更新于:2024-06-30
Bropirimine
溴匹立明
更新于:2024-06-30
概要
基本信息
原研机构 |
在研机构- |
最高研发阶段终止申请上市 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
结构
分子式C10H8BrN3O |
InChIKeyCIUUIPMOFZIWIZ-UHFFFAOYSA-N |
CAS号56741-95-8 |
关联
100 项与 溴匹立明 相关的临床结果
登录后查看更多信息
100 项与 溴匹立明 相关的转化医学
登录后查看更多信息
100 项与 溴匹立明 相关的专利(医药)
登录后查看更多信息
347
项与 溴匹立明 相关的文献(医药)2024-01-25·Journal of medicinal chemistry1区 · 医学
PROTACs Targeting BRM (SMARCA2) Afford Selective In Vivo Degradation over BRG1 (SMARCA4) and Are Active in BRG1 Mutant Xenograft Tumor Models
1区 · 医学
Article
作者: Ye, Xiaofen ; Quinn, Connor ; Berlin, Michael ; Chen, Huifen ; Harbin, Alicia ; Li, Limei ; Cantley, Jennifer ; Pizzano, Jennifer ; Wang, Jing ; Hamman, Brian D ; Chan, Emily W ; Davenport, Kim ; Soto, Leofal ; DiNicola, Dean ; Gordon, Debbie ; Staben, Leanna R ; Pérez-Dorado, Inmaculada ; Cheng, Yunxing ; He, Mingtao ; Januario, Thomas ; Rose, Christopher M ; Tian, Qingping ; Hole, Alison J ; Bookbinder, Mark ; Ye, Crystal S ; Sun, Hongming ; Wang, Weifeng ; Zhang, Penghong ; Dragovich, Peter S ; Bortolon, Elizabeth ; Yauch, Robert ; Chen, Xin ; Cheung, Tommy K ; Broccatelli, Fabio ; Merchant, Mark ; Koenig, Stefan G ; Rousseau, Emma ; Haskell, Roy ; Cadelina, Greg ; Kerry, Philip S ; Zhou, Yuhui
The identification of VHL-binding proteolysis targeting chimeras (PROTACs) that potently degrade the BRM protein (also known as SMARCA2) in SW1573 cell-based experiments is described. These molecules exhibit between 10- and 100-fold degradation selectivity for BRM over the closely related paralog protein BRG1 (SMARCA4). They also selectively impair the proliferation of the H1944 "BRG1-mutant" NSCLC cell line, which lacks functional BRG1 protein and is thus highly dependent on BRM for growth, relative to the wild-type Calu6 line. In vivo experiments performed with a subset of compounds identified PROTACs that potently and selectively degraded BRM in the Calu6 and/or the HCC2302 BRG1 mutant NSCLC xenograft models and also afforded antitumor efficacy in the latter system. Subsequent PK/PD analysis established a need to achieve strong BRM degradation (>95%) in order to trigger meaningful antitumor activity in vivo. Intratumor quantitation of mRNA associated with two genes whose transcription was controlled by BRM (PLAU and KRT80) also supported this conclusion.
2023-12-01·Clinical breast cancer
Triple Negative Breast Cancer and Brain Metastases
Review
作者: Van Swearingen, Amanda E D ; Bansal, Rani ; Anders, Carey K
The treatment of metastatic breast cancer (MBC) has improved over the past decade, however prognosis continues to be mitigated by the fact that about 1 in 5 patients with MBC will develop brain metastases (BrM) during their metastatic disease course. 1 This number is even higher for patients with triple-negative breast cancer (TNBC), with studies showing as high as 40% of patients developing BrM. 2, 3 Studies have shown that TNBC portends a worse survival after a diagnosis of BrM compared with non-TNBC subtypes. 