预约演示
更新于:2025-08-31
NB-6253
更新于:2025-08-31
概要
基本信息
在研机构- |
权益机构- |
最高研发阶段无进展临床前 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
关联
100 项与 NB-6253 相关的临床结果
登录后查看更多信息
100 项与 NB-6253 相关的转化医学
登录后查看更多信息
100 项与 NB-6253 相关的专利(医药)
登录后查看更多信息
2
项与 NB-6253 相关的文献(医药)2022-12-01·Journal of translational medicine
Blocking TIGIT/CD155 signalling reverses CD8+ T cell exhaustion and enhances the antitumor activity in cervical cancer
Article
作者: Guo, Lingyu ; Sun, Yu ; Wang, Aihong ; Zhang, Junhua ; Liu, Lu ; Zhang, Youzhong ; Liu, Xiaoli ; Han, Sai
Abstract:
Objective:
TIGIT/CD155 has attracted widespread attention as a new immune checkpoint and a potential target for cancer immunotherapy. In our study, we evaluated the role of TIGIT/CD155 checkpoints in the progression of cervical cancer.
Methods:
The expression of CD155 and TIGIT in cervical cancer tissues was detected using flow cytometry, immunohistochemistry (IHC) and gene expression profiling. In vivo and in vitro experiments have proven that blocking TIGIT/CD155 restores the ability of CD8+T cells to produce cytokines. Changes in the NF-κB and ERK pathways were detected using western blotting (WB) after blocking TIGIT/CD155 signalling.
Results:
TIGIT expression was elevated in patients with cervical cancer. High TIGIT expression in CD8+T lymphocytes from patients with cervical cancer promotes the exhaustion of CD8+T lymphocytes. In addition, CD155 is expressed at high levels in cervical cancer tissues and is negatively correlated with the level of infiltrating CD8+T cells. We found that TIGIT, upon binding to CD155 and being phosphorylated, inhibited NF-κB and ERK activation by recruiting SHIP-1, resulting in the downregulation of cytokine production. Blocking TIGIT in activated CD8+T cells attenuates the inhibitory effect of SHIP-1 on CD8+T cells and enhances the activation of NF-κB and ERK. In vivo and in vitro experiments have proven that blocking TIGIT/CD155 restores the ability of CD8+T cells to produce cytokines. Injecting the blocking antibody TIGIT in vivo inhibits tumour growth and enhances CD8+T lymphocyte function. Treatment with a combination of TIGIT and PD-1 inhibitors further increases the efficacy of the TIGIT blocking antibody.
Conclusions:
Our research shows that TIGIT/CD155 is a potential therapeutic target for cervical cancer.
Biomedicines3区 · 工程技术
Targeting TIGIT for Immunotherapy of Cancer: Update on Clinical Development
3区 · 工程技术
ReviewOA
作者: Sahasranaman, Srikumar ; Budha, Nageshwar ; Rotte, Anand
Immune checkpoint blockers have dramatically improved the chances of survival in patients with metastatic cancer, but only a subset of the patients respond to treatment. Search for novel targets that can improve the responder rates and overcome the limitations of adverse events commonly seen with combination therapies, like PD-1 plus CTLA-4 blockade and PD-1/PD-L1 plus chemotherapy, led to the development of monoclonal antibodies blocking T-cell immunoglobulin and ITIM domain (TIGIT), a inhibitory checkpoint receptor expressed on activated T cells and NK cells. The strategy showed potential in pre-clinical and early clinical studies, and 5 molecules are now in advanced stages of evaluation (phase II and above). This review aims to provide an overview of clinical development of anti-TIGIT antibodies and describes the factors considered and thought process during early clinical development. Critical aspects that can decide the fate of clinical programs, such as origin of the antibody, Ig isotype, FCγR binding, and the dose as well as dosing schedule, are discussed along with the summary of available efficacy and safety data from clinical studies and the challenges in the development of anti-TIGIT antibodies, such as identifying patients who can benefit from therapy and getting payer coverage.
2
项与 NB-6253 相关的新闻(医药)2025-04-25
iStock,
LevKPhoto
Presentations at this year’s American Association for Cancer Research meeting could have a broad impact on the treatment landscape for head and neck and lung cancer, and implications for specific drug modalities like TIGIT and VEGF.
