预约演示
更新于:2025-03-23
B1R inhibitors(Alpha-9 Theranostics)
更新于:2025-03-23
概要
基本信息
原研机构 |
在研机构 |
非在研机构- |
最高研发阶段临床前 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
结构/序列
使用我们的ADC技术数据为新药研发加速。
登录
或

关联
100 项与 B1R inhibitors(Alpha-9 Theranostics) 相关的临床结果
登录后查看更多信息
100 项与 B1R inhibitors(Alpha-9 Theranostics) 相关的转化医学
登录后查看更多信息
100 项与 B1R inhibitors(Alpha-9 Theranostics) 相关的专利(医药)
登录后查看更多信息
4
项与 B1R inhibitors(Alpha-9 Theranostics) 相关的文献(医药)2023-07-14·BMC plant biology
Two types of GLR channels cooperate differently in light and dark growth of Arabidopsis seedlings.
Article
作者: Lewandowska, Aleksandra ; Gabryś, Halina ; Krzeszowiec, Weronika ; Bebko, Kateryna ; Scholz, Sandra S ; Lyczakowski, Jan Jakub
BACKGROUND:
GLutamate Receptor-like (GLR) channels are multimeric, ionotropic, ligand-gated plant transmembrane receptors. They are homologous to mammalian glutamate receptors, iGLuRs, which are critical to neuronal function. GLRs have been reported several times to play a role in photomorphogenesis. However, to date, no study has looked at the mechanism of their involvement in this process. Here we focused on examining the impact of GLRs on the regulation of early seedling growth in blue light, red light, and in the dark.
RESULTS:
Wild type and six photoreceptor mutant seedlings were grown on media supplemented with known iGLuR/GLR channel antagonists: MK-801, which non-competitively blocks NMDA channels in mammalian cells, and CNQX, known for competitive blocking of AMPA channels in mammalian cells. The lengths of hypocotyls and roots were measured in seedlings of phyA, phyB, phot1, phot2, cry1, and cry2 mutants after 7 days of in vitro culture. Changes in growth parameters, both in light and in darkness upon application of chemical antagonists, show that both types of GLR channels, NMDA-like and AMPA-like, are involved in the regulation of seedling growth irrespective of light conditions. Analysis of seedling growth of photoreceptor mutants indicates that the channels are influenced by signaling from phot1, phot2, and cry1. To extend our analysis, we also evaluated the elicitation of a calcium wave, which is likely to be partially driven by GLRs, in Arabidopsis seedlings. The changes in cellobiose-induced calcium waves observed after applying GLR inhibitors suggest that both types of channels likely cooperate in shaping Arabidopsis seedling growth and development.
CONCLUSIONS:
Our work provides the first experimental evidence that two types of GLR channels function in plants: NMDA-like and AMPA-like. We also demonstrate that the channels are involved in seedling growth and development, at least partially through modulation of calcium signaling, but they are unlikely to play a major role in photomorphogenesis.
2018-08-01·Cytokine3区 · 医学
Role of selective blocking of bradykinin B1 receptor in attenuating immune liver injury in trichloroethylene-sensitized mice
3区 · 医学
Article
作者: Wang, Hui ; Zang, Dandan ; Zhang, Jiaxiang ; Yang, Peng ; Zhu, Qi-Xing ; Li, Na ; Yang, Ling ; Shen, Tong
Trichloroethylene (TCE) is able to induce trichloroethylene hypersensitivity syndrome (THS) with multi-system immune injuries. In our previous study, we found kallikrein-kinin system (KKS) activation, including the bradykinin B1 receptor (B1R), which contributed to immune organ injury in TCE sensitized mice. However, the mechanism of B1R mediating immune dysfunction is not clarified. The present study initiates to investigate the potential mechanism of B1R on liver injury. We establish a TCE sensitized BALB/c mouse model to explore the mechanism with or without a B1R inhibitor R715. We found B1R expression was increased in TCE sensitization-positive mice. As expect, hepatocyte intracellular organelles and mitochondria disappeared, glycogen particles reduced significantly as well in TCE sensitization-positive mice via the transmission electron microscopic examination, meanwhile, R715 alleviated the deteriorate above. The blockade of B1R resulted in a significant decreased p-ERK1/2 and increased p-AKT expression. The expression of CD68 kupffer cell and its relative cytokine, including IL-6 and TNF-α, increased in TCE sensitization-positive mice and decreased in R715 pretreatment TCE sensitization-positive mice. Together, the results demonstrate B1R plays a key role in ERK/MAPK and PI3K/AKT signal pathway activation and inflammation cytokine expression in immune liver injury induced by TCE. B1R exerts a pivotal role in the development of TCE induced liver injury.