4 Given the unique location and biologic properties of BrM, treatment options have historically been limited. Challenges to the treatment of TNBC BrM include a lack of targeted therapies and difficulties in delivery of drug to the brain past the blood-brain barrier (BBB). Herein, we will review the advances in local and systemic therapies to most effectively treat patients with TNBC BrM, including therapies on the horizon currently in clinical trials.
2023-07-09·Journal of chromatographic science
Normal-Phase TLC and Gradient Reversed-Phase HPLC for the Simultaneous Determination of Enrofloxacin and Bromhexine HCl in Presence of Two of Their Official Impurities
Article
作者: Tantawy, Mahmoud A ; Abou Al-Alamein, Amal M ; Mohamed, Ola Y ; Abdulla, Shaaban A
Abstract:
In this work, two chromatographic methods are developed and validated for the determination of enrofloxacin and bromhexine (BRM) HCl in the presence of two of their specified impurities, ciprofloxacin and BRM impurity C. The suggested chromatographic methods included the use of thin layer chromatography (TLC-densitometry) and high-performance liquid chromatography (HPLC). In case of TLC-densitometry, good separation was achieved by using mobile phase of n.butanol:acetone:water:glacial acetic acid:triethylamine (10:3:1:0.5:0.5, by volume) on silica gel stationary phase at 254-nm detection. The developed HPLC method used BDS HYPERSIL C18 column with a mobile phase of water:acetonitrile:methanol:triflouroacetic acid. A linear gradient elution of 75–10%, 20–50% and 5–40% for water, acetonitrile and methanol, respectively, was applied in 13 min at a flow rate of 1.5 mL min−1. These methods were sufficient to separate the four substances simultaneously, and they are validated as per International Conference on Harmonization guidelines.
21
项与 溴匹立明 相关的新闻(医药)2024-05-28
·精准药物
侃言单价分子胶降解剂靶向降解蛋白是消除致病蛋白一种强大的治疗方式。然而,合理设计分子胶降解剂非常具有挑战性。上周聊了聊通过DNA编码化合物库筛选技术合理设计、筛选分子胶降解剂的实例,详见👉 🔗本月中旬美国加州大学伯克利分校Danniel教授课题组在ACS Cent. Sci.(IF ~18.2/Q1)上报道了其关于分子胶靶向降解剂的最新研究。这是一篇非常有意思的研究,Danniel教授发现了一种可以移植安装在不同靶点配体上的共价弹头手柄——乙烯基磺酰基哌嗪,能够将非降解剂配体转化为有效降解其对应靶蛋白的共价降解剂。省流,请看下图,研究人员首先发现了有效、选择性降解BRD4的共价分子胶降解剂;并发现其共价弹头手柄可以移植安装到多种蛋白质靶向配体上,能够有效降解其靶蛋白,包括CDK4、AR、BTK、SMARCA2 /4、BCR-ABL/c-ABL等。1、确定促使降解BRD4的共价手柄2、定位BRD4降解的E3连接酶3、探索共价手柄对其它靶标的适用性接下来展开聊聊研究过程。01确定促使BRD4降解的共价手柄共价分子胶降解剂:近期有多项研究表明,可以通过细微的化学修饰,给非降解剂配体安装上共价手柄,可以将其转变成共价分子胶降解剂:配体靶向POI,共价手柄“连锁”E3连接酶残基形成共价键,进而使POI降解,如下图。这些研究中,已经确定了各种共价手柄靶向E3连接酶底物受体DCAF16、或DCAF11的半胱氨酸残基(Cys)。因此,可以基于POI已知的配体、和E3连接酶受体(如DCAF16)共价靶向手柄,合理设计共价单价分子胶降解剂。而且,共价分子胶降解剂还可以加强POI与E3连接酶受体之间的存在的微弱的相互作用,进而降解POI。如上图,研究人员首先使用BET家族抑制剂JQ1作为测试模板,合成了一系列具有各种亲的弹头手柄的共价JQ1类似物。这些共价弹头手柄可以与E3连接酶受体上的半胱氨酸、赖氨酸、或其它亲核氨基酸残基反应,以降解BRD4。研究人共合成、测试了18个JQ1共价衍生物,最终,只有化合物ML1-50会同时导致HEK293 T细胞中BRD4的长型和短型消失。其共价弹头手柄为乙烯基磺酰基哌嗪。ML1-50以剂量依赖的方式降低BRD4水平,在HEK293T细胞中优先降解短型BRD4亚型,并具有nM级别效力。研究人员还研究了ML1-50在不同细胞类型、不同靶标(包括BRD4的同亚族)的POI降解效力,证明其针对BRD4的特异性。接下来,研究人员设计方案,确定化合物ML1-50降解BRD4的E3连接酶的受体。