更高的最大耐受剂量。
第一个公开披露的正在进行临床开发的位点特异性 ADC 是 SGN-33A,这是一种靶向 CD33 的 ADC,它使用位点特异性工程化半胱氨酸稳定地连接到 PBD 二聚体。这产生了每个抗体有 2 个 PBD 二聚体的 ADC。已经报告的初始毒性包括中性粒细胞减少症(表 2)。SGN-70A 也使用了这项技术,但毒性尚未报告。
10.结论
这篇综述表明,对于大多数化合物而言,毒性是由有效载荷和最终代谢产物的性质驱动的。有一些特定的例子是靶标介导的毒性。了解 ADC 的不同部分在药物毒性中所起的作用(除了对疗效所需的作用),将有助于设计具有更好安全概况的候选药物。在可行的情况下,讨论了与其他治疗选择的比较。进入 3 期测试的化合物(BV、ino-阿霉素-奥佐加米辛、阿多-曲妥珠单抗-埃姆坦辛和 glembatumumab vedotin)确实比标准治疗的毒性更少。然而,大多数 ADC 处于 1 期临床测试中,其中一些报告的毒性是无法管理的,可能会导致它们无法进展。随着该领域扩展到包括具有特定 DARs 的更合理设计的 ADC,监测毒性将变得重要,以确保药物的改进靶向将提高治疗指数。
识别微信二维码,添加生物制品圈小编,符合条件者即可加入
生物制品微信群!
请注明:姓名+研究方向!
版
权
声
明
本公众号所有转载文章系出于传递更多信息之目的,且明确注明来源和作者,不希望被转载的媒体或个人可与我们联系(cbplib@163.com),我们将立即进行删除处理。所有文章仅代表作者观点,不代表本站立场。
抗体药物偶联物临床结果
2024-06-06
·精准药物
抗体偶联药物(ADCs)是一种将特异性的单克隆抗体与毒素小分子相结合的药物,是近年来发展迅速且前景广阔的生物制药领域。ADC与靶点结合后,通过抗原介导的内吞作用将细胞毒性药物递送到肿瘤细胞,然后在晚期溶酶体中通过酶催化(如组织蛋白酶B)释放细胞毒性药物。ADC靶点的选择一直是研究者关注的重要内容。据统计,ADC的靶点主要为肿瘤相关抗原(TAAs),如HER2、CD30等。这些抗原不仅在肿瘤细胞表面高表达,而且在其他体细胞表面也有一定程度的表达。因此,ADC在杀灭肿瘤细胞的同时,也能杀灭部分正常细胞。例如,靶向Her2的ADC可能导致心脏毒性。因此,这类ADC靶点必须面临脱靶问题。而肿瘤特异性抗原(tsa)不存在上述问题,如细胞外区突变的膜蛋白、多肽-主要组织相容性复合体(pMHC)等。它们可以特异性地将肿瘤细胞与正常细胞区分开来,使ADC能够特异性地靶向并杀死肿瘤细胞。同时,对单克隆抗体的特异性和亲和力要求很高。因此,本文将根据肿瘤特异性的程度分别介绍肿瘤相关抗原和肿瘤特异性抗原,并重点介绍肿瘤特异性抗原。
表1.FDA批准上市的ADC靶点
1. 肿瘤相关抗原
肿瘤相关抗原(TAAs)是一类在肿瘤中高表达的蛋白质细胞并且在正常体细胞中低表达或正常表达,其中较常见的抗原有HER2、CD30、CA125等,常用于临床诊断和肿瘤类别。
1.1. 与细胞增殖相关的膜蛋白
人表皮生长因子受体2 (Human epidermal growth factor receptor 2, HER2)是表皮的一员生长因子受体家族,由ERBB2基因编码,位于细胞膜上。据报道,HER2蛋白在乳腺癌、胃癌等肿瘤细胞中高表达。对于乳腺癌,HER2蛋白的表达是影响其预后的重要因素。体内25% ~ 30%的乳腺癌患者受到肿瘤细胞HER2基因过表达的影响。这些肿瘤细胞不仅具有很强的增殖能力,而且容易对某些化疗药物产生耐药性。