2012-08-01·Pain medicine (Malden, Mass.)3区 · 医学
The Clinical Significance of QT Interval Prolongation in Anesthesia and Pain Management: What You Should and Should Not Worry About
3区 · 医学
Review
作者: Carlton Q. Brown ; Christopher Spevak ; Carlyle Hamsher ; Mark C. Haigney ; Erich F. Wedam
The most feared drug-induced complication is fatal cardiac arrest. Torsades de pointes (TdP) is a polymorphic ventricular tachycardia occurring in the setting of a QT interval prolongation and is the most frequent type of drug-induced pro-arrhythmia. The most common mechanism of QT prolongation and TdP is blockade of the rapid component of the delayed rectifier repolarizing potassium conductance IKr. Anesthesiologists have extensive experience with QT prolonging drugs, but there are relatively few reports of TdP occurring in the perioperative setting. Nevertheless, regulatory concern regarding the drug droperidol resulted in a significant reduction in its use. Concern regarding two other agents that potently block IKr, i.e., sevoflurane and methadone, has grown, and practitioners are worried that these valuable agents may meet the same fate. In this review, the data regarding the TdP risk of droperidol, sevoflurane, and methadone are compared with particular emphasis on the different settings in which they are employed. While the three drugs are potent IKr inhibitors, little evidence exists to suggest that droperidol or sevoflurane are associated with significant proarrhythmia in the perioperative setting. Due to factors such as inhibition of the parasympathetic nervous system, prevention of hypoxia and hypercarbia, and attention to serum electrolytes, TdP is a very rare occurrence in the perioperative environment. Methadone, however, is typically given to outpatients, over long periods, and in combination with agents that inhibit its metabolism or are QT prolonging in their own right. Thus, pre- and post-drug electrocardiograms may be appropriate when prescribing methadone for outpatients, while the much lower risk for TdP (and the difficulties inherent in QT measurement in the perioperative period) render this approach unfruitful and worthy of reevaluation.
100 项与 B1R inhibitors(Alpha-9 Theranostics) 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 实体瘤 | 临床前 | 加拿大 | - |
登录后查看更多信息
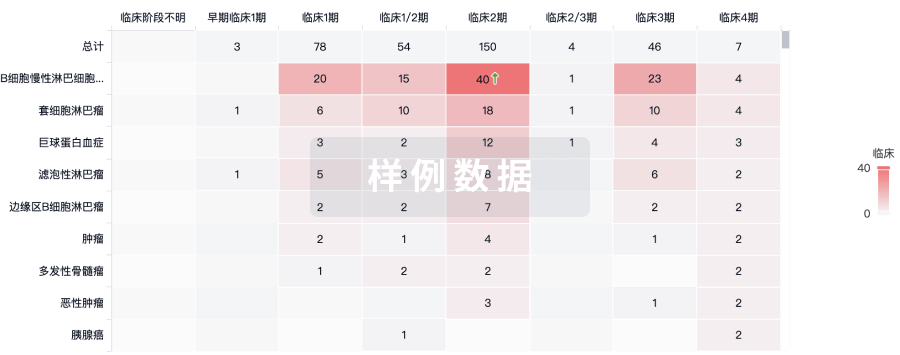
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
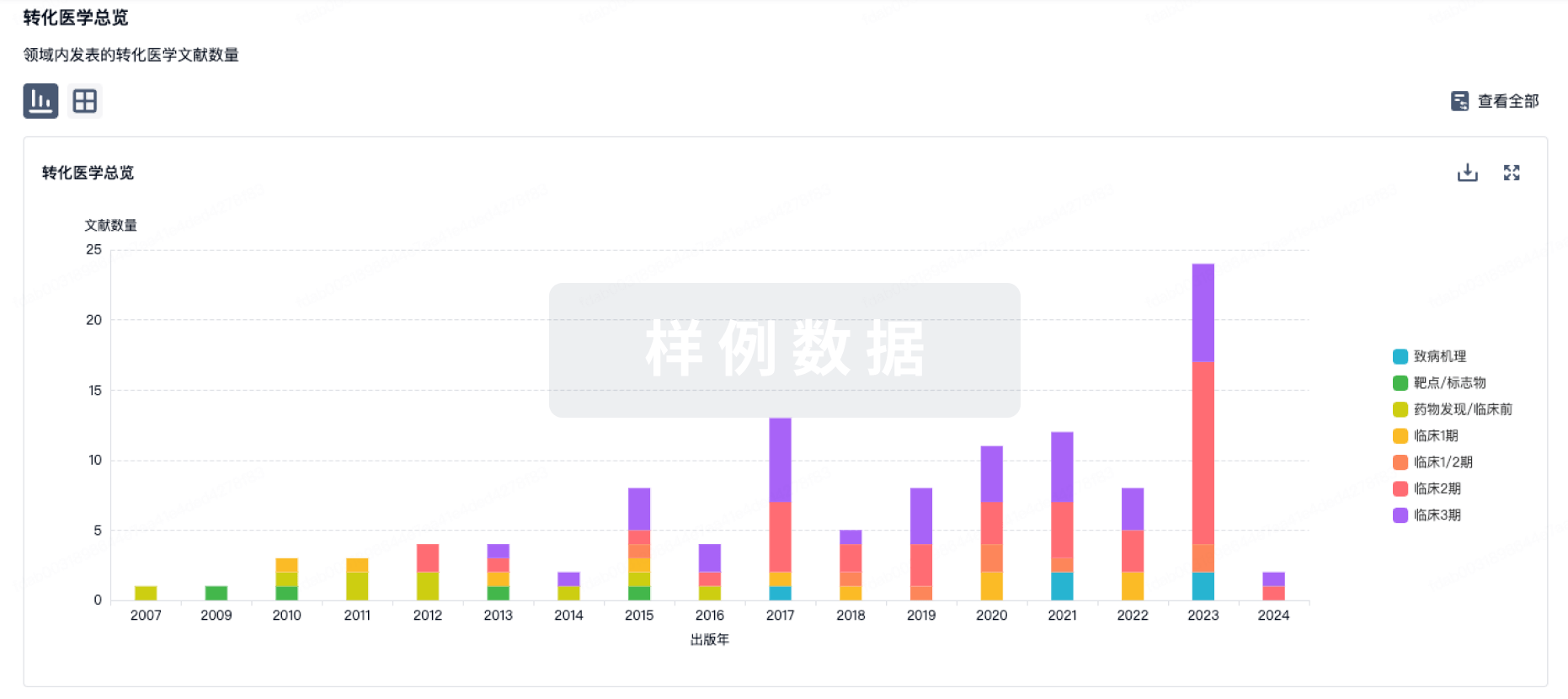
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
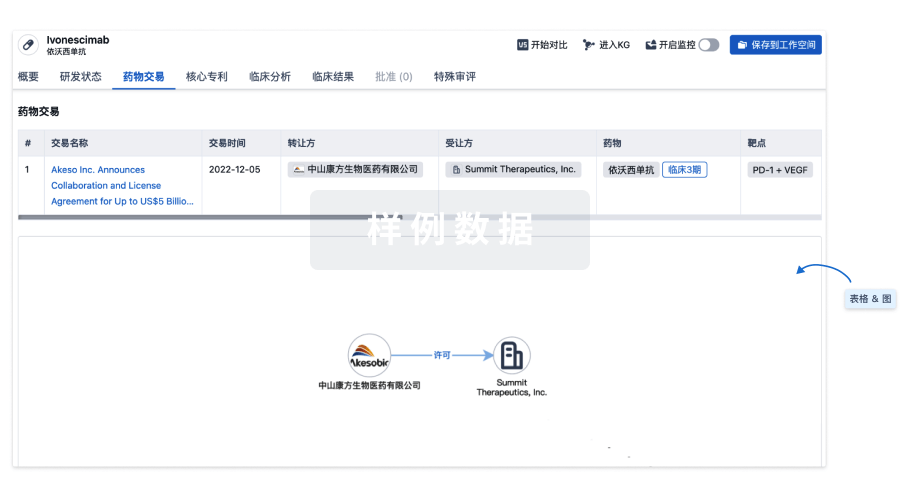
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
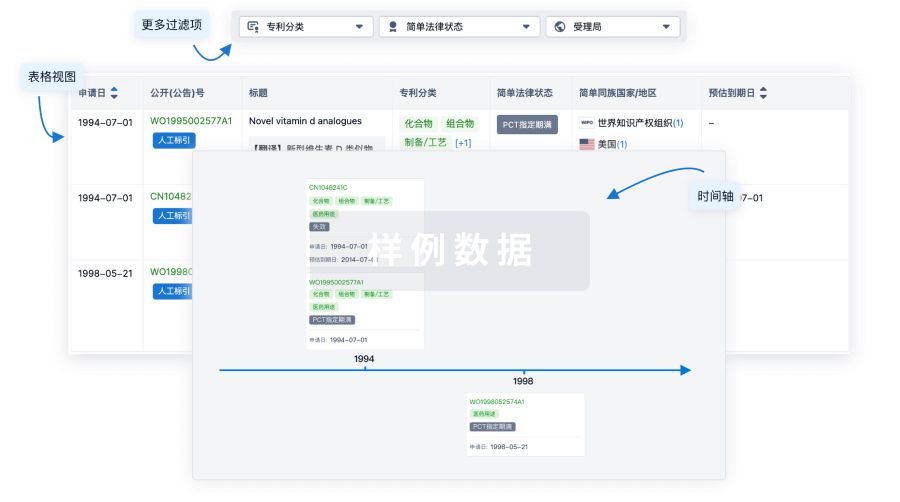
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