02定位BRD4降解的E3连接酶为了确定化合物ML1-50降解BRD4的E3连接酶受体,首先合成了一个包含炔烃功能基团、和乙烯基磺酰基哌嗪手柄的探针——化合物ML2-33,如下图。这个探针比化合物ML1-50更加简单,没有潜在的配体连接,理论上它比化合物ML1-50更加混杂,因此,研究人员针对化合物ML2-33处理的细胞,设计了竞争性下拉蛋白质组学实验,寻找被化合物ML1-50预处理后,明显消失的蛋白。化合物ML2-33下拉蛋白质组学实验得到6259个蛋白,其中有133钟蛋白分布在不同的蛋白类别中,被过量的ML1-50预处理显著胜过。在这些ML1-50预处理显著胜过的靶标中,只有一种E3连接酶是Cullin E3连接酶家族中的一部分,可以被NEDD8-DCAF16调控。利用基于竞争活性的蛋白质谱分析(ABPP),通过半胱氨酸反应性探针,在原位ML1-50靶标结合与非原位全蛋白质组半胱氨酸标记的竞争,显示对照DCAF16细胞的C119与ML1-50处理的探针修饰的胰蛋白酶肽的比率为1.4,表明该半胱氨酸的衔接率为28%。为了进一步证实ML1-50与DCAF16的直接相互作用,研究人员用ML1-50取代了半胱氨酸活性探针,用基于凝胶的ABPP方法标记了纯化的DCAF16蛋白,而不会引起蛋白的任何沉淀。ML1-50介导的BRD4降解可以被过量JQ1预处理的细胞所取代,并且用共价烷基功能化手柄ML2-33单独处理的细胞不会改变BRD4水平,这表明BRD4的损失可能是通过与BRD4的特异性相互作用,而不是通过共价手柄的非特异性作用。同时,研究人员还证明了,可以通过不同的E3连接酶,针对同一靶标的不同亲电降解物。接下来,探索乙烯基磺酰基哌嗪共价手柄对其它靶标的普适性。03探索共价手柄对其它靶标的普适性很多的关于降解剂的研究都是基于BRD4进行研究,这是因为BRD4是一个非常典型的靶向蛋白降解的模型,它对靶向蛋白降解非常的敏感,因此,常被用来作为POC验证的模型。但是,BRD4共价降解剂的成功,并不能说明这个共价弹头手柄就具有其它靶标的更广泛的普适性了。需要进一步的验证,这种在非降解剂配体上安装共价弹头手柄,进而将其转化为特异性靶向的分子胶共价降解剂的靶点普适性。此外,先前的研究已经表明共价和非共价BRD4分子降解剂通过DCAF16起作用,通过加强DCAF16和BRD4之间存在的弱相互作用。亦可以通过共价招募DCAF16,生成针对BRD4以外的其它靶标的异源双功能的PROTACs降解剂。然而,ADCF16靶向共价手柄是否可以广泛移植到其它POI配体上,以产生BRD4以外的新底物蛋白的无连接、单价降解物尚不清楚。因此,本项研究首先将乙烯基磺酰基哌嗪手柄抑制到临床批准的CDK4/6抑制剂Ribociclib上,生成化合物ML1-71,如下图。尽管与ML1-50降解BRD4相比不太明显,但ML1-71能够在细胞依赖剂量显著地降解CDK4,如上图。尽管这种降解剂只有中等效力,但是可以观察到CDK4降解在DCAF16敲除细胞显著衰减。没有发现ML2-33处理对CDK4的降解,也没有观察到ML1-77处理对CDK4的细胞毒性。基于同样的考虑,研究人员也生成了针对SMARCA2/4的安装有乙烯基磺酰基哌嗪共价手柄的单价降解剂ML1-96。ML1-96能够显著降解MV-4-11白血病癌细胞中的SMARCA2、SMARCA4,并且这种降解作用在DCAF16敲除的细胞中显著减弱。同样,ML2-33不影响SMARCA2/4的水平,以及ML1-96对细胞没有毒性。将这种策略应用于雄激素受体(AR)单价降解剂,得到化合物ML2-9,同样降解效果显著,对细胞没有毒性。应用于BCR-ABL/c-ABL、以及BTK生成ML2-5、TH1-9,也证明了这种策略的可行性。总结与展望:总的来说,Daniel教授这种策略对于合理设计分子胶靶向降解剂非常具有参考意义。方法中涉及到共价手柄的探索、E3连接酶受体的探索,以及共价手柄在不同POI中的适用性等。证实了通过共价化学处理E3连接酶受体设计POI降解物的可行性。同时,也可以发现,每个降解物仍然还是表现出了不同程度的脱靶降解现象,可能表明蛋白质靶向配体在使用的浓度下脱靶或由共价降解处理引起的脱靶效应。因此,在后续的进一步药化的研究工作中,需要注意,进一步提高共价降解剂的手柄的选择性。更多详细的实验方案,请阅读原文(开源)。参考文献DCAF16-Based Covalent Handle for the Rational Design of Monovalent Degraders. ACS Central Science,2024, in printDOI: 10.1021/acscentsci.4c00286声明:发表/转载本文仅仅是出于传播信息的需要,并不意味着代表本公众号观点或证实其内容的真实性。据此内容作出的任何判断,后果自负。若有侵权,告知必删!长按关注本公众号 粉丝群/投稿/授权/广告等请联系公众号助手 觉得本文好看,请点这里↓
蛋白降解靶向嵌合体
2024-05-26
最近,Nature报告了一种新的基于氧化还原的色氨酸生物共轭策略,使用氧杂氮丙啶试剂进行环化色氨酸化学连接(Trp-CLiC),以实现高效和特异性的色氨酸标记[1]。色氨酸是蛋白质中罕见的氨基酸,仅占观察到的氨基酸的1%,但由于其独特的化学性质,它在生物学中占有特殊的地位。作为生物合成过程中能量成本最高的氨基酸,色氨酸经常富集在蛋白质的功能位点,有助于蛋白质折叠和信号转导等过程[2, 3]。研究团队通过开发这种新方法,不仅提高了色氨酸标记的效率和特异性,而且还能够揭示色氨酸残基在蛋白质功能中的多样作用,尤其是在调节蛋白质介导的相分离过程中的功能位点。研究团队首先介绍了传统的蛋白质生物共轭化学方法,这些方法通常针对半胱氨酸和赖氨酸残基,因为它们具有较高的亲核性。然而,色氨酸的亲核性相对较弱,这使得开发一种在生理条件下有效的色氨酸生物共轭反应成为一个挑战。为了克服这一难题,研究人员从吲哚生物碱的生物合成途径中得到启发,设计了一种模拟氧化环化反应的新型氧化剂——N-磺酰基氧杂氮丙啶(N-sulfonyl oxaziridine),用于色氨酸的选择性功能化。(图1)图1: Trp-CLiC 策略模拟吲哚类生物碱的生物合成研究人员通过计算18类氧杂氮丙啶的最低未占据分子轨道(LUMO)能量,发现N-磺酰基氧化哌啶具有最高的电子接受能力,从而成为最有效的色氨酸氧化环化试剂(图2)。通过一系列化学反应筛选和优化,研究团队最终确定了几种具有高反应活性和选择性的氧杂氮丙啶试剂,并利用这些试剂成功地在多肽和蛋白质上进行了色氨酸标记。图2:18 类氧化哌啶的 8 个代表性结构的分子轨道计算为了验证Trp-CLiC方法的实用性,研究人员在多种蛋白质和蛋白质组水平上进行了评估。他们选择了具有单个色氨酸残基的GLP-1肽和IL-8蛋白作为模型底物,展示了Trp-CLiC试剂在肽和蛋白质标记中的高效性(图3)。图3:Ox-W18是 GLP-1 肽标记最有效的N-磺酰基氧化哌啶探针此外,研究人员还使用Trp-CLiC方法对溶菌酶(lysozyme)进行了标记,并发现该方法能够快速、特异性地标记蛋白质中的色氨酸残基(图4)。