On Friday, the streets of Chicago, Illinois—or at least, the halls of its McCormick Place Convention Center—will flood with hundreds of cancer experts from across the country. The annual meeting of the American Association for Cancer Research is in town and will host some of the most cutting-edge research in oncology.
Expectations are great for this year’s event, which will host a few highly impactful readouts, some of which could have far-reaching implications for the market and for clinical practice.
One of the most anticipated presentations is a detailed readout for Merck’s Phase III
KEYNOTE-689 study
, which evaluated its blockbuster PD-1 blocker Keytruda in head and neck squamous cell carcinoma (HNSCC).
In October 2024, the pharma provided a
topline peek
at the study’s results, noting that Keytruda elicited a “statistically significant and clinically meaningful improvement” in event-free survival and a “trend toward improvement” in overall survival. Merck did not provide specific data at the time, and Keytruda’s magnitude of efficacy in this indication remains unknown.
However, in an April 16 note to investors, William Blair analysts said they expect KEYNOTE-689 to have “read-through to the broader” HNSCC treatment space.
“Merck’s Keynote-689 will have some impact on the head and neck cancer landscape/market opportunity,” William Blair analyst Matt Phipps added in an email to
BioSpace
. Detailed findings from the trial will likely affect other players in this space, including Merus and its bispecific antibody petosemtamab, and Exelixis, which is advancing its oral tyrosine kinase inhibitor zanzalintinib, according to Phipps.
Aside from HNSCC, AACR 2025 could also witness big readouts in lung cancer. In an email to
BioSpace
, AACR president-elect Lillian Siu, a senior medical oncologist at the Princess Margaret Cancer Centre in Toronto, pointed to Boehringer Ingelheim’s Beamion LUNG-1 trial, which she said could be “practice-changing.”
The trial is studying the HER2 tyrosine kinase blocker zongertinib in non-small cell lung cancer (NSCLC). A Phase Ib readout in September 2024 demonstrated a
66.7% objective response rate
for zongertinib, which also shrank tumors in 94% of treated patients. Boehringer Ingelheim is scheduled to release additional findings from
Beamion LUNG-1 on Monday
.
Modality Movement
Beyond specific indications, many experts also expect this year’s AACR cycle to be critical for certain treatment modalities.
“VEGF-PD(L)1 bispecifics took the oncology world by storm” in September last year, when Summit Therapeutics and Akeso announced that their investigational bispecific antibody ivonescimab
bested
Keytruda as a frontline NSCLC treatment, Christiana Bardon, co-managing partner at biotech investment firm MPM BioImpact, told
BioSpace
in an email.
The oncology partners doubled down on those results on Wednesday,
reporting
that the candidate yielded better progression-free survival than BeiGene’s PD-1 inhibitor Tevimbra in patients with NSCLC.
“Since [September 2024], a wave of VEGF bispecifics and some trispecifics have emerged,” according to Bardon, who noted that at least 13 of these, including ivonescimab and a candidate developed by Hangzhou DAC Biotechnology and dubbed
DXA023-G017
, will have data at AACR. “There’s even a VEGF bispecific with an [antibody-drug conjugate] payload,” she said.
Bardon is quick to caution, however, that assets playing in this space “are still mostly at the preclinical stage.” Nevertheless, “they have the potential to disrupt the oncology landscape if successful.”
William Blair’s Phipps, for his part, is focused on the TIGIT space, which in recent months has been battered by setbacks. Last month, for instance, BeiGene was
forced to scrap
its TIGIT asset ociperlimab after disappointing late-stage data, which showed that the candidate was “unlikely to meet the primary endpoint of overall survival” in the Phase III AdvanTIG-302 trial in NSCLC.
However, arguably the most high-pro belongs to Roche, which in November 2024 reported that its anti-TIGIT antibody tiragolumab
failed
the SKYSCRAPER-01 study in locally advanced unresectable or metastatic NSCLC, unable to significantly improve overall survival versus placebo.
The pharma did not provide specific data at the time, but will do so at AACR. A more comprehensive readout “will have important implications for the TIGIT inhibitor space,” Phipps said. “We know the trial failed, but are there reasons that can be gleaned that might allow others to still succeed with this target?”