目前上市的靶向HER2的单克隆药物包括曲妥珠单抗(Herceptin®)和帕妥珠单抗(Perjeta®),主要基于阻断HER2二聚化。曲妥珠单抗(Trastuzumab)的T-DM1 (Kadcyla®)(图1)于2013年被批准用于治疗晚期her2阳性转移性乳腺癌。但也可引起肺毒性(间质性肺疾病,包括局部急性肺炎、急性呼吸窘迫综合征)、输液相关反应、过敏反应、血小板减少、神经毒性以及注射部位溢液(红肿、疼痛等)。
图1. T-DM1(Kadcyla®)结构图
1.2. 癌胚抗原
滋养层糖蛋白(Trophoblast glycoprotein, TPBG)又称5T4,是一种典型的癌胚蛋白抗原,表达于肿瘤细胞和胚胎细胞表面很少表达于其他正常体细胞表面。其中,5T4抗原在结直肠癌、胃癌、卵巢癌和其他肿瘤。研究表明,5T4在胃癌组织中的表达结直肠癌和胃癌分别达到85%和81%。因此,5T4作为抗体药物或ADC的潜在抗原已受到研究者的广泛关注。
1.3. 白细胞分化抗原
白细胞分化抗原是细胞系的细胞表面标记(包括血小板、血管内皮细胞等),在正常分化成熟、不同阶段和激活过程中出现或消失。其中,抗体药物靶点的选择中,分化簇(CD)更为常见。其中之一是CD30,它广泛表达于霍奇金淋巴瘤细胞,并且是已上市的ADC本妥昔单抗 (Adcetris®)的靶点。此外,CD33和CD22是另外两种已上市ADC(奥加伊妥珠单抗和吉妥珠单抗奥唑米星)的靶点。
2. 肿瘤特异性抗原
仅存在于细胞膜上的膜蛋白或膜蛋白复合物肿瘤细胞表面被定义为肿瘤特异性抗原,可作为特异性区分肿瘤细胞和正常体细胞的潜在标志物。因此,我们认为肿瘤细胞表面突变的膜蛋白复合物和肿瘤细胞表面胞外区突变的膜蛋白可作为肿瘤特异性抗原。如图2所示,突变的胞内蛋白通过蛋白酶降解并由主要组织相容性复合体(MHC)ⅰ类分子呈递含有突变氨基酸的多肽到细胞表面,形成突变的pMHC;而胞外突变膜蛋白直接定位于细胞膜。两者都是基于突变的氨基酸形成肿瘤特异性抗原,成为抗体或ADC的潜在靶点。接下来,将重点详细介绍上述两种肿瘤特异性抗原。
图2 由突变基因形成的突变膜蛋白和突变pMHC
2.1. 肿瘤特异性pMHC
pMHC由3部分组成:MHC的 I类分子、β2-微球蛋白(β2M)、以及由8 ~ 11个氨基酸组成的抗原肽,如图3所示。其中,MHC 的I类分子由α1、α2、α3三个结构域组成。α3结构域位于细胞膜上,α1和α2结构域位于细胞膜外。抗原肽位于α1和α2结构域的两个大α螺旋上,在主槽内;β2-微球蛋白与MHC的I类分子的α1和α3结构域相互作用。在细胞中,一些翻译后被错误折叠和修饰的新蛋白质,以及一些与泛素连接的蛋白质,被蛋白酶体降解,产生一系列长度从3个氨基酸到21个氨基酸不等的多肽。这些长度为8-11个氨基酸的多肽通过多肽转运体转运到内质网。经进一步N端修饰后,与MHC的I类分子和β2-微球蛋白结合形成pMHC,经高尔基体转运至细胞膜。
图3.pMHC 结构
2.1.1. 肿瘤特异性pMHC作为抗体药物靶点的研究