图4 Trp-CLiC标记溶菌酶的反应动力学分析研究人员进一步探讨了Trp-CLiC标记的化学产物的稳定性,发现在不同的水溶液介质和pH条件下,标记的色氨酸残基显示出良好的稳定性。此外,通过在HEK293T细胞裂解液中测试Trp-CLiC的蛋白质组选择性,研究人员证实了Ox-W18试剂对色氨酸具有高度的选择性(图5)。总而言之,这些数据表明Trp-CLiC是跨肽、蛋白质和蛋白质组水平的色氨酸生物共轭的有效策略,其中N-磺酰基氧杂氮丙啶衍生物成为促进色氨酸氧化环化反应产生相应吲哚环加合物产物的特殊试剂支架。图5:与其他竞争性氨基酸相比,Trp-CLiC 方法对色氨酸标记表现出高选择性文章还详细描述了Trp-CLiC在全蛋白质组水平上的应用,通过活性基团蛋白质组学(ABPP)策略,研究人员能够系统地鉴定细胞中新的功能性色氨酸位点。他们利用Ox-W18探针处理HEK293T细胞裂解液,并结合铜介导的炔-叠氮环加成点击化学,富集标记的蛋白质,并通过串联质谱(LC-MS/MS)分析鉴定了探针修饰的多肽。通过这种方法,研究人员鉴定了906个独特的高反应性色氨酸位点,涉及591个被修饰的蛋白质。这些蛋白质广泛分布于多种蛋白质类别中,包括酶、分子伴侣和核蛋白,以及许多结构蛋白。研究人员还发现,许多被Trp-CLiC标记的蛋白质高度富集在多样的相分离区室中,如核仁、核体、paraspeckle和应激颗粒。(图6)图6:使用 Trp-CLiC 系统鉴定蛋白质组中反应性色氨酸残基的示意图此外,研究人员还探讨了色氨酸参与的阳离子-π相互作用,这是一种在化学和生物学中普遍存在的非共价相互作用。通过AlphaFold结构筛查,研究人员预测了Trp-CLiC ABPP数据集中的阳离子-π对,并发现这些相互作用与蛋白质的溶剂可及性密切相关。研究人员还发现,这些高反应性色氨酸位点在蛋白质的相分离行为中发挥了功能作用,特别是NONO蛋白中的Trp271位点,其突变显著影响了NONO介导的相分离和paraspeckle形成。最后,研究人员探讨了通过翻译后修饰调控色氨酸参与的阳离子-π相互作用的可能性。他们以NPM1蛋白为模型,研究了色氨酸和赖氨酸残基的突变对NPM1相分离和细胞分布的影响,并发现这些阳离子-π对可以通过赖氨酸的翻译后修饰进行功能调节。Comments:本研究开发了一种新的色氨酸标记方法,可以对蛋白质中的色氨酸侧链进行选择性、快速和高效的化学修饰。不仅提高了标记的效率和特异性,而且为研究蛋白质功能、蛋白质组学分析和疾病相关蛋白质的靶向治疗提供了新的工具和策略。通过Trp-CLiC方法,研究人员能够系统地鉴定和分析蛋白质中的功能性色氨酸位点,揭示了这些位点在蛋白质介导的相分离和细胞功能中的重要作用。参考文献[1] Xie X, Moon PJ, Crossley SWM, Bischoff AJ, He D, Li G, Dao N, Gonzalez-Valero A, Reeves AG, McKenna JM, Elledge SK, Wells JA, Toste FD, Chang CJ. Oxidative cyclization reagents reveal tryptophan cation-π interactions. Nature. 2024 Mar;627(8004):680-687.[2] Barik, S. The uniqueness of tryptophan in biology: properties, metabolism, interactions and localization in proteins. Int. J. Mol. Sci. 21, 8776–8797 (2020).[3] Murthy, S. N. et al. Conserved tryptophan in the core domain of transglutaminase is essential for catalytic activity. Proc. Natl Acad. Sci. USA 99, 2738–2742 (2002).识别微信二维码,添加生物制品圈小编,符合条件者即可加入生物制品微信群!请注明:姓名+研究方向!版权声明本公众号所有转载文章系出于传递更多信息之目的,且明确注明来源和作者,不希望被转载的媒体或个人可与我们联系(cbplib@163.com),我们将立即进行删除处理。所有文章仅代表作者观点,不代表本站立场。
诊断试剂
2024-05-12
·同写意
本次大会以“创新工艺,成就医药产业未来”为主题,设1场主旨会议与3场平行分会,汇聚50余位来自抗体、ADC、GLP-1与多肽领域的专业讲者,聚焦下游工艺,进阶分离纯化新技术方法,共话医药产业未来!被称作魔法子弹的ADC近几年风光无限,这类药物的设计思路很清楚,就是利用抗原与抗体的高特异结合能力将毒素选择性递送到抗原阳性细胞中选择性杀伤目标细胞、从而减少对正常细胞的杀伤。但是和其它药物一样,ADC到了真正的战场上如何表现并不一定与设计思路完全一致。随着越来越多数据的出现,关于成功ADC真实工作机制的图像也越来越清晰。这是一类非常复杂的药物,所以没有一个单一研究能充分描述ADC机制。如同盲人摸象的故事,我们需要把很多研究结果综合起来才能得到一个相对完整的图像。这篇文章我会把近期的一些重要数据列出,读者可以把这些点链接起来、看看大家能看到一类什么样的抗癌药物。1ADC正在从分子靶向递送向组织靶向演化ADC的本质是一个前药,本身没有活性(除了抗体介导疗效外)、只有在胞内释放毒素后才会有杀伤力。这个分布次序与传统小分子药物(先接触正常细胞然后分布到肿瘤)相反,本身对于肿瘤杀伤就是一个优势。当然更重要的是这个激活策略令靶向杀伤成为可能,如果靶点抗原表达足够特异、那么携带超级杀手的ADC只需要极低剂量就可以精准杀死肿瘤细胞而不骚扰正常细胞的日常生活。但高度特异的肿瘤抗原很少见,多多少少在正常组织都有一定表达。而肿瘤相对对于人体是个微小组织,所以多数抗体在低剂量主要被正常组织靶点吸收了,这也是诊断抗体显影剂通常要与抗体联用的原因、也有人用单抗联用解决结合位点障碍问题(binding site barrier)。另外抗体非特异结合、内吞势力也十分猖獗,毕竟抗体不能永远在系统内游荡、所以机体演化出一整套不依赖抗原的抗体代谢清除机制。ADC也不是法外之地,同样会在规定时间、规定地点被降解清除。还有一个非常重要的因素是虽然肿瘤抗原表达量可能很高,但是能够与药物接触的抗原通常是有限的,因为肿瘤组织的残缺血管结构和细胞间的紧密链接。用抗体显影剂诊断肿瘤有很高实用价值但是目前为止除了几个多肽显影剂外,基于抗体的显影剂还没有上市使用的。一个原因是肿瘤抗原本身特异性有限,背景噪音比较大。另一个重要原因则是肿瘤组织太混乱,大分子进入比较难。曲妥珠显影剂在1/3 HER2阳性病灶无法显影,而在肝肾脾等无HER2组织却大量蓄积。可以想象如果曲妥珠携带的不是显影剂而是payload治疗窗口会怎样。