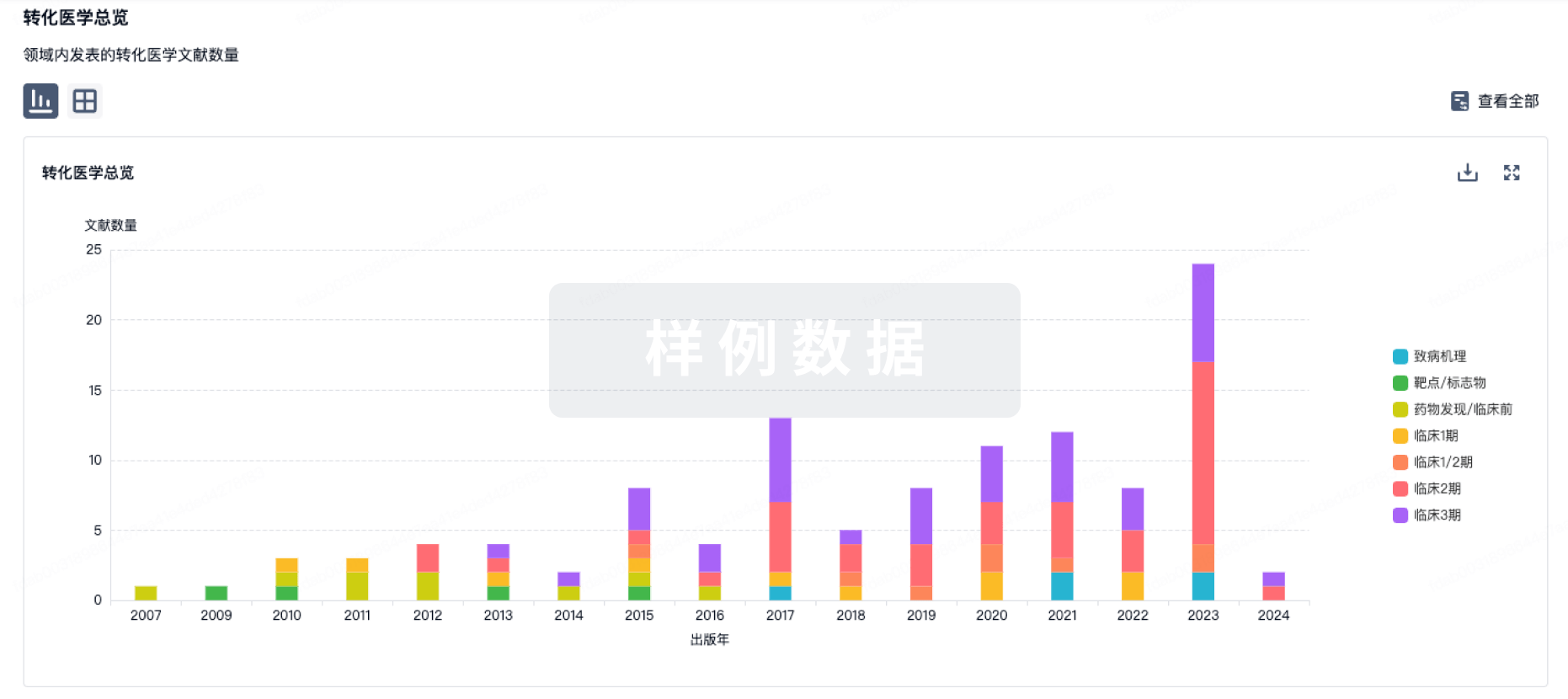
Bunitrolol (BTL), a β-adrenoceptor blocking agent, is predominantly biotransformed to 4-OH BTL in rat liver microsomes.Extensive studies in the rat and human have indicated that hydroxylation of other β-adrenoceptor blocking agents, for example propranolol 4-hydroxylation and bufuralol 1'-hydroxylation, are catalyzed by debrisoquine (DB) 4-hydroxylase.These findings suggest the involvement of DB 4-hydroxylase in BTL metabolismTo examine this possibility, the authors investigated BTL 4-hydroxylase activities in liver microsomes of adult Wistar and Dark Agouti rats of both sexes.Sex and strain differences of DB 4-hydroxylation in both strains of rats were very similar to those of the BTL oxidationThese results suggest that P 450 isoenzyme(s) catalyzing DB 4-hydroxylation mediates also BTL 4-hydroxylation in rat liver microsomes.However, enzymic studies using purified P 450 isoenzyme(s), namely, BTL 4-hydroxylase, are necessary to prove this possibility.The authors are now performing experiments along this line.