肿瘤突变抗原是一类仅在肿瘤细胞中表达的突变抗原。因此,靶向肿瘤突变抗原的药物可以从根本上区分肿瘤细胞和正常体细胞,最大限度地减少药物脱靶引起的毒副作用。pMHC可为抗体药物提供肿瘤特异性突变靶点。Skora利用噬菌体展示技术筛选出针对人类白细胞抗原(HLA)-A*02/克尔斯特鼠肉瘤病毒癌基因(KRAS) G12V突变型和HLA-A *03/EGFR L858R突变型pMHC的scFv,并在细胞水平分析和评价其特异性和亲和力。结果表明,scFv能与仅含1个突变氨基酸的突变型pMHC特异性结合,亲和力为48.65 nmol/L,而野生型pMHC无亲和力。这一结果充分证明了单克隆抗体能够区分只有一个氨基酸差异的pMHC,由肿瘤特异性突变肽组成的pMHC有望成为一种很有前景的抗体和ADC靶点。
2.1.2. 肿瘤特异性pMHC作为抗体药物的潜在靶点
费城染色体是一种特殊的慢性染色体异常髓系白血病(CML)细胞,导致融合基因BCR-ABL的形成。在费城染色体阳性(ph +)细胞中,有三种不同融合蛋白b2a2, e1a2和b3a2是由剪接变异体分别产生的。来源于这些融合蛋白连接的几个新肽与MHC I亚型具有高亲和力。与HLA-A03结合的多肽KQSSKALQR具有体外免疫原性。采用串联纳米喷雾质谱检测K562细胞(ph+);Cyto对HLA-B08结合肽GFKQSSKAL具有特异性的毒性T淋巴细胞(CTLs)CML患者与健康供者之间已分离,但阳性频率低于HLA-A03结合肽这意味着肿瘤负荷可能会影响产生肿瘤的能力特定的CTL。对于HLA-A02结合肽SSKALQRPV, Yotnda提高了其表达水平特异性CTL成功裂解靶HLA-A02转染的CML细胞。更重要的是,Kessler从三种融合蛋白和系统检测体外蛋白酶体消化后的降解肽质谱分析,最终鉴定出AEALQRPVA为一种多肽HLA-B61具有CTL表位。
B-raf V600E突变来源于第600位点的氨基酸替换(缬氨酸到谷氨酸)。据报道,37-50%的黑色素瘤存在V600突变,其中约80-90%的突变为V600E。B-raf的V600E突变增加了激酶活性和侵袭性,也导致黑素瘤细胞凋亡减少。由于V600E突变在黑素瘤细胞内的高频表达,被广泛认为是T细胞的潜在免疫治疗靶点。利用突变肽刺激黑色素瘤患者来源的CD4+ (CD4+) T细胞和CD8+ (CD8+) T细胞,观察其对靶细胞的比活性。尤其是CD8+ HLA-B2705限制性突变体特异性T细胞,携带V600E的黑色素瘤细胞被选择性识别和杀灭。巧合的是,类似的结果已在V600E突变和HLA-A02阳性的黑色素瘤患者中得到验证。这些研究表明V600E突变肽能与HLA-B2705和HLA-A2结合,诱导CD8+ CTL反应,为免疫治疗提供了一个有前景的表位。
G12突变是K-ras基因的主要致癌突变,其中G12D和G12V突变在60-70%的胰腺癌、20-30%的结直肠癌和20-30%的非小细胞肺癌(NSCLC)中均有报道。目前,针对突变K-ras的免疫治疗方法已被开发。将含有G12V突变的多肽接种于胰腺癌患者,CD8+特异性T细胞在体内杀伤携带G12V突变的自体肿瘤细胞,并验证HLA-B35限制性多肽VVVGAVGVG为效应T细胞的作用靶点。利用树突状细胞(dc)提呈K-ras突变体G12V多肽,激活突变体特异性T细胞,在体外和小鼠胰腺肿瘤模型中发挥抗胰腺癌的作用。G12D K-ras突变DNA疫苗作为一种新型的免疫治疗药物在小鼠NSCLC肿瘤模型中进行了研究,观察到接种后辅助性T细胞1免疫应答增强,肿瘤浸润的CD8+ T细胞数量增加。巧合的是,通过免疫HLA-A1101转基因小鼠和基因工程人外周血淋巴细胞,获得了HLA-A1101限制性G12D突变表位特异性t细胞受体(TCR)。在小鼠异种移植模型中,转导的TCR-T细胞显著抑制携带G12D突变的HLA-A1101阳性肿瘤生长。