肿瘤抗原的隐蔽性加上正常组织在靶、脱靶抗体结合的干扰导致目前ADC导航能力不足,进而造成99%以上的ADC无法找到肿瘤组织、被迫在正常组织释放毒素。ADC与小分子前药有个本质区别,就是本身因为分子量太大无法通过肾排泄、必须经过降解才能把毒素释放。半衰期一周的ADC意味着一周之内无论它找到多少抗原都得释放50%的毒素,如同一个城市的垃圾站容量有限、也很难找到,市民又不能不到垃圾、所以市容很难保证。即使在微环境内,肿瘤细胞也不总是唯一的毒素释放细胞,有很多非特异的释放机制可能参与了ADC的毒素释放,这也淡化了靶点介导效应的治疗价值。因此高毒性ADC并不成功,只是在毒性较低、并可以自由进出细胞的毒素如DXd出现后ADC才真正进入主流。对于可过膜毒素而言胞内释放不等于胞内定居,而是会扩散到微环境其它细胞。如果在肿瘤组织释放就会把杀伤力扩大到没有靶点抗原表达的肿瘤细胞、即所谓的旁观者杀伤,所以疗效会更好。但是这个性质是把双刃剑,正常细胞里释放的毒素也会同样扩散到其它正常细胞、造成更大范围杀伤,你不能socialize肿瘤杀伤的同时指望capitalize正常组织损伤。临床上ADC在靶、脱组织毒性比较罕见,同一毒素无论做成靶向什么抗原的ADC它们毒性谱和最高耐受剂量都差不多,而且都与毒素本身很接近,说明无论毒素从哪里释放最后的毒性都是差不多的、会被整个compartment socialized。DXd ADC都脱发,MMAF ADC都有眼毒性。当然有可能有在靶毒性的ADC在开发过程中被淘汰了,但是根据我对临床前评价体系的了解这个可能性不是很大。疗效方面情况类似,虽然有些ADC在抗原高表达人群收益更大、但多数ADC在多数应用场景应答与靶点表达关系较弱。多数ADC没有可靠的预测疗效生物标记,目前全球上市了15个ADC产品,只有3款要求用靶点抗原检测选择患者。有些高密度抗原如HER2可能在阴性肿瘤也有一定表达、所以还有进入细胞的机会,但正常组织如肾可以表达IHC3+水平的HER2。这不是靶向递送、而是把正常组织当作人体盾牌了(当然不同毒素对不同正常组织杀伤力不同),魔法子弹先打正常组织再打肿瘤都有疗效那还不如直接把DXd达到静脉里了。造成这个结果有几个理论上的可能性。一是小分子化疗药物的扩散速度通常远快于细胞杀伤速度。传统化疗药物一般是滴注到患者静脉,但药物不会在血液里停留很长时间、而是会在较短时间内分布到全身组织中,所以静脉给药的化疗并不能叫做靶向血液的药物。同样胞内释放的ADC毒素也通常会在杀伤释放毒素的肿瘤细胞前扩散到微环境,这也是瘤内注射化疗并没有更好控制肿瘤的原因之一。如果释放毒素的细胞被杀死后毒素才能继续杀伤其它细胞,那么抗原阳性细胞应该优先被清除、剩余的肿瘤细胞应该以靶点阴性为主。但在Enhertu的DAISY临床试验中产生应答患者进展后HER2表达只有微小下降,很多患者HER2水平没有变化、有个别患者HER2水平还上升了,这种顶风作案的行为说明DXd在微环境里进行了无差别杀伤。二是ADC与抗原结合只是实现靶向递送的一个步骤,后面还需要其它步骤的配合才能实现这个递送步骤的价值。比如内吞要足够快、能进入溶酶体、链接子能被切断、毒素能够逃出溶酶体等,否则细胞有一系列运营机制能把ADC赶出去,如FcRn介导的抗体循环。所以ADC与抗原结合是个比较脆弱的联姻,后面很多柴米油盐的琐事可以让这个唯一能实现选择递送的事件失效。作为小结ADC虽然设计是分子靶向的递送技术,但在临床治疗窗口的引导下已经定向演化成一个组织水平的递送技术,无论疗效和毒性都是组织水平的、由组织的游离毒素AUC或Cmax决定。当然不可过膜毒素或不可裂解ADC另当别论,但这种ADC已经越来越少。这个宏观图像对ADC靶点选择和分子设计有着深远的影响。2ADC不是魔法子弹、而是魔法扳机ADC被称作魔法子弹,意思是接上抗体后的化疗药物可以准确杀伤目标细胞。但这是基于抗体如果能特异性与抗原结合,毒素就能特异性杀伤抗原阳性细胞这个假设。实际上抗体与抗原结合只是一小步,如果内吞比较慢那么ADC药物还会与抗原解离,即使能高效内吞如果链接子水解速度太慢ADC还会被FcRn给外排到胞外。肿瘤相对于正常组织只是一个极小区域,所以ADC更多时间还是与正常细胞打交道,而正常细胞也有内吞ADC、释放毒素的机制如Fc受体。尽管单个正常细胞因为不表达靶标抗原所以与肿瘤细胞比可能效率较低,但是数量上正常细胞占绝对优势。加上正常组织血管结构比肿瘤组织更完整,药物渗透更好,所以其实只有不到1%的ADC是在肿瘤里释放的。即使ADC能把毒素释放在肿瘤细胞里,能否完成选择性杀伤还需要毒素在靶标细胞里存在足够时间。小分子自由扩散能力很强、肿瘤细胞也会表达一些外排泵,所以对于DXd这样过膜性较好的毒素无论在哪个细胞中释放出来都要到操场集合、然后统一行动去杀伤微环境里的肿瘤细胞,像DM1这样过膜性较差的毒素工作机制可能不同。如果把ADC比喻成伞兵,不可过膜的T-DM1在落地细胞中完成杀伤然后就算完成任务, 而T-DXd则先在敌人后院降落、然后集体去行动,这样就可以同时杀伤靶点抗原阳性和阴性的肿瘤细胞、直到被人体排除。前面讲过这个机制在肿瘤微环境就是旁观者杀伤效应,在正常组织就是脱靶毒性。所以ADC真正能做到的是在一定程度上选择性在靶标阳性抗原细胞里释放毒素,其工作机制更像是一个魔法扳机、即在某些情况下能在肿瘤细胞里扣动扳机,至于是否能成为魔法子弹要看子弹飞多长时间、飞到哪里。两年前Zymeworks的科学家发表一篇影响很大的文章指出ADC与小分子毒素比绝对安全性并没有改善,即最高耐受剂量(MTD)并没有提高。按理说如果ADC只见到恐怖分子才开枪、吃瓜群众应该不用担心被误伤,而实际情况是同样火力下被误伤的比例与小分子毒素比一点没下降。不仅被误伤人数没有下降,被误伤的还都是同一类吃瓜群众、比如分裂较快的血液细胞无论面对ADC还是小分子毒素都是被薅羊毛的不幸群体。其实这个核心观点早在2015年FDA的科学家就已经提出,即无论靶点是什么ADC的毒性强弱及毒性谱基本由毒素决定。究其原因还是子弹虽然主要在肿瘤细胞里射出,但飞的时间太长、最后在哪里产生杀伤还是由每个compartment的毒素AUC决定。尽管绝对安全性没有改善,但不等于ADC没有附加收益。同样是毒素血药浓度达到MTD,ADC与小分子毒素在肿瘤微环境里的毒素浓度可能根据抗原表达量不同而有不同程度差异、因此绝对疗效可能是有改善的。ADC与小分子毒素比具体在肿瘤里面富集了多少是不太容易研究的,尤其是在真正患者肿瘤中的富集。疗效不能作为毒素蓄积的代替指标,一是不同肿瘤对同一毒素的敏感度不同,抗原高表达肿瘤可能对毒素更敏感或更耐受。二是难以去掉抗体端的影响,对于靶点高表达肿瘤抗体本身也会介导一些免疫杀伤、与毒素的叠加效应不好量化。这跟第三个因素即缓释掺和在一起,因为ADC是个缓释毒素机制所以本身也会增加一定治疗窗口。一个办法是用各种显影剂接在抗体上看看显影剂留在哪些地方,从而倒推毒素的可能释放机制、但这些技术很难精确到细胞类型。有些研究确实精确到细胞类型了,但结果有时却比较尴尬。