Among these other players are partners Gilead and Arcus, which in November 2024 reported Phase III data showing that their anti-TIGIT antibody domvanalimab, when combined with the PD-1 therapy zimberelimab,
significantly boosted
progression-free and overall survival in patients with a specific type of NSCLC, as compared with zimberelimab or chemotherapy.
The partners are also trialing the domvanalimab-zimberelimab combo in HSNCC, for which they are scheduled to
present mid-stage data
at AACR.
临床结果临床3期AACR会议临床1期ASCO会议
2022-02-17
BeiGene (NASDAQ: BGNE; HKEX: 06160; SSE: 688235), a global, science-driven biotechnology company focused on developing innovative and affordable medicines, today announced that BeiGene’s BTK inhibitor BRUKINSA (zanubrutinib) received approval from Swissmedic for the treatment of adult patients with Waldenström’s macroglobulinemia (WM) who have received at least one prior line of therapy, or for treatment-naïve patients who are not suited for standard chemo-immunotherapy. BRUKINSA had previously been granted orphan drug status.
“The authorization of BRUKINSA will bring a new option and an innovative medicine that has potential to offer deep and durable response for eligible patients with WM in Switzerland," said Pr. Davide Rossi, Deputy Head of the Division of Hematology of the Oncology Institute of Southern Switzerland IOSI. “BRUKINSA is a next-generation BTK inhibitor which has also provided meaningful improvements in tolerability for some patients with WM compared to ibrutinib, as treatment discontinuation remains a concern.”
Reto Kessler, Country Manager, Switzerland at BeiGene added, “This approval is a significant development for people living with WM in Switzerland and for BeiGene’s expansion in Europe. Our teams are committed to collaborating with the Federal Office of Public Health and healthcare professionals to ensure access to BRUKINSA for patients in Switzerland.”
The Marketing Authorization Application (MAA) is supported by data from the global Phase 3 ASPEN clinical trial, a Phase 3 randomized, open-label, multicenter trial (NCT03053440) that evaluated BRUKINSA compared to ibrutinib in patients with relapsed/refractory (R/R) or treatment-naïve (TN) WM who harbor a MYD88 mutation (MYD88MUT). In the ASPEN trial, BRUKINSA demonstrated a numerically higher very good partial response (VGPR) rate and a favorable safety pro ibrutinib, although the primary endpoint of statistical superiority related to deep response (VGPR or better) was not met. As assessed by independent review committee (IRC) per adaptation of the response criteria updated at the Sixth International Workshop on Waldenström’s Macroglobulinemia (IWWM), the combined complete response (CR) + VGPR rate in the overall intention-to-treat (ITT) population was 29% with BRUKINSA (95% CI: 20, 40), compared to 19% with ibrutinib (95% CI: 12, 30).
In the ASPEN trial, of the 101 patients with WM randomized and treated with BRUKINSA, four percent of patients discontinued due to adverse events, including cardiomegaly, neutropenia, plasma cell myeloma, and subdural hemorrhage. Adverse events leading to dose reduction occurred in 14% of patients, with the most common being neutropenia (3%) and diarrhea (2%).
The recommended dose of BRUKINSA is either 160 mg twice daily or 320 mg once daily, taken orally with or without food. The dose may be adjusted for adverse reactions and reduced for patients with severe hepatic impairment and certain drug interactions.
About Waldenström’s Macroglobulinemia
WM is a rare B-cell lymphoma that occurs in less than two percent of patients with non-Hodgkin lymphomas.2 The disease usually affects older adults and is primarily found in bone marrow, although lymph nodes and the spleen may be involved.1 Throughout Europe, the estimated incidence rate of WM is approximately seven for every one million men and four for every one million women.2
About BRUKINSA
BRUKINSA is a small molecule inhibitor of Bruton’s tyrosine kinase (BTK) discovered by BeiGene scientists that is currently being evaluated globally in a broad clinical program as a monotherapy and in combination with other therapies to treat various B-cell malignancies. Because new BTK is continuously synthesized, BRUKINSA was specifically designed to deliver complete and sustained inhibition of the BTK protein by optimizing bioavailability, half-life, and selectivity. With differentiated pharmacokinetics compared to other approved BTK inhibitors, BRUKINSA has been demonstrated to inhibit the proliferation of malignant B cells within a number of disease relevant tissues.