EGFR T790M首次作为获得耐药性突变被研究于EGFR酪氨酸激酶抑制剂(TKIs)治疗,约50% ~ 60%的NSCLC患者在接受EGFR TKIs治疗后出现疾病进展后检测到该突变。最近的研究发现,79.9%的EGFR存在T790M突变TKI治疗前NSCLC患者的发生率为0.009% ~ 26.9%。2015年,奥希替尼(Tagrisso)被FDA批准为第三代药物EGFR TKI,与EGFR的某些突变形式(T790M, L858R, 19外显子)结合缺失)的浓度约为野生型的9倍。然而,在体外实验中,在临床相关浓度的奥希替尼也抑制了HER2, HER3, HER4, ACK1和BLK的活性。可以说,EGFR T790M是近年来抗肿瘤药物研究的热门靶点。研究人员希望找到一种专门针对这种突变的药物,从而特异性地杀死肿瘤细胞,而不是其他组织。通过HLA-A2限制性内切酶验证了2条T790M突变肽为肿瘤特异性表位。此外,HLA-A2+健康供试者的PBMCs经突变肽刺激后可诱导出突变特异性CTI,并选择性地对携带HLA-A2+的T790M的NSCLC细胞产生反应,这表明EGFR T790M突变可能是EGFR- TKI耐药患者的潜在免疫治疗靶点。
最近,针对P53来源的pMHC的抗体优先表达黑色素瘤抗原(PRAME)、甲胎蛋白(AFP)等肿瘤相关抗原也被一一筛选出来,并用于临床前抗肿瘤研究。这表明肿瘤相关和肿瘤特异性的pMHC越来越受到关注。研究者希望通过这类靶点的发现,拓宽抗肿瘤药物靶点的选择,为发现一类新的抗肿瘤药物和治疗方法奠定坚实的基础。
2.2. 肿瘤特异性突变胞外跨膜蛋白
跨膜蛋白的胞外结构域位于细胞表面,可以与抗体或抗体衍生物结合。一旦突变发生在跨膜蛋白的胞外区,可能产生一个新的突变特异性B细胞表位。国际癌症基因组联盟(International Cancer Genome Consortium, ICGC)数据库和癌症体细胞突变目录(Catalogue Of Somatic mutation In Cancer, COSMIC)数据库是两个重要的肿瘤测序数据库,基于ICGC数据库,分析了9155例肿瘤样本,在胞外跨膜蛋白发现了3328个突变,其中17个突变发生频率超过5次,频率超过1%(表1)。
表2. 胞外跨膜蛋白17种突变概述
EGFR变异体II(EGFRvIII)突变是由EGFR外显子2-7缺失引起的在恶性胶质瘤和NSCLC中发现。近20年来,EGFRv III作为一种肿瘤特异性的抗原表位的研究在我国呈上升趋势,人们对其进行了大量的研究,尤其在单克隆抗体中。AMG595是一种高度选择性EGFRv III特异性ADC,由Amgen开发,显示出有效的对肿瘤生长抑制的活性。这是第一个靶向肿瘤特异性表位的ADC进入I期临床试验。