比如一个曲妥珠显影剂在HER2阳性实体瘤里面主要与巨噬细胞结合、并没有去找HER2,高度特异的曲妥珠为何成了渣男有多种可能。当然你可以直接策略肿瘤组织里面的药物浓度,这也有几个干扰因素。一是如果测总毒素浓度你得考虑不同组织的药物蛋白结合率,即有些小分子毒素如MMAE相对于血液在肿瘤组织虽有蓄积但是通过与蛋白非特异结合、游离药物并没有富集。还有就是小分子容易从肿瘤逃逸,所以即使很多在肿瘤释放,但逃逸太快也会导致AUC下降,这对快速杀伤或慢解离(slow Koff)毒素是个干扰。今年阿斯利康的科学家发表了一个Enhertu在小鼠模型的药物释放数据,Enhertu释放DXd的AUC在HER2高表达肿瘤(N87,每个细胞表达350万HER2)为493,而在HER2低表达MB468(每个细胞表达4800 HER2受体)则为156。HER2从导航能力看应该说是ADC靶点的天花板,强阳性肿瘤与几乎阴性肿瘤比三倍的毒素富集反映了目前ADC设计框架下这个递送机制的极限。三倍富集虽然没有达到很多人的预期,但是对抗热力学分配规律如同逆水行舟并不是一件容易的事情。化疗的窗口通常非常小,增加3倍还是一个非常显著的提升。加上抗体端起到了一定辅助作用、尤其是靶点高表达人群和ADC的毒素缓释机制,令Enhertu无论与曲妥珠还是伊利替康比都显著提高了治疗效果。但是ADC也是一类设计、生产、开发都高度复杂的药物,这三倍的富集是否值得如此繁琐的设计、能否通过更简单的设计实现?如果愿意承担与抗体偶联的高昂代价,我们需要重新思考如何能真正实现大水漫灌微环境、显著优于更简单的分子构建模式。ADC已经到了Deming所说的survival isn’t mandatory时刻,或者reinvent自己、或者被其它模式取代。这两个选择都是新的机会。3IO-化疗、甚至IO-IO组合对魔法子弹设计者来说比魔法扳机更伤自尊的说法是你这武器不过是火药和枪把子的组合、瞄准和导航系统没你想的那么重要,但是真正成功的ADC确实是同时借助了抗体端和毒素端的协同或叠加效应。这种组合效应是肿瘤治疗自古至今所依赖的,有着复杂的背景。肿瘤药物开发最大障碍是肿瘤的异质性难题,如果每个肿瘤细胞结构功能都一样、与药物关系都一样那么治愈晚期肿瘤可能早就是现实了。肿瘤既有基因异质性,即每个肿瘤细胞基因组不同、表达关键蛋白结构和数量各不相同;也有时空异质性,即在不同发展阶段、肿瘤不同区域的肿瘤细胞中蛋白表达也不同。药物可浸润区域也在每个病灶都不一样、加上肿瘤干细胞的存在,导致晚期肿瘤治疗几乎是一个不可完成的任务。因为肿瘤异质性所以绝大多数疗法只能杀伤部分肿瘤,靶向疗法难以杀伤靶点阴性肿瘤,免疫疗法没有微环境中免疫警察的支持也是光杆司令。小分子选择性差,大分子不能有效浸润微环境。即使化疗也难以杀伤肿瘤干细胞、不能有效防止肿瘤再生。因此组合疗法是几乎所有肿瘤治疗的通用策略。比较干净的靶向疗法即便组合起来也只有在少数特殊场景下可以有效控制晚期肿瘤,现在绝大多数治疗方案都是有化疗参与的。多数靶向疗法要与化疗联用才真正能显示治疗价值,任何新的治疗方案都还需要有点化疗的影子。化疗药物可以大量杀伤一批死硬分子、当然代价是会误伤一些无辜群众,也可以在肿瘤细胞和肿瘤微环境制造足够混乱、把黑帮份子从地堡赶到外面,为更精准温和的现代疗法靶向杀伤提供便利、因此提供一个疗效基础。化疗药物的发现和开发虽然选拔机制是看杀伤能力,但是在真正患者体内却不一定是通过直接杀伤。多数在临床显示足够治疗窗口的化疗药物在耐受剂量下其实并不能直接杀伤肿瘤细胞,因为患者体内的肿瘤比实验室里的肿瘤长得要慢很多。一个假说是化疗药物在达到杀伤剂量之前已经会在肿瘤细胞内造成极大混乱、从而激活了免疫系统间接杀伤肿瘤。这如同一个杀手到了黑帮聚会的酒馆去清理黑帮,他虽然是有战斗能力的、但在人数处于劣势的时候可以在酒馆里制造混乱吸引警察来围剿这些以前没有被警察发现的黑恶势力。江湖上早有传闻化疗都有免疫疗法的成分,现在IO-ADC组合也是临床开发的一个重要方向。抗体药物则更要借助免疫系统,多数抗体药靶点只是在肿瘤细胞表达较高、不一定参与肿瘤发展的重要决策,当然表达高于正常细胞本身也是个令人生疑的迹象、可能参与的比较隐蔽。即使像HER2这样与肿瘤生长高度相关的靶点,HER2抗体阻断信号通路本身对肿瘤影响也很有限,更多是通过ADCC、ADCP等免疫介导效应。所以ADC在肿瘤微环境里既有抗体介导免疫杀伤、也有化疗的直接杀伤或间接介导的免疫杀伤,至少在某些场景下模拟一个化疗与抗体药物的组合。当然有些ADC的MTD较低,抗体端达不到治疗剂量、但成功的ADC都达到抗体有效剂量。有些ADC的抗体端根本没有明显单方活性,但是在肿瘤微环境的局部药物组合还是可能产生治疗效果的。上个世纪就有研究显示一个药物在远低于治疗剂量时就可以增敏另一个药物的治疗效果,这也是小分子双靶点药物设计的一个重要理论根据,所以ADC抗体端的贡献不能轻易忽视。前面讲过多数ADC在多数场景应答与抗原表达量相关性较差、同一毒素做成靶点分布大相径庭的不同ADC毒性和毒性谱非常接近,说明靶点介导生物学效应主要不在细胞水平体现、而是在组织水平体现。有些ADC显示了一定靶点相关应答和疗效、如Enhertu,即HER2高表达患者应答更好。但这是因为HER2高表达患者肿瘤毒素释放吞吐量更高,还是因为对曲妥珠介导的免疫杀伤更敏感并不太容易区分。动物模型中HER2高阳性肿瘤毒素蓄积比几乎无表达肿瘤高几倍,但患者肿瘤可能更复杂、结果可能不同。HER2显影剂在一定程度能区分阳性和阴性病灶,但是假阳性、假阴性率还是很高,达不到商业应用水平。另外前面讲过抗体在抗原阳性细胞富集不等于就能在这类细胞释放毒素、释放了也不一定停留足够长时间。Enhertu已经批准用于所有HER2阳性实体瘤治疗,但是在HER2阳性乳腺癌70%应答率与HER2阳性胰腺癌5%的应答率相差巨大。这似乎更与曲妥珠在这两类肿瘤的基础疗效而不是导航Enhertu能力相关,因为二者都是HER2阳性,当然两种肿瘤对DXd的敏感度可能不同、毒素释放能力可能也不同。肿瘤治疗从来都依靠以化疗为支柱的组合疗法,ADC是唯一既有化疗广谱杀伤又有抗体靶向杀伤的治疗板块。因此ADC是唯一能对肿瘤发起立体战争的单方药物,与抗癌多年来的组合疗法经验不谋而合。遗憾的是ADC的抗体靶点很多没有单抗药物上市,一个原因可能是这些抗体也需要合适的化疗组合才能真正起效,而上市化疗药物或者机制与之不匹配、或者药代动力学不平行,导致组合命中率较低。事实上实体瘤有一定单方活性的抗体药物靶点十分有限,我数了数只有EGFR, HER2, PDL1, CLDN18.2, VEGFR2, PDGRFα, EpCAM, GD2有上市药物,多数还是较小产品,所以任何有点单方活性的抗体都应该做成ADC看看成药性。ADC火热一个后果是在临床验证了哪些毒素是优质抗体组合伙伴,比如DXd接到多个抗体似乎效果都不错、至少在缓释条件下,这些宝贵数据为快速验证新靶点ADC提供了技术支撑。