BRUKINSA is supported by a broad clinical program which includes more than 3,900 subjects in 35 trials across 28 markets. To date, BRUKINSA has received more than 20 approvals covering more than 40 countries and regions, including the U.S., European Union, China, Australia, Great Britain and Switzerland. Currently, more than 40 additional regulatory submissions are in review around the world.
BeiGene Oncology
BeiGene is committed to advancing best- and first-in-class clinical candidates internally or with like-minded partners to develop impactful and affordable medicines for patients across the globe. We have a growing R&D and medical affairs team of approximately 2,900 colleagues dedicated to advancing more than 100 clinical trials that have involved more than 14,500 subjects. Our expansive portfolio is directed predominantly by our internal colleagues supporting clinical trials in more than 45 countries and regions. Hematology-oncology and solid tumor targeted therapies and immuno-oncology are key focus areas for the Company, with both mono- and combination therapies prioritized in our research and development. BeiGene currently has three approved medicines discovered and developed in our own labs: BTK inhibitor BRUKINSA in the United States, China, the EU and U.K., Canada, Australia and additional international markets; and the non-FC-gamma receptor binding anti-PD-1 antibody tislelizumab as well as the PARP inhibitor pamiparib in China.
BeiGene also partners with innovative companies who share our goal of developing therapies to address global health needs. We commercialize a range of oncology medicines in China licensed from Amgen, Bristol Myers Squibb, EUSA Pharma and Bio-Thera. We also plan to address greater areas of unmet need globally through our other collaborations including with Mirati Therapeutics, Seagen, and Zymeworks.
In January 2021 BeiGene and Novartis announced a collaboration granting Novartis rights to co-develop, manufacture, and commercialize BeiGene’s anti-PD1 antibody tislelizumab in North America, Europe, and Japan. Building upon this productive collaboration, including a biologics license application (BLA) under FDA review, BeiGene and Novartis announced an option, collaboration and license agreement in December 2021 for BeiGene’s TIGIT inhibitor ociperlimab that is in Phase 3 development. Novartis and BeiGene also entered into a strategic commercial agreement through which BeiGene will promote five approved Novartis Oncology products across designated regions of China.
About BeiGene
BeiGene is a global, science-driven biotechnology company focused on developing innovative and affordable medicines to improve treatment outcomes and access for patients worldwide. With a broad portfolio of more than 40 clinical candidates, we are expediting development of our diverse pipeline of novel therapeutics through our own capabilities and collaborations. We are committed to radically improving access to medicines for two billion more people by 2030. BeiGene has a growing global team of over 8,000 colleagues across five continents.
References:
1 Lymphoma Research Foundation. Getting the Facts: Waldenström Macroglobulinemia. Accessed July 2021. Available at
2 Buske, C, et al. Treatment and outcome patterns in European patients with Waldenström’s macroglobulinaemia: a large, observational, retrospective chart review. The Lancet Haematology 2018; 5: e0299-309.
合作抗体孤儿药First in Class免疫疗法
100 项与 NB-6253 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 肿瘤 | 临床前 | 加拿大 | 2017-11-09 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

生物类似药
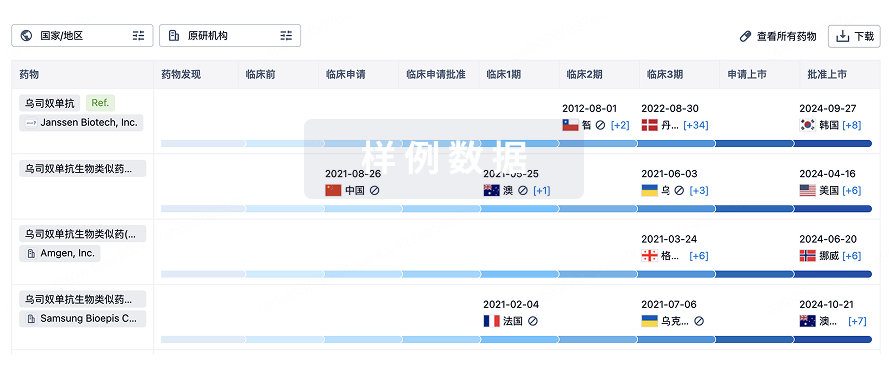
生物类似药在不同国家/地区的竞争态势。请注意临床1/2期并入临床2期,临床2/3期并入临床3期
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或

Eureka LS:
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