此外,EGFR S492R突变也是一种潜在的肿瘤特异性ADC靶点,是膜蛋白胞外结构域的一个点突变显著影响西妥昔单抗与EGFR的结合。在西妥昔单抗治疗后的转移性结直肠癌(mCRC)活检组织和mCRC细胞系中检测到这种变异,而所有治疗前的活检组织和细胞均为EGFR和K-ras野生型,并在体外验证了西妥昔单抗耐药。此外,建立了一种基于微滴式数字PCR的方法检测帕尼单抗和西妥昔单抗治疗的mCRC患者血浆中循环游离DNA (cfDNA)中EGFR S492R突变,西妥昔单抗组16%(46/285)的患者检测到S492R突变,帕尼单抗组1%(3/261)的患者检测到S492R突变。此外,EGFR S492R突变也是一种潜在的肿瘤特异性ADC靶点,是膜蛋白胞外结构域的一个点突变显著影响西妥昔单抗与EGFR的结合。这种变异在转移性结直肠癌(mCRC)活检和mCRC细胞中被检测到西妥昔单抗治疗后的细胞系,而所有治疗前的活检和细胞均为野生型体外验证其对西妥昔单抗耐药。此外,建立了一种基于微滴式数字PCR的敏感检测血浆中循环游离DNA (cfDNA) EGFR S492R突变的方法帕尼单抗联合西妥昔单抗治疗的mCRC患者,S492R突变16%(46/285)接受西妥昔单抗治疗的患者和1%(3/261)采用帕尼单抗治疗。以上研究表明,EGFR S492R突变可能是西妥昔单抗治疗后的一种独特的获得性耐药突变,与原发耐药无关,其在西妥昔单抗治疗后的频繁发生使其成为潜在的肿瘤特异性抗原,可用于突变特异性抗体治疗。
3. 结论
目前正在进行临床试验的ADC大部分靶点仍然是肿瘤相关抗原,但许多肿瘤特异性抗原已经在进行临床或临床前研究。随着测序技术和检测技术的发展,更多原发性肿瘤特异性抗原和获得性肿瘤特异性抗原将被发现和鉴定。因此,在抗体和ADC靶点的选择上,更倾向于选择肿瘤特异性抗原,以减少靶点选择带来的副作用。关于ADC靶点的选择有非常多的讲究,后续会为大家带来更多分享。
参考文献
1.Jun Lai and Shuqing Chen,Relationship Between Target and Specific Action of Antibody-Drug Conjugates.
2.Xiaoxiao Wang,The target atlas for antibody-drug conjugates across solid cancers.
声明:发表/转载本文仅仅是出于传播信息的需要,并不意味着代表本公众号观点或证实其内容的真实性。据此内容作出的任何判断,后果自负。若有侵权,告知必删!
长按关注本公众号
粉丝群/投稿/授权/广告等
请联系公众号助手
觉得本文好看,请点这里↓
抗体药物偶联物
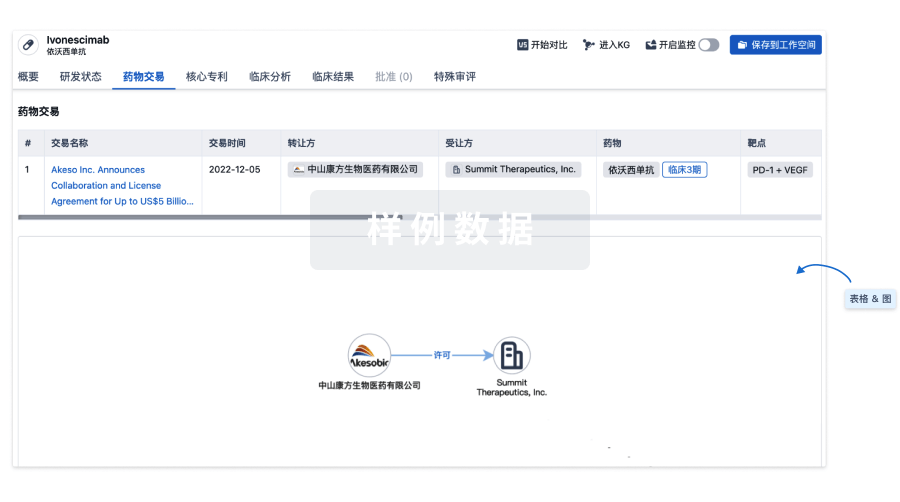
100 项与 AMG-595 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 多形性胶质母细胞瘤 | 临床1期 | 美国 | 2012-02-01 | |
| 多形性胶质母细胞瘤 | 临床1期 | 澳大利亚 | 2012-02-01 | |
| 复发性恶性胶质瘤 | 临床1期 | 美国 | 2012-02-01 | |
| 复发性恶性胶质瘤 | 临床1期 | 澳大利亚 | 2012-02-01 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
生物类似药
生物类似药在不同国家/地区的竞争态势。请注意临床1/2期并入临床2期,临床2/3期并入临床3期
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或

Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