4Curtin-Hammett规则ADC设计的·一个基本假设是抗体与靶点抗原结合后会启动杀伤过程,如果ADC只与靶点结合那么ADC就只杀伤表达靶点的细胞,如果只有肿瘤表达靶点那么ADC就只杀伤肿瘤细胞。当然实际执行中这个逻辑链条经常支离破碎,人体没有只在肿瘤细胞表达的抗原、肿瘤表面抗原也不是都能被ADC药物找到。不表达抗原的正常细胞也能内吞大量ADC、释放毒素,即使在肿瘤微环境内有多少ADC毒素是通过抗原阳性肿瘤细胞释放的也存在很多疑问。这些不可告人的little dirty secrects我们以后慢慢讲,今天讲一下即使非肿瘤细胞不干扰ADC结合和毒素释放,ADC要完成肿瘤特异杀伤也是天时地利人和缺一不可的。化学反应动力学为预测多步骤过程的最终结果铺垫了一些理论基础,其中一个叫做Curtin-Hammett规则的规律对理解ADC杀伤肿瘤细胞这个多步过程的最终结局尤其应景。这个规则用化学语言讲是指一个化学反应不同路径的产物比例是由各路径的活化能决定,而与每条反应路径的中间体浓度高低无关、前提是中间体之间转化速度较快。我来把它翻译成生活中的一个场景以帮助非化学专业人士理解。说一条街上开了甲乙两个餐馆,各雇了一个小哥在门口招揽顾客。甲餐馆的小哥更加能说会道所以90%的人流都被他引到自己的店里,但这是否意味着甲餐馆销售额一定比乙餐馆多呢?不一定,如果甲餐馆店内就餐环境脏乱差、服务员态度恶劣、菜品不佳,那么大多数顾客最后还是会在乙餐馆消费、因为乙餐馆就在旁边。当然如果甲餐馆在北京、乙餐馆在上海,这个规则就失效了、对应着中间体转换速度较慢的化学反应。ADC与靶点抗原结合是同一个道理。通常抗体与抗原结合速度很快、很多情况就是分子的扩散速度,但是结合后是否能内吞到细胞内就与每个抗原的生物功能和抗体的构建模式有关了。进了细胞也不是就能立刻把毒素卸载,而是要到一个降解酶比较丰富叫做溶酶体的特殊部门才能卸货,而每个细胞溶酶体里面的组织蛋白酶(多数ADC的链接子含有这类酶的识别多肽序列)数量不同、活性也不一样,所以链接子在不同肿瘤细胞水解速度不同。溶酶体停车时间是有限制的,如果水解速度太慢ADC分子会被细胞内抗体循环机制如FcRn运载到细胞外,还得重新排队才能回到溶酶体。溶酶体里面的蛋白酶也不仅只是降解链接子,你的毒素如果长的像多肽也会被按照废物处理掉。以前有人用Velcade作为ADC毒素,结果在溶酶体里面被当作多肽给打碎处理了。即使你的ADC不用反复排队、顺利在靶点所在细胞释放了所有毒素,这也并不等于刺杀任务结束、可以去领赏金了,因为相对于逃离肿瘤细胞、肿瘤杀伤是个慢功夫。毒素释放后与抗体就彻底失去联系,基本完全随机扩散、放飞自我了。有些毒素因为过膜速度很慢可能长时间留在肿瘤细胞内、所以会有足够时间杀伤这个肿瘤细胞,但是代价是只能杀伤这个肿瘤细胞、因为不能进入其它细胞。现在的ADC毒素大多数可以产生所谓的旁观者杀伤效应、即可以自由进出细胞膜,这类毒素进出细胞膜的时间以秒计算。但是肿瘤细胞杀伤在体外也是需要几天的,患者肿瘤因为长的慢很多,所以临床最敏感的肿瘤也需要几周才能看到反应。因此毒素离开肿瘤细胞与杀伤肿瘤肿瘤完全不在一个时间尺度上,除了极高活性配体或者不可逆配体(解离常数Koff非常大,所以与靶点解离需要很长时间)不需要毒素暴露水平要长时间保持在一定水平外,多数小分子毒素即使在目的地释放也会在执行任务之前作鸟兽散。和所有靶向递送技术平台一样, ADC也是希望能在肿瘤微环境长时间保持高于系统药物暴露的药物浓度,但这是违反热力学第二定律的。要远离热力学平衡就必须有能量持续输入,这个能量输入就是抗体与抗原的结合能。不过这只是一个必要但不充分条件,通过上面分析可以看出这点有限的结合能使用效率与很多因素有关,如果使用不当很容易就成为月光族。同样靶点与ADC结合也是必要但不充分条件,如同提高结婚率不等于提高人口出生率,后者还与很多其它因素有关。5魔法子弹还是子弹ADC虽然比化疗药物大很多、也复杂很多,但本质还是一个化疗药物。虽然作为司机的抗体部分在某些情况下可能起到暗中保驾护航的作用,但杀伤还是主要看毒素部分,魔法子弹也还是子弹。尽管ADC这个概念由来已久、也很快产生了像T-DM1这样的重磅产品,但是在Enhertu横空出世之前ADC并没有受到主流药厂的重视。2020年路罗氏CEO对说:"We have shifted our technology priorities," Roche CEO Severin Schwan told Reuters. "Maybe others will be luckier, but we failed to master the complexity."罗氏在抗癌药领域的地位类似手机领域的苹果,当时已经有T-DM1和Polivy两款重磅ADC上市,罗氏老大说话业界还是会认真听的。回头看当时应该说是万事俱备只欠一个优质毒素,这个优质毒素就是DXd。第一三共做了多年的喜树碱衍生物,并上市了伊利替康。伊利替康是SN38的前药,虽然也有不错的疗效但与紫杉醇、铂类药物比还是有差距。一三共后来发现一个活性更好的类似物exatecan,但是毒性也更大、临床试验做了不少也没找到一个可用的适应症。当时一三共想把exatecan做成高分子偶联前药,并把一个叫做DE310的高分子前药推进临床。这个前药释放exatecan十分平缓,但是还是无法实现足够治疗窗口。后来ADC进入大家视野,一三共就把DE310的经验平移到了ADC设计,包括DE310用的GGFG链接子。连Enhertu最早的专利都是把exatecan作为毒素:Exatecan虽然比SN38活性更好,但是作为ADC毒素还是算是比较弱的、这个缺点可以用高DAR值来解决。Exatecan有个胺基可以直接与GGFG通过酰胺键偶联,但是这样偶联酶切时会有一部分在G-F之间断裂、释放G-exatecan。加上一些理化性质的考虑,一三共科学家在exatecan加上一个羟乙酰胺基、即现在大名鼎鼎的DXd(deruxtecan)。在此之前GGFG如何与羟基化合物偶联已经是已知技术,所以做成Enhertu并不复杂。DXd比exatecan还要弱5倍左右,所以与当时的主流ADC设计理念并不一致,当然现在已经没人再计较DXd的体外活性问题。Enhertu的成功多少有点意外成分,这个从一三共过去20年的股票可以看出来。至于Enhertu到底为何如此成功、尤其是在HER2 ultralow乳腺癌也有显著疗效现在业界还在研究,但是估计主要是DXd有某些与众不同的超能力。因为现在至少有10款使用其它毒素的HER2 ADC产品在临床试验阶段失败,其它更致命的弹头如CAR-T、CAR-M、免疫毒素等也全军覆没。而DXd目前的五个不同靶点ADC各个价值连城、以总交易额350亿美元分别转让给阿斯利康和默沙东。至于神秘剑客DXd到底有哪些特异功能估计还需要很长时间才能搞清楚,可能仅仅是安全窗口比exatecan高、本身就是一个优质小分子化疗药物,但遗憾的是DXd从来没有进入临床研究、所以没有头对头比较数据。也可能是在非杀伤剂量产生某种免疫激活效应,或者直接产生ICD(免疫原性细胞死亡)、或者增敏了抗体的ADCC等免疫效应。DXd的半衰期较短,这对于小分子药物来说是个劣势,但对于ADC毒素来说是个优势、因为毒素进入血液循环经过肝肾等代谢排泄器官会很快被排除,维持一个肿瘤组织与血液系统的药物梯度。或许是这些因素的组合,现在公开数据有限、所以难以判断。现在的新药发现是一个超级复杂体系,目的是通过一些列所谓去风险筛选程序降低临床失败风险、避免到临床试验神农尝百草。但遗憾的是真正首创新药基本还是通过临床试验筛出来的,理性药物设计还是一个靠天吃饭的行当。即使所谓的me-too跟踪药物可能会容易一点但也要看具体情况,比如紫杉醇、质子泵抑制剂、沙利度胺类分子胶、波立维类P2Y12受体激动剂等重要药物虽然有所谓的跟踪药物,但与首创药物只有非常微小的结构差异。说明机制本身不足以筛到优质药物,否则早有人去筛新结构类型了。很多用了几十年的主流抗癌药其实到现在我们也不清楚真正的工作机制,比如紫杉醇在临床有效剂量下胞内的药物浓度不足以抑制微管蛋白解离,其它重要药物如铂类、他莫昔芬也是一样。DXd只是最新的一桩悬案,当然这样的悬案越多越好。6谁在施展魔法?ADC本身没有活性(除了抗体介导的免疫杀伤外)、只有毒素被释放出来才有肿瘤杀伤活性,但是到目前为止体内、尤其是患者体内毒素释放的细节还比较模糊。虽然ADC的设计是通过抗原阳性肿瘤细胞内吞后释放毒素,但是蛋白水解酶不仅存在于肿瘤细胞中、甚至肿瘤细胞并非主要来源。ADC链接子主要是通过组织蛋白酶水解,但也可能成为其它蛋白酶底物。所以谁在背后施展魔法并非是一个泾渭分明的问题。抗体药物虽然有了30年的历史,但是我们对抗体在人体降解过程的了解仍然存在很多空白。同位素标记研究发现小鼠代谢降解抗体效率最高的器官为肝、脾、肾,但因为皮肤、肌肉组织更多所以降解抗体蛋白绝对量最大的器官为肝、皮肤、肌肉,清除约75%的抗体。很多完全肿瘤患者体重下降严重,有假说认为降解肌肉机制失调与抗体药物降解提高是同一套机制。一般认为网站内皮吞噬系统是主要的降解抗体机制,单核细胞、巨噬细胞为主要细胞类型。因为天然生物功能就是吞噬降解废弃蛋白和其它生物物质,所以这个系统吞吐量较大、内吞和降解能力都很强,而这些性质也是ADC毒素释放所需要的。肿瘤微环境里面也有大量肿瘤相关巨噬细胞(TAM),通常已经被肿瘤收编成为肿瘤增长的帮凶。TAM绝对量很大、是微环境中数量最大免疫细胞,在有些病灶甚至超过肿瘤细胞。与肿瘤细胞相互链接成为一个紧密组织不同,TAM可以在微环境中自由活动,因为在成为帮凶之前这些细胞需要在所处组织巡逻以清除废旧蛋白。巨噬细胞根据化学信号寻找肇事份子,所以也有人用巨噬细胞作为化疗药物载体、利用它识别肿瘤等非正常组织能力把药物递送到疾病组织。药物可以在体外装进巨噬细胞,也可以在体内利用巨噬细胞贪吃的本性粘在巨噬细胞上、叫做in vivo hitchhiking。大家可能比较熟知前几年比较火热的红细胞递送药物,巨噬细胞递送只是没有那么高调。巨噬细胞表达各种Fc受体,可以与IgG抗体Fc区结合并内吞整个抗体。抗体的一个主要杀伤机制抗体介导细胞吞噬(ADCP)就是利用Fc受体结合抗体内吞这个功能,如果可变区与肿瘤细胞结合则整个复合物被吞噬。虽然FcR内吞与Fc构象和密度有关(与肿瘤细胞表面抗原结合后改变),单个抗体也是可以被巨噬细胞通过Fc受体内吞的。虽然Fc受体与Fc亲和力不如抗体抗原结合力强,但是因为TAM在微环境中数量巨大、又整天闲逛,所以对抗体药物影响还是很大。PD-1抗体与PD-1短暂结合后会逐渐转移到巨噬细胞表面,成为耐药的一个机制。PD-1也是在免疫细胞表达的受体,而ADC靶向的肿瘤表面受体因为暴露更低所以可能受巨噬细胞影响更大。既然TAM可以影响单抗的代谢那么ADC也可能通过TAM降解。Seattle Genetics早在2017年发现有些ADC药物虽然靶向的是肿瘤抗原,但在微环境中却主要是通过巨噬细胞介导内吞释放的毒素、靶点介导释放跟不上巨噬细胞的节奏。后来陆续有人研究发现如果清除宿主的巨噬细胞则ADC活性显著下降,如果用单抗阻断ADC与肿瘤细胞结合虽然微环境中ADC水平下降但毒素释放却没受影响、但如果静默抗体的Fc则毒素释放大幅度减慢。TAM能这样显著影响ADC降解不仅与其数量和内吞能力有关,TAM的水解酶水平因为工作需要也很高。有人用ABPP化学标记追踪微环境中活性组织蛋白酶的来源,发现至少在某些肿瘤微环境中90%以上的组织蛋白酶来自巨噬细胞而不是肿瘤细胞。当然这些观测只是在特定实验环境下进行的,但这些数据指向微环境中ADC也不完全只与肿瘤细胞接触。动物模型毕竟与人体肿瘤有本质区别,遗憾的是ADC在人体肿瘤的代谢和分布数据很少,其中一半的知识可能是来自Enhertu的DAISY二期临床试验。这个试验不仅第一次发现Enhertu在HER2极低表达患者也有相当应答率(最近在DB06试验中得到验证),而且作者做了非常详细的疗效和耐药机制研究。其中有关PK/PD关系研究发现HER2表达与Enhertu在肿瘤的蓄积关系不明显,肿瘤微环境Enhertu水平低也不排除应答可能。作者没有测量毒素DXd的水平、更没有测游离DXd的水平,所以没有严格的PK/PD关联数据。耐药患者的HER2水平通常只有微小下降或没有下降,有几位患者耐药后发现HER2反而上调了。这些数据令人怀疑HER2之外还有神秘力量参与了Enhertu的毒素释放。抗体是经高度演化、功能繁多的生物分子,毒素经过多年临床前、临床筛选已经淘汰了多数“劣质毒素”。ADC是一类高度复杂的药物,我们对这类药物的理解还在不断加深。这个系列短文如盲人摸象试图从不同侧面勾画ADC的图像,链接所有这些数据点您看到的是一类怎样的抗癌药物?【转载声明】本文转载自“美中药源”微信公众号。更多优质内容,欢迎关注↓↓↓
抗体药物偶联物
100 项与 溴匹立明 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 膀胱癌 | 申请上市 | 美国 | - | |
| 膀胱癌 | 申请上市 | - | - | |
| 单纯疱疹 | 临床3期 | 美国 | - | |
| 淋巴瘤 | 临床3期 | 美国 | - |
登录后查看更多信息
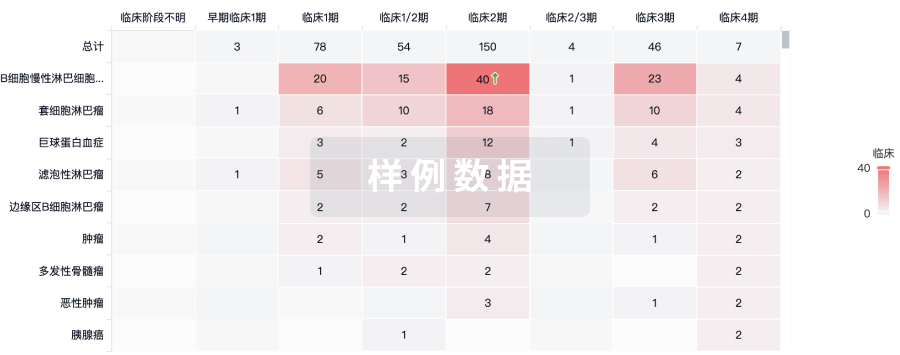
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
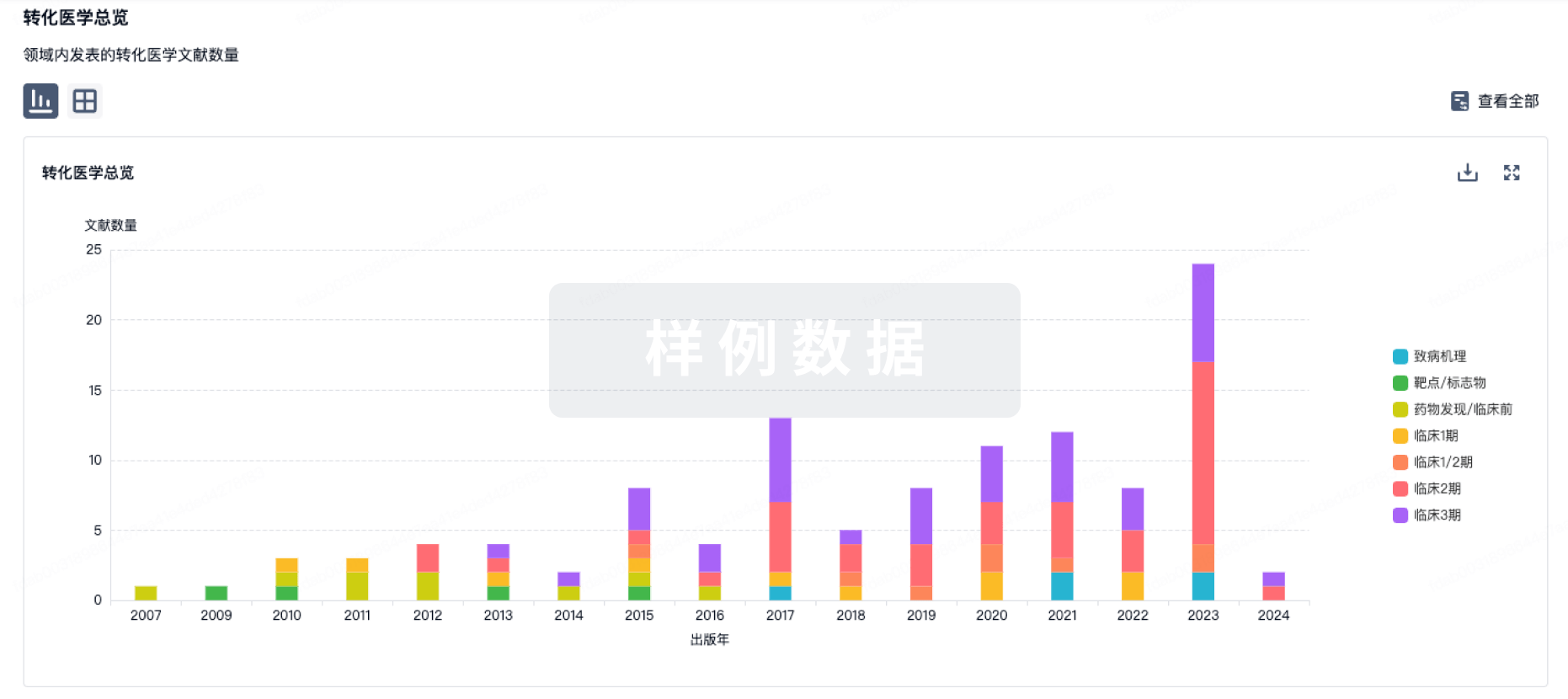
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
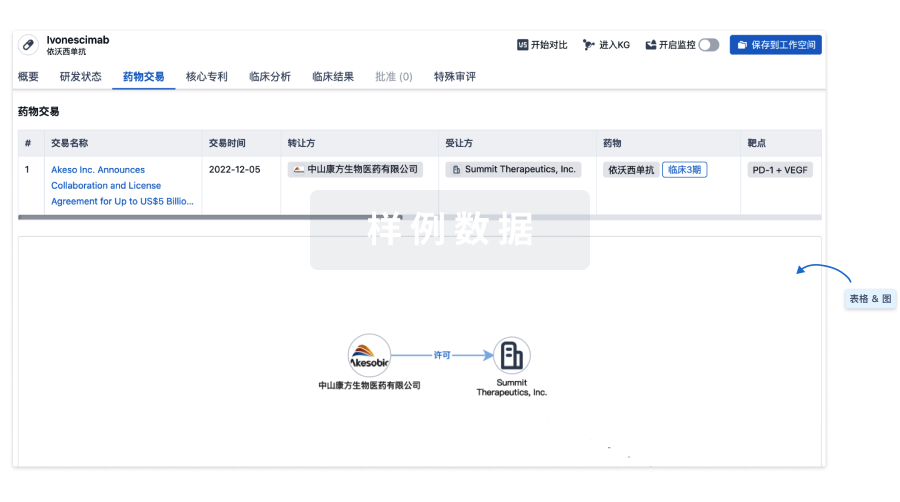
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
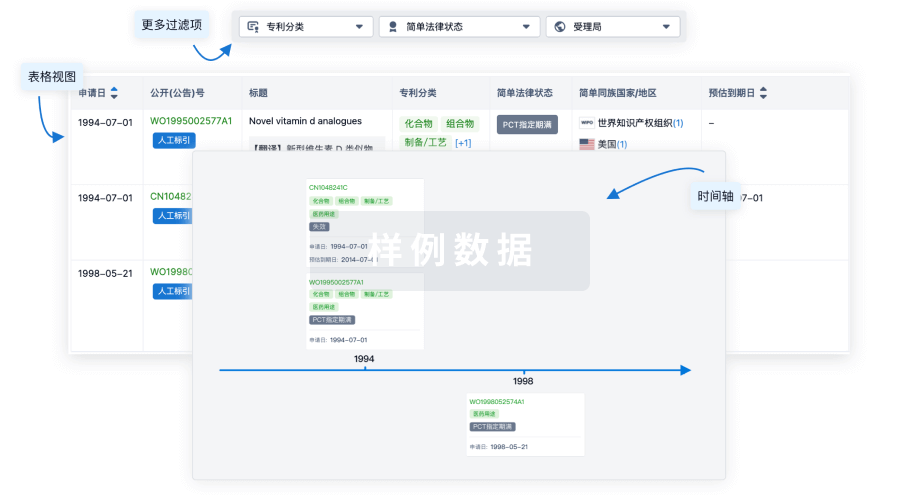
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


